Abstract
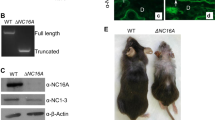
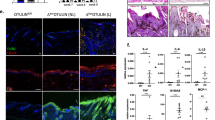
Basaloid skin tumors, including basal cell carcinoma (BCC) and basaloid follicular hamartoma, are associated with aberrant Hedgehog (Hh) signaling1 and, in the case of BCC, an expanding set of genetic variants including keratin 5 (encoded by KRT5)2, an intermediate filament-forming protein. We here show that genetic ablation of keratin 17 (Krt17) protein, which is induced in basaloid skin tumors3,4 and co-polymerizes with Krt5 in vivo5, delays basaloid follicular hamartoma tumor initiation and growth in mice with constitutive Hh signaling in epidermis6,7. This delay is preceded by a reduced inflammation and a polarization of inflammatory cytokines from a Th1- and Th17-dominated profile to a Th2-dominated profile. Absence of Krt17 also attenuates hyperplasia and inflammation in models of acute dermatitis. Re-expression of Krt17 in Gli2tg; Krt17−/− keratinocytes induces select Th1 chemokines that have established roles in BCC. Our findings establish an immunomodulatory role for Krt17 in Hh driven basaloid skin tumors that could impact additional tumor settings, psoriasis and wound repair.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Epstein, E.H. Basal cell carcinomas: attack of the hedgehog. Nat. Rev. Cancer 8, 743–754 (2008).
Stacey, S.N. et al. New common variants affecting susceptibility to basal cell carcinoma. Nat. Genet. 41, 909–914 (2009).
Markey, A.C., Lane, E.B., Macdonald, D.M. & Leigh, I.M. Keratin expression in basal cell carcinomas. Br. J. Dermatol. 126, 154–160 (1992).
Yu, M. et al. Superficial, nodular, and morpheiform basal-cell carcinomas exhibit distinct gene expression profiles. J. Invest. Dermatol. 128, 1797–1805 (2008).
Larouche, D., Tong, X., Fradette, J., Coulombe, P.A. & Germain, L. Vibrissa hair bulge houses two populations of skin epithelial stem cells distinct by their keratin profile. FASEB J. 22, 1404–1415 (2008).
Grachtchouk, M. et al. Basal cell carcinomas in mice overexpressing Gli2 in skin. Nat. Genet. 24, 216–217 (2000).
Grachtchouk, V. et al. The magnitude of hedgehog signaling activity defines skin tumor phenotype. EMBO J. 22, 2741–2751 (2003).
Jih, D. et al. Familial basaloid follicular hamartoma: lesional characterization and review of the literature. Am. J. Dermatopathol. 25, 130–137 (2003).
Callahan, C.A. et al. MIM/BEG4, a Sonic hedgehog-responsive gene that potentiates Gli-dependent transcription. Genes Dev. 18, 2724–2729 (2004).
McGowan, K.M. et al. Keratin 17 null mice exhibit age- and strain-dependent alopecia. Genes Dev. 16, 1412–1422 (2002).
Kerns, M.L., DePianto, D., Dinkova-Kostova, A.T., Talalay, P. & Coulombe, P.A. Reprogramming of keratin biosynthesis by sulforaphane restores skin integrity in epidermolysis bullosa simplex. Proc. Natl. Acad. Sci. USA 104, 14460–14465 (2007).
Cataisson, C. et al. Inducible cutaneous inflammation reveals a protumorigenic role for keratinocyte CXCR2 in skin carcinogenesis. Cancer Res. 69, 319–328 (2009).
Elias, P.M. & Schmuth, M. Abnormal skin barrier in the etiopathogenesis of atopic dermatitis. Curr. Opin. Allergy Clin. Immunol. 9, 437–446 (2009).
Demehri, S., Morimoto, M., Holtzman, M.J. & Kopan, R. Skin-derived TSLP triggers progression from epidermal-barrier defects to asthma. PLoS Biol. 7, e1000067 (2009).
Bradley, P.P., Priebat, D.A., Christensen, R.D. & Rothstein, G. Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. J. Invest. Dermatol. 78, 206–209 (1982).
Hardman, M.J., Sisi, P., Banbury, D.N. & Byrne, C. Patterned acquisition of skin barrier function during development. Development 125, 1541–1552 (1998).
Berger, A. Th1 and Th2 responses: what are they? Br. Med. J. 321, 424 (2000).
Steinman, L. A brief history of TH17, the first major revision in the TH1/TH2 hypothesis of T cell-mediated tissue damage. Nat. Med. 13, 139–145 (2007).
Nickoloff, B.J. Cracking the cytokine code in psoriasis. Nat. Med. 13, 242–244 (2007).
Phillips, W.G., Feldmann, M., Breathnach, S.M. & Brennan, F.M. Modulation of the IL-1 cytokine network in keratinocytes by intracellular IL-1 α and IL-1 receptor antagonist. Clin. Exp. Immunol. 101, 177–182 (1995).
Lund, S.A., Giachelli, C.M. & Scatena, M. The role of osteopontin in inflammatory processes. J. Cell Commun. Signal. 3, 311–322 (2009).
Jee, S.H. et al. Interleukin-6 induced basic fibroblast growth factor-dependent angiogenesis in basal cell carcinoma cell line via JAK/STAT3 and PI3-kinase/Akt pathways. J. Invest. Dermatol. 123, 1169–1175 (2004).
Hattori, Y. et al. Vascular expression of matrix metalloproteinase-13 (collagenase-3) in basal cell carcinoma. Exp. Mol. Pathol. 74, 230–237 (2003).
Ying, S. et al. C–C chemokines in allergen-induced late-phase cutaneous responses in atopic subjects: association of eotaxin with early 6-hour eosinophils, and of eotaxin-2 and monocyte chemoattractant protein-4 with the later 24-hour tissue eosinophilia, and relationship to basophils and other C–C chemokines (monocyte chemoattractant protein-3 and RANTES). J. Immunol. 163, 3976–3984 (1999).
Yuspa, S.H. The pathogenesis of squamous cell cancer: lessons learned from studies of skin carcinogenesis–thirty-third G. H. A. Clowes Memorial Award Lecture. Cancer Res. 54, 1178–1189 (1994).
Eckert, R.L. et al. S100 proteins in the epidermis. J. Invest. Dermatol. 123, 23–33 (2004).
Braff, M.H., Bardan, A., Nizet, V. & Gallo, R.L. Cutaneous defense mechanisms by antimicrobial peptides. J. Invest. Dermatol. 125, 9–13 (2005).
Miyazaki, H. et al. Down-regulation of CXCL5 inhibits squamous carcinogenesis. Cancer Res. 66, 4279–4284 (2006).
Lo, B.K. et al. CXCR3/ligands are significantly involved in the tumorigenesis of basal cell carcinoma. Am. J. Pathol. 176, 2435–2446 (2010).
Pasparakis, M. Regulation of tissue homeostasis by NF-κB signalling: implications for inflammatory diseases. Nat. Rev. Immunol. 9, 778–788 (2009).
Tensen, C.P. et al. Genomic organization, sequence and transcriptional regulation of the human CXCL 11(1) gene. Biochim. Biophys. Acta 1446, 167–172 (1999).
Smith, J.B. et al. Cloning and genomic localization of the murine LPS-induced CXC chemokine (LIX) gene, Scyb5. Immunogenetics 54, 599–603 (2002).
Bunting, K. et al. Genome-wide analysis of gene expression in T cells to identify targets of the NF-κ B transcription factor c-Rel. J. Immunol. 178, 7097–7109 (2007).
Tong, X. & Coulombe, P.A. Keratin 17 modulates hair follicle cycling in a TNFα-dependent fashion. Genes Dev. 20, 1353–1364 (2006).
Kim, S., Wong, P. & Coulombe, P.A. A keratin cytoskeletal protein regulates protein synthesis and epithelial cell growth. Nature 441, 362–365 (2006).
van de Rijn, M. et al. Expression of cytokeratins 17 and 5 identifies a group of breast carcinomas with poor clinical outcome. Am. J. Pathol. 161, 1991–1996 (2002).
Sarbia, M. et al. Differentiation between pancreaticobiliary and upper gastrointestinal adenocarcinomas: is analysis of cytokeratin 17 expression helpful? Am. J. Clin. Pathol. 128, 255–259 (2007).
Paus, R., Ito, N., Takigawa, M. & Ito, T. The hair follicle and immune privilege. J. Investig. Dermatol. Symp. Proc. 8, 188–194 (2003).
McGowan, K.M. & Coulombe, P.A. Onset of keratin 17 expression coincides with the definition of major epithelial lineages during skin development. J. Cell Biol. 143, 469–486 (1998).
Bernot, K.M., Coulombe, P.A. & McGowan, K.M. Keratin 16 expression defines a subset of epithelial cells during skin morphogenesis and the hair cycle. J. Invest. Dermatol. 119, 1137–1149 (2002).
Bernot, K.M., Coulombe, P.A. & Wong, P. Skin: an ideal model system to study keratin genes and proteins. Methods Cell Biol. 78, 453–487 (2004).
Acknowledgements
We thank M. Han for technical support and J. Seykora for expertise. These studies were supported in part by grants CA123530 and AR44232 to P.A.C., fellowship grant F32 CA110618 to D.D. and grant CA087837 to A.A.D., all from the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
D.D. conceived and led the execution of all experiments and participated in the interpretation of the results and manuscript production. M.L.K. contributed expertise about inflammatory and immune cytokines and assisted D.D. in the execution and interpretation of many experiments.
A.A.D. contributed expertise on mouse skin tumor models and skin tumor histology and participated in manuscript production. P.A.C. conceived the experiments along with D.D. and participated in the interpretation of the results and manuscript production.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Table 1 (PDF 1237 kb)
Rights and permissions
About this article
Cite this article
DePianto, D., Kerns, M., Dlugosz, A. et al. Keratin 17 promotes epithelial proliferation and tumor growth by polarizing the immune response in skin. Nat Genet 42, 910–914 (2010). https://doi.org/10.1038/ng.665
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.665
This article is cited by
-
Interference KRT17 reverses doxorubicin resistance in triple-negative breast cancer cells by Wnt/β-catenin signaling pathway
Genes & Genomics (2023)
-
B7-H4 is a potential diagnostic and prognostic biomarker in colorectal cancer and correlates with the epithelial-mesenchymal transition
BMC Cancer (2022)
-
A stem cell population at the anorectal junction maintains homeostasis and participates in tissue regeneration
Nature Communications (2021)
-
Indirubin inhibits Wnt/β-catenin signal pathway via promoter demethylation of WIF-1
BMC Complementary Medicine and Therapies (2020)
-
Incorporation of differentiated dysplasia improves prediction of oral leukoplakia at increased risk of malignant progression
Modern Pathology (2020)