Abstract
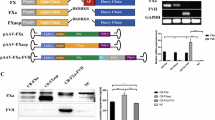
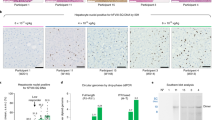
Pre-clinical studies in mice and haemophilic dogs have shown that introduction of an adeno-associated viral (AAV) vector encoding blood coagulation factor IX (F.IX) into skeletal muscle results in sustained expression of F.IX at levels sufficient to correct the haemophilic phenotype1,2. On the basis of these data and additional pre-clinical studies demonstrating an absence of vector-related toxicity, we initiated a clinical study of intramuscular injection of an AAV vector expressing human F.IX in adults with severe haemophilia B. The study has a dose-escalation design, and all patients have now been enrolled in the initial dose cohort (2×1011 vg/kg). Assessment in the first three patients of safety and gene transfer and expression show no evidence of germline transmission of vector sequences or formation of inhibitory antibodies against F.IX. We found that the vector sequences are present in muscle by PCR and Southern-blot analyses of muscle biopsies and we demonstrated expression of F.IX by immunohistochemistry. We observed modest changes in clinical endpoints including circulating levels of F.IX and frequency of F.IX protein infusion. The evidence of gene expression at low doses of vector suggests that dose calculations based on animal data may have overestimated the amount of vector required to achieve therapeutic levels in humans, and that the approach offers the possibility of converting severe haemophilia B to a milder form of the disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Herzog, R.W. et al. Stable gene transfer and expression of human blood coagulation factor IX after intramuscular injection of recombinant adeno-associated virus . Proc. Natl Acad. Sci. USA 94, 5804– 5809 (1997).
Herzog, R. et al. Long-term correction of canine hemophilia B by gene transfer of blood coagulation factor IX mediated by adeno-associated viral vector. Nature Med. 5, 56–63 ( 1999).
Ljung, R.C.R. Annotation. Can haemophilic arthropathy be prevented? Br. J. Haematol. 101, 215–219 ( 1998).
Lofqvist, T., Nilsson, I.M., Berntorp, E. & Pettersson, H. Haemophilia prophylaxis in young patients—a long term follow up. J. Intern. Med. 241, 395–400 (1997).
Mannucci, P.M. & Weitz, J.I. The hemophilias: progress and problems . Semin. Hematol. 36, 104– 117 (1999).
Parks, W.P., Boucher, D.W., Melnick, J.L., Taber, L.H. & Yow, M.D. Seroepidemiological and ecological studies of the adenovirus-associated satellite viruses. Infect. Immun. 2, 716–722 ( 1970).
Summerford, C. & Samulski, R.J. Membrane-associated heparan sulfate proteoglycan is a receptor for adeno-associated virus type 2 virions. J. Virol. 72, 1438–1445 (1998).
Qing, K. et al. Human fibroblast growth growth factor receptor 1 is a co-receptor for infection by adeno-associated virus 2. Nature Med. 5, 71–77 (1999).
Cheung, W.F. et al. Identification of the endothelial cell binding site for factor IX. Proc. Natl Acad. Sci. USA 93, 11068– 11073 (1996).
Matsushita, T. et al. Adeno-associated virus vectors can be efficiently produced without helper virus. Gene Ther. 5, 938– 945 (1998).
Burton, M. et al. Coexpression of factor VIII heavy and light chain adeno-associated viral vectors produces biologically active protein. Proc. Natl Acad. Sci. USA 96, 12725–12730 (1999).
Yoshitake, S., Schach, B.G., Foster, D.C., Davie, E.W. & Kuracki, K. Nucleotide sequence of the gene for human factor IX (antihemophilic factor B). Biochemistry 24, 3736–3750 (1985).
Kurachi, S., Hitomi, Y., Midori, F. & Kurachi, K. Role of intron I in expression of the human factor IX gene. J. Biol. Chem. 270, 5276–5281 (1995).
Manno, C.S., Larson, P.J., Cohen, A.R. & Flake, A.W. Guidelines for Research Involving Recombinant DNA Molecules (NIH Guidelines). Appendix M. Points to consider in the design and submission of protocols for the transfer of recombinant DNA molecules into one or more human subjects. (NIH Recombinant DNA Advisory Committee, Washington DC, 1999).
Walter, J., You, Q., Hagstrom, J.N., Sands, M. & High, K.A. Successful expression of human factor IX following repeat administration of adenoviral vector in mice. Proc. Natl Acad. Sci. USA 93, 3056–3061 ( 1996).
Miller, J.H. Experiments in Molecular Genetics 352–355 (Cold Spring Harbor Press, Cold Spring Harbor, New York, 1972).
Acknowledgements
We thank A. Radu for assistance with immunohistochemical staining of muscle; M. Tanzer for assistance with coagulation assays; D. Leonard for assistance with PCR assays on human samples; the Nucleic Acid/Protein Research Core Facility at CHOP; R. Barth and M. King for assistance with ultrasound; and S.G. Madison for clinical support. This work was supported by National Institutes of Health Grants R01 HL53682 to M.A.K. and R01 HL53668, R01 HL61921, P50 HL54500 and a grant from the Hoxie Harrison-Smith Foundation to K.A.H. The work was also supported in part by NIH grant M01 RR00070 to the GCRC at Stanford University, by NIH grant M01 RR00240 to the GCRC at The Children's Hospital of Philadelphia and by Avigen.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kay, M., Manno, C., Ragni, M. et al. Evidence for gene transfer and expression of factor IX in haemophilia B patients treated with an AAV vector. Nat Genet 24, 257–261 (2000). https://doi.org/10.1038/73464
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/73464
This article is cited by
-
Perioperative Management of Hemophilia Patients
Current Anesthesiology Reports (2024)
-
Adeno-associated virus mediated expression of monoclonal antibody MR191 protects mice against Marburg virus and provides long-term expression in sheep
Gene Therapy (2022)
-
Current progress and future direction in the treatment for hemophilia
International Journal of Hematology (2020)
-
Advances in gene therapy for hemophilia: basis, current status, and future perspectives
International Journal of Hematology (2020)
-
Gene Therapy for Hemophilia: Progress to Date
BioDrugs (2018)