Abstract
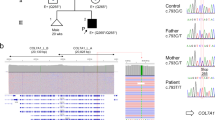
Epidermolysis bullosa simplex (EBS) is a group of epidermal blistering diseases almost invariably transmitted as a dominant trait, which has recently been shown to arise from mutations in keratins 14 and 5 (K14 and K5). We describe a family with recessive EBS in which the disease is tightly linked to the substitution of the highly conserved glutamic acid-144 to alanine in the first helical segment of the rod domain of keratin 14. In contrast, linkage with keratin 5 was excluded. The loss of an ionic interaction with keratin 5 is likely to affect K14–K5 heterodimer formation. Our data suggest that this mutation underlies EBS in our family, and that mutations in keratin genes may impair the mechanical integrity of basal keratinocytes in a recessive as well as dominant fashion.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Fine, J.-D. et al. Revised clinical and laboratory criteria for subtypes of inherited epidermolysis bullosa: a consensus report by the subcommittee on diagnosis and classification of the national epidermolysis bullosa registry. J. Am. Acad. Dermatol. 24, 119–135 (1991).
Lin, A.N. & Carter, D.M. in Epidermolysis Bullosa: Basic and Clinical Aspects (eds Lin, A.N. & Garter, D.M.) 89–117 (Springer Verlag, New York, 1992).
Bonifas, J.M., Rothman, A.L. & Epstein, E.H., Jr. Epidermolysis bullosa simplex: evidence in two families for keratin gene abnormalities. Science 254, 1202–1205 (1991).
Coulombe, P.A. et al. Point mutations in human keratin 14 genes of epidermolysis bullosa simplex patients: genetic and functional analyses. Cell 66, 1301–1311 (1991).
Lane, E.B. et al. A mutation in the conserved helix termination peptide of keratin 5 in hereditary skin blistering. Nature 356, 244–246 (1992).
Romano, V. et al. Chromosomal assignments of human type I and type II cytokeratin genes to different chromosomes. Cytogenet. Cell Genet. 48, 148–151 (1988).
Ryynänen, M., Knowiton, R.G. & Uitto, J. Mapping of epidermolysis bullosa simplex mutation to chromosome 12. Am. J. hum. Genet. 49, 978–984 (1991).
Vassar, R., Coulombe, P.A., Degenstein, L., Albers, K. & Fuchs, E. Mutant keratin expression in transgenic mice causes marked abnormalities resembling a human genetic skin disease. Cell 64, 365–380 (1991).
Coulombe, P.A., Hutton, M.E., Vassar, R. & Fuchs, E. A function for keratins and a common thread among different types of epidermolysis bullosa simplex diseases. J. cell Biol. 115, 1661–1674 (1991).
Coulombe, P.A., Chan, Y.-M., Albers, K. & Fuchs, E. Deletions in epidermal keratins leading to alterations in filament organization in vivo and in intermediate filament assembly in vitro. J. cell Biol. 111, 3049–3064 (1990).
Letai, A., Coulombe, P.A. & Fuchs, E. Do the ends justify the mean? Proline mutations at the ends of the keratin coiled-coil rod segment are more disruptive than internal mutations. J. cell Biol. 116, 1181–1195 (1992).
Epstein, E.H., Jr. Molecular genetics of epidermolysis bullosa. Science 256, 799–803 (1992).
Fuchs, E. & Coulombe, P.A. Of mice arid men: genetic skin diseases of keratin. Cell 69, 899–902 (1992).
Salih, M.A.M., Lake, B.D., El Hag, M.A. & Atherton, D.J. Lethal epidermolytic epidermolysis bullosa: a new autosomal recessive type of epidermolysis bullosa. Br. J. Dermatol. 113, 135–143 (1985).
Nielsen, P.G. & Sjolund, E. Epidermolysis bullosa simplex localisata associated with anodontia, hair and nail disorders: a new syndrome. Acta Derm. Veneorol. 65, 526–530 (1985).
Niemi, K.-M., Sommer, H., Kero, M., Kanerva, L. & Haltia, M. Epidermolysis bullosa simplex associated with muscular dystrophy with recessive inheritance. Arch. Dermatol. 124, 551–554 (1988).
Fine, J.-D. et al. Autosomal recessive epidermolysis bullosa simplex. Arch. Dermatol. 125, 931–938 (1989).
Fine, J.-D., Johnson, L., Wright, T. & Horiguchi, Y. Epidermolysis bullosa simplex: identification of a kindred with autosomal recessive transmission of the Weber-Cockayne variety. Ped. Dermatol. 6, 1–5 (1989).
Rosenfeld, P.J. et al. A null mutation in the rhodopsin gene causes rod photoreceptor dysfunction and autosomal recessive retinitis pigmentosa. Nature Genet. 1, 209–213 (1992).
Marchuk, D., McCrohon, S. & Fuchs, E. Complete sequence of a gene encoding a human type I keratin: sequences homologous to enhancer elements in the regulatory region of the gene. Proc. natn Acad. Sci. U.S.A. 82, 1609–1613 (1985).
Marchuk, D., McCrohon, S. & Fuchs, E. Remarkable conservation of structure among intermediate filament genes. Cell 39, 491–498 (1984).
Korge, B.P. et al. Extensive size polymorphism of the human keratin 10 chain resides in the C-terminal V2 subdomain due to variable numbers and sizes of glycine loops. Proc. natn. Acad. Sci. U.S.A. 89, 910–914 (1992).
Rosenberg, M., Raychaudhury, A., Shows, T.B., LeBeau, M.M. & Fuchs, E. A group of type I keratin genes on human chromosome 17: characterization and expression. Molec. cell Biol. 8, 722–736 (1988).
Lander, E.S. & Bostein, D. Homozygosity mapping: a way to map human recessive traits with the DNA of inbred children. Science 236, 1567–1570 (1987).
Compton, J.G. et al. Linkage of epidermolytic hyperkeratosis to the type II keratin gene cluster on chromosome 12q. Nature Genet. 1, 301–305 (1992).
Lee, L.D. & Baden, H.P. Organization of the polypeptide chains in mammalian keratin. Nature 264, 377–379 (1976).
Parry, D.A.D., Crewther, W.G., Fraser, R.D.B. & McRae, T.P. Structure of α-keratin: structural implication of the amino acid sequences of the type I and type II chain segments. J. molec. Biol. 113, 449–454 (1977).
Mc Lachlan, A.D. Coiled-coil formation and sequence regularities in the helical regions of α-keratins. J. molec. Biol. 124, 297–304 (1978).
Fuchs, E.V., Coppock, S.M., Green, H. & Cleveland, D.W. Two distinct classes of keratin genes and their evolutionary significance. Cell 27, 75–84 (1981).
Steinert, P.M., Steven, A.C. & Roop, D.R. The molecular biology of intermediate filaments. Cell 42, 411–419 (1985).
Parry, D.A.D., Steven, A.C. & Steinert, P.M. The coiled-coil molecules of intermediate filaments consist of two parallel chains in exact axial register. Biochem. Biophys. Res. Comm. 127, 1012–1018 (1985).
Fraser, R.D.B., MacRae, T.P., Parry, D.A.D. & Suzuki, E. Intermediate filaments in α-keratins. Proc. natn. Acad. Sci. U.S.A. 83, 1179–1183 (1986).
Steinert, P.M. & Freedberg, I.M. in Physiology, Biochemistry, and Molecular Biology of the Skin (ed. Goldsmith, L.A.) 113–147 (Oxford University Press, New York, 1992).
Coulombe, P.A. & Fuchs, E. Elucidating the early stages of keratin filament assembly. J. cell Biol. 111, 153–169 (1990).
Eckert, R.L. & Rorke, E.A. The sequence of the human epidermal 58-kD (♯5) type II keratin reveals an absence of 5′ upstream sequence conservation between coexpressed epidermal keratins. DNA 7, 337–345 (1988).
Rothnagel, J.A. et al. Mutations in the rod domains of keratins 1 and 10 in epidermolytic hyperkeratosis. Science 257, 1128–1130 (1992).
Cheng, J. et al. The genetic basis of epidermolytic hyperkeratosis: a disorder of differentiation-specific epidermal keratin genes. Cell 70, 811–819 (1992).
Chipev, C.C. et al. A Leucine→Proline mutation in the cH1 subdomain of keratin 1 causes epidermolytic hyperkeratosis. Cell 70, 821–828 (1992).
Byers, P.H. Brittle bones-fragile molecules: disorders of collagen gene structure and expression. Trends Genet. 6, 293–300 (1990).
Kuivaniemi, H., Tromp, G. & Prockop, D.J. Mutations in collagen genes: causes of rare and some common diseases in humans. Faseb J. 5, 2052–2060 (1991).
Prockop, D.J. Mutations in collagen genes as a cause of connective-tissue diseases. New Engl. J. Med. 326, 540–546 (1992).
Krane, S.M., Pinnell, S.R. & Erbe, R.W. Lysyl-protocollagen hydroxylase deficiency in fibroblasts from siblings with hydroxylysine-deficient collagen. Proc. natn. Acad. Sci. U.S.A. 69, 2899–2903 (1972).
Nusgens, B.V. et al. Evidence for a relationship between Ehlers-Danlos type VII C in humans and bovine dermatosparaxis. Nature Genet. 1, 214–217 (1992).
Pope, F.M. et al. Patients with Ehlers-Danlos syndrome type IV lack type III collagen. Proc. natn. Acad. Sci. U.S.A. 72, 1314–1316 (1975).
Tromp, G. & Prockop, D. Single base mutation in the proα2(I) collagen gene that causes efficient splicing of RNA from exon 27 to exon 29 and synthesis of a shortened but in-frame proa2(I) chain. Proc. natn. Acad. Sci. U.S.A. 85, 5254–5258 (1988).
Pihlajaniemi, T. et al. Osteogenesis imperfecta: cloning of a pro-α2(I) collagen gene with a frameshift mutation. J. biol. Chem. 259, 12941–12944 (1984).
Cohn, D.H., Starman, B.J., Blumberg, B. & Byers, P.M. Recurrence of lethal osteogenesis imperfecta due to parental mosaicism for a dominant mutation in a human type I collagen gene. Am. J. hum. Genet. 46, 591–601 (1990).
Anton-Lamprecht, I., Jovanovic, V., Arnold, M.-L., Rauskolb, R. & Schenck, W. Prenatal diagnosis of genetic disorders of the skin by means of electron microscopy. Hum. Genet. 59, 392–405 (1981).
Rheinwald, J.G. & Green, H. Serial cultivation of strains of human epidermal keratinocytes: the formation of keratinizing colonies from single cells. Cell 6, 331–334 (1975).
Chomczynski, P. & Sacchi, N. Single step method of RNA isolation of acid guanidium thibcyanate-phenol-chloroform extraction. Anal. Biochem. 162, 156–159 (1987).
Gyllensten, U.B. & Erlich, H.A. Generation of single-strand DNA by the polymerase chain reaction and its application to direct sequencing of the HLA-DQA locus. Proc. natn. Acad. Sci. U.S.A. 85, 7652–7656 (1988).
Dessen, P., Fondrat, C., Valencien, C. & Mugnier, C. Bisance: a French service for access to biomolecular sequences databases. Cabios. 6, 355–356 (1990).
Lathrop, G.M., Lalouel, J.-M., Julier, C. & Ott, J. Strategies for multitocus linkage analysis in humans. Proc. natn. Acad. Sci. U.S.A. 81, 3443–3446 (1984).
Nakamura, Y. et al. Isolation and mapping of a polymorphic DNA sequence pYNH15 on chromosome 12q [D12S17]. Nucl. Acids Res. 16, 779 (1988).
Priestley, L., Kumar, D. & Sykes, B. Amplification of the COL2A1 3′ variable region used for segregation analysis in a family with the Stickler syndrome. Hum. Genet. 85, 525–526 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Hovnanian, A., Pollack, E., Hilal, L. et al. A missense mutation in the rod domain of keratin 14 associated with recessive epidermolysis bullosa simplex. Nat Genet 3, 327–332 (1993). https://doi.org/10.1038/ng0493-327
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0493-327
This article is cited by
-
Blistering disease: insight from the hemidesmosome and other components of the dermal-epidermal junction
Cell and Tissue Research (2015)
-
Epidermal cell junctions and their regulation by p63 in health and disease
Cell and Tissue Research (2015)
-
Defining Keratin Protein Function in Skin Epithelia: Epidermolysis Bullosa Simplex and Its Aftermath
Journal of Investigative Dermatology (2012)
-
Epidermolysis bullosa
Der Hautarzt (2009)
-
Novel Keratin 14 Mutations in Patients with Severe Recessive Epidermolysis Bullosa Simplex
Journal of Investigative Dermatology (2006)