Abstract
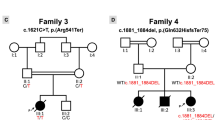
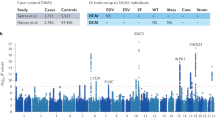
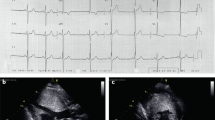
Longitudinal evaluation of a seven generation kindred with an inherited conduction system defect and dilated cardiomyopathy demonstrated autosomal dominant transmission of a progressive disorder that both perturbs atrioventricular conduction and depresses cardiac contractility. To elucidate the molecular genetic basis for this disorder, a genome–wide linkage analysis was performed. Polymorphic loci near the centromere of chromosome 1 demonstrated linkage to the disease locus (maximum multipoint lod score = 13.2 in the interval between D1S305 and D1S176). Based on the disease phenotype and map location we speculate that gap junction protein connexin 40 is a candidate for mutations that result in conduction system disease and dilated cardiomyopathy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wynne, J. & Braunwald, E. The cardiomyopathies and myocarditides: toxic, chemical, and physical damage to the heart. In Heart Disease (ed. Braunwald, E.) 1394–1450 (Saunders, Philadelphia, 1992).
Codd, M.B., Sugrue, D.D., Gersh, B.J. & Melton, L.J. III. Epidemiology of idiopathic dilated and hypertrophic cardiomyopathy: a population-based study in Olmsted County, Minnesota, 1975–1984. Circulation 80, 564–572 (1989).
Manolio, T.A. et al. Prevalence and etiology of idiopathy dilated cardiomyopathy (Summary of a National Heart, Lung, and Blood Institute Workshop). Am. J. Cardiol. 69, 1458–1466 (1992).
Michels, V.V. et al. The frequency of familial dilated cardiomyopathy in a series of patients with idiopathic dilated cardiomyopathy. New Engl. J. Med. 326, 77–82 (1992).
Berko, B.A. & Swift, M. X-linked dilated cardiomyopathy. New Engl. J. Med. 316, 1186–1181 (1987).
Muntoni, F. et al. Deletion of the dystrophin muscle-promoter region associated with X-linked dilated cardiomyopathy. New Engl. J. Med. 329, 921–925.
Michels, V.V. et al. Dystrophin analysis in idiopathic dilated cardiomyopathy. J. med. Genet. 30, 955–957 (1993).
Channer, K.S., Channer, J.L., Campbell, M.J. & Rees, J.R. Cardiomyopathy in the Kearns-Sayre syndrome. Br. Heart J. 59, 486–490 (1988).
Graber, H.L. et al. Evolution of hereditary cardiac conduction and muscle disorder: a study involving a family with six generations affected. Circulation 74, 21–35 (1986).
Voss, E.G. et al. Familial dilated cardiomyopathy. Am. J. Cardiol. 54, 456–457 (1984).
Lynch, H.T. et al. Hereditary progressive atrioventricular conduction defect. A new syndrome? J.A.M.A. 225, 1465–1470 (1973).
Mestroni, L. et al. Clinical and pathologic study of familial dilated cardiomyopathy. Am. J. Cardiol. 65, 1449–1453 (1990).
Seldin, M.F., Prins, J.-B., Rodrigues, N.R., Todd, J.A. & Meisler, M.H. Mouse chromosome 3. Mamm. Genome 4, S47–S57 (1993).
Watkins, H. et al. A disease locus for familial hypertrophic cardiomyopathy maps to chromosome 1q3. Nature Genet. 3, 333–336 (1993).
Carrier, L. et al. Mapping of a novel gene for familial hypertrophic cardiomyopathy to chromosome 11. Nature Genet. 4, 311–314 1993.
Watkins, H. et al. Characteristics and prognostic implications of myosin missense mutations in familial hypertrophic cardiomyopathy. New Engl. J. Med. 326, 1108–1114 (1992).
Thierfeider, L. et al. A familial hypertrophic cardiomyopathy locus maps to chromosome 15q2. Proc. natn. Acad. Sci. U.S.A. 90, 6270–6274 (1993).
Calquist, J.F., Menlove, R.L., Murray, M.B., O'Connell, J.B. & Anderson, J.L. HLA Class II (DR and DQ) antigen associations in idiopathic dilated cardiomyopathy.Validation study and meta-analysis of published HLA association studies. Circulation 83, 515–527 (1991).
Limas, C. et al. T-cell receptor gene polymorphisms in familial cardiomyopathy: Correlation with anti-β-receptor autoantibodies. Am. Heart J. 124, 1258–1263 (1992).
NIH-CEPH Collaborative mapping group. Science 258, 67–86 (1992).
Weissenbach, J. et al. A second-generation linkage map of the human genome. Nature 359, 794–801.
Buetow, K.H. et al. Integrated genome wide maps constructed using the CEPH reference panel. Nature Genet. 6, 391–393 (1994).
Engelstein, M. et al. A PCR-based linkage map of human chromosome 1. Genomics 15, 251–258 (1993).
Irwin, M., Cox, N. & Kong, A. Sequential imputation for multilocus linkage analysis. Genetic Epidemiology 10, 483–488 (1993).
Beyer, E.C., Paul, D.L. & Goodenough, D.A. Connexin family of gap junction proteins. J. membrane Biol. 116, 187–194 (1990).
Bennet, M.V.L. et al. Gap junctions: New tools, new answers, new questions. Neuron 6, 305–320 (1991).
Willecke, K. et al. Six genes of the human connexin gene family coding for gap junction proteins are assigned to four different human chromosomes. Eur. J. Cell Biol. 53, 275–280 (1990).
Lathrop, G.M., Lalouel, J.M., Julier, C. & Ott, J. Strategies for multilocus linkage analysis in humans. Proc. natn. Acad. Sci. U.S.A. 81, 3443–3446 (1984).
Haefliger, J.-A. et al. Four novel members of the connexin family of gap junction proteins. J. biol. Chem. 267, 2057–2064 (1992).
Kanter, H.L., Saffitz, J.E. & Beyer, E.C. Cardiac myocytes express multiple gap junction proteins. Circ. Res. 70, 438–444 (1992).
Kanter, H.L., Laing, J.G., Beau, S.L., Beyer, E.C. & Saffitz, J.E. Distinct patterns of connexin expression in canine purkinje fibers and ventricular muscle. Circ. Res. 72, 1124–1131 (1993).
Peters, N.S., Green, C.R., Poole-Wilson, P.A. & Severs, N.J. Reduced content of connexin43 gap junctions in ventricular myocardium from hypertrophied and ischemic human hearts. Circulation 88, 864–875 (1993).
Reed, K.E. et al. Molecular cloning and functional expression of human connexin37, an endothelial cell gap junction protein. J. clin. Invest. 91, 997–1004 (1993).
Surawicz, B. et al. Task Force 1: standardization of terminology and interpretation. Am. J. Cardiol. 41, 130–145 (1978).
Feigenbaum, H. Echocardiography. in Heart Disease (ed. Braunwald, E.) 64–115 (Saunders, Philadelphia,1992).
Boehnke, M. Allele frequency estimation from data on relatives. Am. J. hum. Genet. 48, 22–25 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kass, S., MacRae, C., Graber, H. et al. A gene defect that causes conduction system disease and dilated cardiomyopathy maps to chromosome 1p1–1q1. Nat Genet 7, 546–551 (1994). https://doi.org/10.1038/ng0894-546
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0894-546
This article is cited by
-
Closing the ‘phenotype gap’ in precision medicine: improving what we measure to understand complex disease mechanisms
Mammalian Genome (2019)
-
Beyond membrane channelopathies: alternative mechanisms underlying complex human disease
Acta Pharmacologica Sinica (2011)
-
Sudden cardiac death in a patient with lamin A/C mutation in the absence of dilated cardiomyopathy or conduction disease
Clinical Research in Cardiology (2011)
-
Lamin A/C truncation in dilated cardiomyopathy with conduction disease
BMC Medical Genetics (2003)
-
The failing heart
Nature (2002)