Abstract
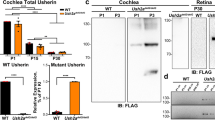
Hearing loss is the most common sensory deficit in humans. Because the auditory systems of mice and humans are conserved, studies on mouse models have predicted several human deafness genes and identified new genes involved in hearing1,2. The deafwaddler (dfw) mouse mutant is deaf and displays vestibular/motor imbalance. Here we report that the gene encoding a plasma membrane Ca2+-ATPase type 2 pump (Atp2b2 , also known as Pmca2) is mutated in dfw. An A→G nucleotide transition in dfw DNA causes a glycine-to-serine substitution at a highly conserved amino-acid position, whereas in a second allele, dfw2J, a 2-base-pair deletion causes a frameshift that predicts a truncated protein. In the cochlea, the protein Atp2b2 is localized to stereocilia and the basolateral wall of hair cells in wild-type mice, but is not detected in dfw2J mice. This indicates that mutation of Atp2b2 may cause deafness and imbalance by affecting sensory transduction in stereocilia3 as well as neurotransmitter release from the basolateral membrane4. These mutations affecting Atp2b2 in dfw and dfw2J are the first to be found in a mammalian plasma membrane calcium pump and define a new class of deafness genes that directly affect hair-cell physiology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Steel, K.P. & Brown, S.D.M. Genetics of deafness. Curr. Opin. Neurobiol. 6, 520–525 (1996).
Petit, C. Genes responsible for human hereditary deafness: symphony of a thousand. Nature Genet. 14, 385–391 ( 1996).
Yamoah, E.N. et al. Plasma membrane Ca2+-ATPase extrudes Ca2+ from hair cell stereocilia. J. Neurosci. 18, 610–624 (1998).
Tucker, T. & Fettiplace, R. Confocal imaging of calcium microdomains and calcium extrusion in turtle hair cells. Neuron 15, 1323–1335 (1995).
Lane, P.W. New mutants and linkages: deafwaddler (dfw). Mouse News Lett. 77, 129 (1987).
Street, V.A., Robinson, L.C., Erford, S.K. & Tempel, B.L. Molecular genetic analysis of distal mouse chromosome 6 defines gene order and positions of the deafwaddler and opisthotonos mutations. Genomics 29, 123–130 (1995).
McKee-Johnson, J.W., Street, V.A., Erford, S.K., Robinson, L.C. & Tempel, B.L. Physical and genetic maps of the deafwaddler region on distal mouse chromosome six. Genomics 49, 371–377 (1998).
Noben-Trauth, K., Zheng, Q.Y., Johnson, K.R. & Nishina, P.M. mdfw: a deafness susceptibility locus that interacts with deafwaddler (dfw) . Genomics 44, 266–272 (1997).
Gao, J. et al. Cloning and characterization of a mouse gene with homology to the human von Hippel-Lindau disease tumor suppressor gene: implications for the potential organization of the human von Hippel-Lindau disease gene. Cancer Res. 55, 743–747 (1995).
Kuzmin, I. et al. One-megabase yeast artificial chromosome and 400-kilobase cosmid-phage contigs containing the von Hippel-Lindau tumor suppressor and Ca2+-transporting adenosine triphosphate isoform 2 genes. Cancer Res. 54, 2486–2491 (1994).
Carafoli, E. & Stauffer, T. The plasma membrane calcium pump: Functional domains, regulation of the activity, and tissue specificity of isoform expression. J. Neurobiol. 25, 312 –324 (1994).
Stahl, W.L. et al. Plasma membrane Ca2+-ATPase isoforms: distribution of mRNAs in rat brain by in situ hybridization. Molec. Brain Res. 16, 222–231 (1992).
Crouch, J.J. & Schulte, B.A. Expression of plasma membrane Ca-ATPase in the adult and developing gerbil cochlea. Hear. Res. 92, 112–119 ( 1995).
Crouch, J.J. & Schulte, B.A. Identification and cloning of site C splice variants of plasma membrane Ca-ATPase in the gerbil cochlea . Hear. Res. 101, 55–61 (1996).
Andersen, J.P., Vilsen, B., Leberer, E. & MacLennan, D.H. Functional consequences of mutations in the ÿ-strand sector of the Ca2+-ATPase of sarcoplasmic reticulum. J. Biol. Chem. 264, 21018–21023 (1989).
Ghislain, M., Schlesser, A. & Goffeau, A. Mutation of a conserved glycine residue modifies the vanadate sensitivity of the plasma membrane H+-ATPase from Schizosaccharomyces pombe. J. Biol. Chem. 262, 17549–17555 (1987).
Apicella, S. et al. Plasmalemmal ATPase calcium pump localizes to inner and outer hair bundles . Neurosci. 79, 1145–1151 (1997).
Denk, W., Holt, J.R., Shepherd, G.M.G. & Corey, D.P. Calcium imaging of single stereocilia in hair cells: localization of transduction channels at both ends of tip links. Neuron 15, 1311 –1321 (1995).
Lumpkin, E.A., Marquis, R.E. & Hudspeth, A.J. The selectivity of the hair cell's mechanoelectrical-transduction channel promotes Ca2+ flux at low Ca2+ concentrations . Proc. Natl Acad. Sci. USA 94, 10997– 11002 (1997).
Eatock, R.A., Corey, D.P. & Hudspeth, A.J. Adaptation of mechanoelectrical transduction in hair cells of the bullfrog's sacculus. J. Neurosci. 7, 2821–2836 (1987).
Norton, S.J., Tempel, B.L., Steel, K.P. & Rubel, E.W. Physiological and anatomical status of the deafwaddler (dfw) mutant mouse cochlea. Assoc. Res. Oto. Abstr. 19, 82 ( 1996).
McKee-Johnson, J. & Reeves, R.H. Fragmentation and integrative modification of yeast artificial chromosomes. in Protocols for Yeast Artificial Chromosomes . Methods in Molecular Biology (ed. Markie, D.) 167 –186 (Humana Press Inc., Totowa, 1996 ).
Noben-Trauth, K., Naggert, J.K., North, M.A. & Nishina, P.M. A candidate gene for the mouse mutation tubby. Nature 380, 534–538 (1996).
Acknowledgements
We thank E. Rubel, S. Sullivan and D. Wu for comments; and D. Cunningham, N. M. del Puerto and L. Robinson for expert technical assistance. This work was supported by grants from the NIH and the NIDCD Division of Intramural Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Street, V., McKee-Johnson, J., Fonseca, R. et al. Mutations in a plasma membrane Ca2+-ATPase gene cause deafness in deafwaddler mice. Nat Genet 19, 390–394 (1998). https://doi.org/10.1038/1284
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/1284
This article is cited by
-
Biallelic ATP2B1 variants as a likely cause of a novel neurodevelopmental malformation syndrome with primary hypoparathyroidism
European Journal of Human Genetics (2024)
-
Identification and characterisation of spontaneous mutations causing deafness from a targeted knockout programme
BMC Biology (2022)
-
De novo and inherited loss-of-function variants of ATP2B2 are associated with rapidly progressive hearing impairment
Human Genetics (2019)
-
Structure of the human plasma membrane Ca2+-ATPase 1 in complex with its obligatory subunit neuroplastin
Nature Communications (2018)
-
Neuronal calcium signaling: function and dysfunction
Cellular and Molecular Life Sciences (2014)