Abstract
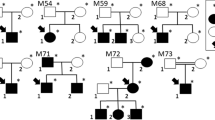
Malattia Leventinese (ML) and Doyne honeycomb retinal dystrophy (DHRD) refer to two autosomal dominant diseases characterized by yellow-white deposits known as drusen that accumulate beneath the retinal pigment epithelium1,2,3,4 (RPE). Both loci were mapped to chromosome 2p16-21 (Refs 5,6) and this genetic interval has been subsequently narrowed6,7. The importance of these diseases is due in large part to their close phenotypic similarity to age-related macular degeneration (AMD), a disorder with a strong genetic component8,10 that accounts for approximately 50% of registered blindness in the Western world11,12,13,14. Just as in ML and DHRD, the early hallmark of AMD is the presence of drusen15,16. Here we use a combination of positional and candidate gene methods to identify a single non-conservative mutation (Arg345Trp) in the gene EFEMP1 (for EGF-containing fibrillin-like extracellular matrix protein 1) in all families studied. This change was not present in 477 control individuals or in 494 patients with age-related macular degeneration. Identification of this mutation may aid in the development of an animal model for drusen, as well as in the identification of other genes involved in human macular degeneration.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Doyne, R.W. Peculiar condition of choroiditis occurring in several members of the same family. Trans. Ophthalmol. Soc. UK 19, 71 –71 (1899).
Forni, S. & Babel, J. Étude clinique et histologique de la malattia leventinse: affection appartenant au groupe des dégénérescences hyalines du pôle postérieur. Ophthalmologica 143, 313–322 (1962).
Vogt, A. in Handbuch der gesammten Augenheilkunde. Untersuchungsmethoden (eds Graefe, A. & Saemisch, T.) 1–118 (Verlag von Wilhelm Engelman, Berlin, 1925).
Collins, T. A pathological report upon a case of Doyne's choroiditis ('honey-comb' or 'family choroidits'). Ophthalmoscope 11, 537–538 (1913).
Heon, E. et al. Linkage of autosomal-dominant radial drusen (malattia leventinese) to chromosome 2p16-21. Arch. Ophthalmol. 114, 193–198 (1996).
Gregory, C.Y. et al. The gene responsible for autosomal dominant Doyne's honeycomb retinal dystrophy (DHRD) maps to chromosome 2p16. Hum. Mol. Genet. 5, 1055–1059 ( 1996).
Edwards, A.O. et al. Malattia leventinese: refinement of the genetic locus and phenotypic variability in autosomal dominant macular drusen. Am. J. Ophthalmol. 126, 417–424 (1998).
Silvestri, G., Johnston, P.B. & Hughes, A.E. Is genetic predisposition an important risk factor in age-related macular degeneration? Eye 8, 564–568 (1994).
Meyers, S.M. A twin study on age-related macular degeneration. Trans. Am. Ophthalmol. Soc. 92, 775–843 ( 1994).
Heiba, I.M., Elston, R.C., Klein, B.E. & Klein, R. Sibling correlations and segregation analysis of age-related maculopathy: the Beaver Dam eye study. Genet. Epidemiol. 11, 51–67 (1994).
Bressler, N.M., Bressler, S.B. & Fine, S.L. Age-related macular degeneration. Surv. Ophthalmol. 32, 375–413 (1988).
Evans, J. & Wormald, R. Is the incidence of registrable age-related macular degeneration increasing? Br. J. Ophthalmol. 80, 9–14 (1996 ).
Klein, R., Klein, B.E. & Linton, K.L. Prevalence of age-related maculopathy. The Beaver Dam eye study. Ophthalmology 99, 933– 943 (1992).
Vingerling, J.R. et al. The prevalence of age-related maculopathy in the Rotterdam study. Ophthalmology 102, 205– 210 (1995).
Sarks, J.P., Sarks, S.H. & Killingsworth, M.C. Evolution of soft drusen in age-related macular degeneration. Eye 8, 269–283 (1994).
Bird, A.C. et al. An international classification and grading system for age- related maculopathy and age-related macular degeneration. The International ARM Epidemiological Study Group. Surv. Ophthalmol. 39, 367–374 (1995).
Jay, M., Plant, C., Evans, K. & Gregory, C.Y. Doyne revisited. Eye 10, 469–472 (1996).
Tran, H., Mattei, M., Godyna, S. & Argraves, W.S. Human fibulin-1D: molecular cloning, expression and similarity with S1-5 protein, a new member of the fibulin gene family. Matrix Biol. 15, 479–493 (1997).
Lecka-Czernik, B., Lumpkin, C.K.J. & Goldstein, S. An overexpressed gene transcript in senescent and quiescent human fibroblasts encoding a novel protein in the epidermal growth factor-like repeat family stimulates DNA synthesis. Mol. Cell. Biol. 15, 120–128 (1995).
Dietz, H.C. et al. Marfan syndrome caused by a recurrent de novo missense mutation in the fibrillin gene. Nature 352, 337–339 (1991).
Dietz, H.C., Saraiva, J.M., Pyeritz, R.E., Cutting, G.R. & Francomano, C.A. Clustering of fibrillin (FBN1) missense mutations in Marfan syndrome patients at cysteine residues in EGF-like domains. Hum. Mutat. 1, 366– 374 (1992).
Wells, J. et al. Mutations in the human retinal degeneration slow (RDS) gene can cause either retinitis pigmentosa or macular dystrophy. Nature Genet. 3, 213–218 ( 1993).
Weber, B.H.F., Vogt, G., Pruett, R.C., Stohr, H. & Felbor, U. Mutations in the tissue inhibitor of metalloproteinases-3 (timp3) in patients with sorsbys fundus dystrophy. Nature Genet. 8, 352–356 ( 1994).
Allikmets, R. et al. A photoreceptor cell-specific ATP-binding transporter gene (ABCR) is mutated in recessive Stargardt macular dystrophy. Nature Genet. 15, 236–246 (1997).
Petrukhin, K. et al. Identification of the gene responsible for Best macular dystrophy. Nature Genet. 19, 241– 247 (1998).
Marquardt, A. et al. Mutations in a novel gene, VMD2, encoding a protein of unknown properties cause juvenile-onset vitelliform macular dystrophy (Best's disease). Hum. Mol. Genet. 7, 1517– 1525 (1998).
Buffone, G.J. & Darlington, G.J. Isolation of DNA from biological specimens without extraction with phenol. Clin. Chem. 31, 164–165 (1985).
Nishimura, D.Y. et al. The forkhead transcription factor gene FKHL7 is responsible for glaucoma phenotypes which map to 6p25. Nature Genet. 19, 140–147 (1998).
Ikegawa, S., Toda, T., Okui, K. & Nakamura, Y. Structure and chromosomal assignment of the human S1-5 gene (FBNL) that is highly homologous to fibrillin. Genomics 35, 590– 592 (1996).
Fingert, J.H. et al. Characterization and comparison of the human and mouse GLC1A glaucoma genes. Genome Res. 8, 377– 384 (1998).
Acknowledgements
We thank S. Sneed, T. Polk, J.D.M. Gass, J. Slakter, L. Yannuzzi, D. Keenum, A. Franceschetti, S. Forni, C. Anastasi-Forni, S. Sarks, C.A. Harper, P. Allen, R. Buttery, C. McCarty, G. Morgan, S. Kirmani and S. Bhattacharya for sharing patients or DNA samples; L. Streb, R. Hockey, H. Haines, L. Affatigato, G. Beck, C. Taylor, S. Krob, G. Metthez, F. Ahmad, V. Buchillier, V. Kaltenrieder, R. McNeil and M. Cain for technical assistance; and C. Fasser for support of retinal degeneration research. Supported in part by NIH grants EY10539 and EY11515, the Carver Charitable Trust, The Ruth and Milton Steinbach Foundation, The Grousbeck Family Foundation, The Foundation Fighting Blindness, the Swiss National Science Foundation grant 32-053750.98, the Fondation Telethon Action Suisse, The Royal Victorian Institute for the Blind and an unrestricted grant from Research to Prevent Blindness.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Stone, E., Lotery, A., Munier, F. et al. A single EFEMP1 mutation associated with both Malattia Leventinese and Doyne honeycomb retinal dystrophy. Nat Genet 22, 199–202 (1999). https://doi.org/10.1038/9722
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/9722
This article is cited by
-
Oxidative stress differentially impacts apical and basolateral secretion of angiogenic factors from human iPSC-derived retinal pigment epithelium cells
Scientific Reports (2022)
-
The second Japanese family with Malattia Leventinese/Doyne honeycomb retinal dystrophy
Documenta Ophthalmologica (2022)
-
Clinically-identified C-terminal mutations in fibulin-3 are prone to misfolding and destabilization
Scientific Reports (2021)
-
Complement C5 is not critical for the formation of sub-RPE deposits in Efemp1 mutant mice
Scientific Reports (2021)