Abstract
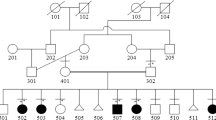
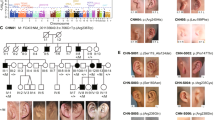
The genetic analysis of congenital skull malformations provides insight into normal mechanisms of calvarial osteogenesis1. Enlarged parietal foramina (PFM) are oval defects of the parietal bones caused by deficient ossification around the parietal notch, which is normally obliterated during the fifth fetal month2. PFM are usually asymptomatic, but may be associated with headache, scalp defects and structural or vascular malformations of the brain3,4. Inheritance is frequently autosomal dominant, but no causative mutations have been identified in non-syndromic cases. We describe here heterozygous mutations of the homeobox gene MSX2 (located on 5q34–q35) in three unrelated families with PFM. One is a deletion of approximately 206 kb including the entire gene and the others are intragenic mutations of the DNA-binding homeodomain (RK159-160del and R172H) that predict disruption of critical intramolecular and DNA contacts. Mouse Msx2 protein with either of the homeodomain mutations exhibited more than 85% reduction in binding to an optimal Msx2 DNA-binding site. Our findings contrast with the only described MSX2 homeodomain mutation5 (P148H), associated with craniosynostosis, that binds with enhanced affinity to the same target6. This demonstrates that MSX2 dosage is critical for human skull development and suggests that PFM and craniosynostosis result, respectively, from loss and gain of activity in an MSX2-mediated pathway of calvarial osteogenic differentiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Mundlos, S. et al. Mutations involving the transcription factor CBFA1 cause cleidocranial dysplasia. Cell 89, 773– 779 (1997).
Currarino, G. Normal variants and congenital anomalies in the region of the obelion. Am. J. Roentgenol. 127, 487–494 (1976).
Pang, D. & Lin, A. Symptomatic large parietal foramina . Neurosurgery 11, 33–37 (1982).
Preis, S., Engelbrecht, V. & Lenard, H.-G. Aplasia cutis congenita and enlarged parietal foramina (Catlin marks) in a family. Acta Paediatr. 84, 701–702 (1995).
Jabs, E.W. et al. A mutation in the homeodomain of the human MSX2 gene in a family affected with autosomal dominant craniosynostosis Cell 75, 443–450 ( 1993).
Ma, L., Golden, S., Wu, L. & Maxson, R. The molecular basis of Boston-type craniosynostosis: the Pro148–His mutation in the N-terminal arm of the MSX2 homeodomain stabilizes DNA binding without altering nucleotide sequence preferences. Hum. Mol. Genet. 5 , 1915–1920 (1996).
Bartsch, O. et al. Delineation of a contiguous gene syndrome with multiple exostoses, enlarged parietal foramina, craniofacial dysostosis, and mental retardation, caused by deletions on the short arm of chromosome 11. Am. J. Hum. Genet. 58, 734–742 ( 1996).
Wuyts, W. et al. Molecular and clinical examination of an Italian DEFECT 11 family . Eur. J. Hum. Genet. 7, 579– 584 (1999).
Wilkie, A.O.M. et al. Saethre-Chotzen syndrome associated with balanced translocations involving 7p21.2: three further families. J. Med. Genet. 32, 174–180 (1995).
Johnson, D. et al. A comprehensive screen for TWIST mutations in patients with craniosynostosis identifies a new microdeletion syndrome of chromosome band 7p21.1. Am. J. Hum. Genet. 63, 1282 –1293 (1998).
Golabi, M., Carey, J. & Hall, B.D. Parietal foramina clavicular hypoplasia. An autosomal dominant syndrome. Am. J. Dis. Child. 138, 596–599 (1984).
Salamanca, A. et al. Prenatal sonographic appearance of foramina parietalia permagna . Prenat. Diagn. 14, 766– 769 (1994).
Kutilek, S., Baxova, A., Bayer, M., Leiska, A. & Kozlowski, K. Foramina parietalia permagna: report of nine cases in one family. J. Paediatr. Child Health 33, 168–170 (1997).
Bürglin, T.R. A comprehensive classification of homeobox genes. in Guidebook to the Homeobox Genes (ed. Duboule, D.) 27–71 (Oxford University Press, Oxford, 1994).
Gehring, W.J. et al. Homeodomain-DNA recognition. Cell 78 , 211–223 (1994).
Li, H. et al. Homology modeling using simulated annealing of restrained molecular dynamics and conformational search calculations with CONGEN: application in predicting the three-dimensional structure of murine homeodomain Msx-1. Protein Sci. 6, 956–970 (1997).
Clarke, N.D. Covariation of residues in the homeodomain sequence family. Protein Sci. 4, 2269–2278 ( 1995).
Isaac, V.E., Sciavolino, P. & Abate, C. Multiple amino acids determine the DNA binding specificity of the Msx-1 homeodomain. Biochemistry 34, 7127–7134 (1995).
Vastardis, H., Karimbux, N., Guthua, S.W., Seidman, J.G. & Seidman, C.E. A human MSX1 homeodomain missense mutation causes selective tooth agenesis. Nature Genet. 13, 417–421 ( 1996).
Hu, G. et al. Haploinsufficiency of MSX1 : a mechanism for selective tooth agenesis. Mol. Cell. Biol. 18, 6044– 6051 (1998).
van den Boogaard, M-J.H., Dorland, M., Beemer, F.A. & Ploos van Amstel, H.K. MSX1 mutation is associated with orofacial clefting and tooth agenesis in humans. Nature Genet 24, 342– 343 (2000).
Catron, K.M., Iler, N. & Abate, C. Nucleotides flanking a conserved TAAT core dictate the DNA binding specificity of three murine homeodomain proteins. Mol. Cell. Biol. 13, 2354–2365 (1993).
Liu, Y.-H. et al. Msx2 gene dosage influences the number of proliferative osteogenic cells in growth centers of the developing murine skull: a possible mechanism for MSX2-mediated craniosynostosis in humans. Dev. Biol. 205, 260–274 (1999).
Satokata, I. et al. Msx2 deficiency in mice causes pleiotropic defects in bone growth and ectodermal organ function. Nature Genet. 24, 391–395 (2000).
Wilkie, A.O.M. The molecular basis of genetic dominance. J. Med. Genet. 31, 89–98 (1994).
Gripp, K.W. et al. TWIST gene mutation in a patient with radial aplasia and craniosynostosis: further evidence for heterogeneity of Baller-Gerold syndrome. Am. J. Med. Genet. 82, 170– 176 (1999).
Stankiewicz, P. et al. The TWIST gene is triplicated in trisomy 7p syndrome. Eur. J. Hum. Genet. 6, 61 (1998).
Dib, C. et al. A comprehensive genetic map of the human genome based on 5,264 microsatellites. Nature 380, 152– 154 (1996).
Hodgkinson, J.E., Davidson, C.L., Beresford, J. & Sharpe, P.T. Expression of a human homeobox-containing gene is regulated by 1,25(OH) 2D3 in bone cells. Biochim. Biophys. Acta 1174, 11–16 (1993).
Ioannou, P.A. & de Jong, P.J. Construction of bacterial artificial chromosome libraries using the modified P1 (PAC) system. in Current Protocols in Human Genetics (eds Dracopoli, N.C. et al.) 5.15.1–5.15.24 (Wiley, New York, 1996 ).
Jaju, R. et al. A new recurrent translocation, t(5;11)(q35;p15.5), associated with del(5q) in childhood acute myeloid leukemia. Blood 94, 773–780 (1999).
Acknowledgements
We thank family members for their participation; A. Baxova, S. Kutilek, A. Salamanca, L. Solymosi, H. Urbach and S. White for help with collection of samples; R. Jaju and L. Kearney for FISH analysis; P. Anslow for radiological advice, R. Maas for sharing unpublished data; and S. Robertson for comments on the manuscript. PAC clones were provided by the UK HGMP Resource Centre. This work was funded by a Wellcome Trust Senior Fellowship in Clinical Science (A.O.M.W.) and grants from the NIDCD (R.E.M.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wilkie, A., Tang, Z., Elanko, N. et al. Functional haploinsufficiency of the human homeobox gene MSX2 causes defects in skull ossification. Nat Genet 24, 387–390 (2000). https://doi.org/10.1038/74224
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/74224
This article is cited by
-
Computed-tomography evaluation of parietal foramen topography in adults: a retrospective analysis
Surgical and Radiologic Anatomy (2024)
-
Multisuture craniosynostosis: a case report of unusual presentation of chromosome 14q32 deletion
Child's Nervous System (2023)
-
Expression of VEGFA-mRNA in classical and MSX2-mRNA in non-classical monocytes in patients with spondyloarthritis is associated with peripheral arthritis
Scientific Reports (2021)
-
Regulation of MDM2 E3 ligase-dependent vascular calcification by MSX1/2
Experimental & Molecular Medicine (2021)
-
Lens-specific conditional knockout of Msx2 in mice leads to ocular anterior segment dysgenesis via activation of a calcium signaling pathway
Laboratory Investigation (2019)