Abstract
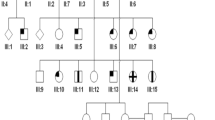
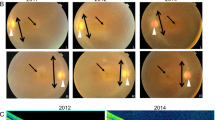
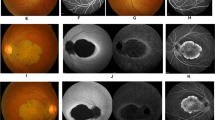
Achromatopsia (ACHM) is an autosomal recessive disorder characterized by color blindness, photophobia, nystagmus and severely reduced visual acuity. Using homozygosity mapping and whole-exome and candidate gene sequencing, we identified ten families carrying six homozygous and two compound-heterozygous mutations in the ATF6 gene (encoding activating transcription factor 6A), a key regulator of the unfolded protein response (UPR) and cellular endoplasmic reticulum (ER) homeostasis. Patients had evidence of foveal hypoplasia and disruption of the cone photoreceptor layer. The ACHM-associated ATF6 mutations attenuate ATF6 transcriptional activity in response to ER stress. Atf6−/− mice have normal retinal morphology and function at a young age but develop rod and cone dysfunction with increasing age. This new ACHM-related gene suggests a crucial and unexpected role for ATF6A in human foveal development and cone function and adds to the list of genes that, despite ubiquitous expression, when mutated can result in an isolated retinal photoreceptor phenotype.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
Change history
15 June 2015
In the version of this article initially published online, the ATF6 protein alteration corresponding to the smaller aberrantly spliced band resulting from the variant c.1533+1G>C was incorrectly reported as p.Gly512Valfs*11 on page 3 of the PDF and in the legend for Figure 6. The correct protein alteration is p.Leu479Valfs*11. The same error occurred in Table 1 for the following five patients: CHRO593-IV:1, CHRO593-II:3, MOGL411-MOGL467-III:4, MOGL411-MOGL467-IV:1 and MOGL5414-II:1. The errors have been corrected for the print, PDF and HTML versions of this article.
References
Thiadens, A.A. et al. Progressive loss of cones in achromatopsia: an imaging study using spectral-domain optical coherence tomography. Invest. Ophthalmol. Vis. Sci. 51, 5952–5957 (2010).
Thomas, M.G. et al. Structural grading of foveal hypoplasia using spectral-domain optical coherence tomography a predictor of visual acuity? Ophthalmology 118, 1653–1660 (2011).
Genead, M.A. et al. Photoreceptor structure and function in patients with congenital achromatopsia. Invest. Ophthalmol. Vis. Sci. 52, 7298–7308 (2011).
Sundaram, V. et al. Retinal structure and function in achromatopsia: implications for gene therapy. Ophthalmology 121, 234–245 (2014).
Aboshiha, J. et al. A prospective longitudinal study of retinal structure and function in achromatopsia. Invest. Ophthalmol. Vis. Sci. 55, 5733–5743 (2014).
Kohl, S. et al. Mutations in the cone photoreceptor G-protein α–subunit gene GNAT2 in patients with achromatopsia. Am. J. Hum. Genet. 71, 422–425 (2002).
Aligianis, I.A. et al. Mapping of a novel locus for achromatopsia (ACHM4) to 1p and identification of a germline mutation in the α subunit of cone transducin (GNAT2). J. Med. Genet. 39, 656–660 (2002).
Chang, B. et al. A homologous genetic basis of the murine Cpfl1 mutant and human achromatopsia linked to mutations in the PDE6C gene. Proc. Natl. Acad. Sci. USA 106, 19581–19586 (2009).
Thiadens, A.A. et al. Homozygosity mapping reveals PDE6C mutations in patients with early-onset cone photoreceptor disorders. Am. J. Hum. Genet. 85, 240–247 (2009).
Kohl, S. et al. A nonsense mutation in PDE6H causes autosomal-recessive incomplete achromatopsia. Am. J. Hum. Genet. 91, 527–532 (2012).
Kohl, S. et al. Total colourblindness is caused by mutations in the gene encoding the α-subunit of the cone photoreceptor cGMP-gated cation channel. Nat. Genet. 19, 257–259 (1998).
Sundin, O.H. et al. Genetic basis of total colourblindness among the Pingelapese islanders. Nat. Genet. 25, 289–293 (2000).
Kohl, S. et al. Mutations in the CNGB3 gene encoding the β-subunit of the cone photoreceptor cGMP-gated channel are responsible for achromatopsia (ACHM3) linked to chromosome 8q21. Hum. Mol. Genet. 9, 2107–2116 (2000).
Zhu, C., Johansen, F.E. & Prywes, R. Interaction of ATF6 and serum response factor. Mol. Cell. Biol. 17, 4957–4966 (1997).
Yoshida, H., Haze, K., Yanagi, H., Yura, T. & Mori, K. Identification of the cis-acting endoplasmic reticulum stress response element responsible for transcriptional induction of mammalian glucose-regulated proteins. Involvement of basic leucine zipper transcription factors. J. Biol. Chem. 273, 33741–33749 (1998).
Haze, K., Yoshida, H., Yanagi, H., Yura, T. & Mori, K. Mammalian transcription factor ATF6 is synthesized as a transmembrane protein and activated by proteolysis in response to endoplasmic reticulum stress. Mol. Biol. Cell 10, 3787–3799 (1999).
Walter, P. & Ron, D. The unfolded protein response: from stress pathway to homeostatic regulation. Science 334, 1081–1086 (2011).
Wang, S. & Kaufman, R.J. The impact of the unfolded protein response on human disease. J. Cell Biol. 197, 857–867 (2012).
Hai, T.W., Liu, F., Coukos, W.J. & Green, M.R. Transcription factor ATF cDNA clones: an extensive family of leucine zipper proteins able to selectively form DNA-binding heterodimers. Genes Dev. 3, 2083–2090 (1989).
Shoulders, M.D. et al. Stress-independent activation of XBP1s and/or ATF6 reveals three functionally diverse ER proteostasis environments. Cell Rep. 3, 1279–1292 (2013).
Tanimoto, N., Sothilingam, V. & Seeliger, M.W. Functional phenotyping of mouse models with ERG. Methods Mol. Biol. 935, 69–78 (2013).
Seeliger, M.W. et al. In vivo confocal imaging of the retina in animal models using scanning laser ophthalmoscopy. Vision Res. 45, 3512–3519 (2005).
Joly, S. et al. Cooperative phagocytes: resident microglia and bone marrow immigrants remove dead photoreceptors in retinal lesions. Am. J. Pathol. 174, 2310–2323 (2009).
Alexander, J.J. et al. Restoration of cone vision in a mouse model of achromatopsia. Nat. Med. 13, 685–687 (2007).
Michalakis, S. et al. Restoration of cone vision in the CNGA3−/− mouse model of congenital complete lack of cone photoreceptor function. Mol. Ther. 18, 2057–2063 (2010).
Komáromy, A.M. et al. Gene therapy rescues cone function in congenital achromatopsia. Hum. Mol. Genet. 19, 2581–2593 (2010).
Carvalho, L.S. et al. Long-term and age-dependent restoration of visual function in a mouse model of CNGB3-associated achromatopsia following gene therapy. Hum. Mol. Genet. 20, 3161–3175 (2011).
Wu, J. et al. ATF6α optimizes long-term endoplasmic reticulum function to protect cells from chronic stress. Dev. Cell 13, 351–364 (2007).
Wang, Y. et al. Activation of ATF6 and an ATF6 DNA binding site by the endoplasmic reticulum stress response. J. Biol. Chem. 275, 27013–27020 (2000).
Roy, B. & Lee, A.S. The mammalian endoplasmic reticulum stress response element consists of an evolutionarily conserved tripartite structure and interacts with a novel stress-inducible complex. Nucleic Acids Res. 27, 1437–1443 (1999).
Thuerauf, D.J., Morrison, L.E., Hoover, H. & Glembotski, C.C. Coordination of ATF6-mediated transcription and ATF6 degradation by a domain that is shared with the viral transcription factor, VP16. J. Biol. Chem. 277, 20734–20739 (2002).
Desterro, J.M., Rodriguez, M.S. & Hay, R.T. Regulation of transcription factors by protein degradation. Cell. Mol. Life Sci. 57, 1207–1219 (2000).
Thuerauf, D.J., Morrison, L. & Glembotski, C.C. Opposing roles for ATF6α and ATF6β in endoplasmic reticulum stress response gene induction. J. Biol. Chem. 279, 21078–21084 (2004).
Haze, K. et al. Identification of the G13 (cAMP-response-element-binding protein-related protein) gene product related to activating transcription factor 6 as a transcriptional activator of the mammalian unfolded protein response. Biochem. J. 355, 19–28 (2001).
Yoshida, H. et al. Endoplasmic reticulum stress-induced formation of transcription factor complex ERSF including NF-Y (CBF) and activating transcription factors 6α and 6β that activates the mammalian unfolded protein response. Mol. Cell. Biol. 21, 1239–1248 (2001).
Yamamoto, K. et al. Transcriptional induction of mammalian ER quality control proteins is mediated by single or combined action of ATF6α and XBP1. Dev. Cell 13, 365–376 (2007).
Rutkowski, D.T. et al. UPR pathways combine to prevent hepatic steatosis caused by ER stress-mediated suppression of transcriptional master regulators. Dev. Cell 15, 829–840 (2008).
Arensdorf, A.M., Dezwaan McCabe, D., Kaufman, R.J. & Rutkowski, D.T. Temporal clustering of gene expression links the metabolic transcription factor HNF4α to the ER stress-dependent gene regulatory network. Front. Genet. 4, 188 (2013).
Thuerauf, D.J., Marcinko, M., Belmont, P.J. & Glembotski, C.C. Effects of the isoform-specific characteristics of ATF6α and ATF6β on endoplasmic reticulum stress response gene expression and cell viability. J. Biol. Chem. 282, 22865–22878 (2007).
Marmor, M.F. et al. International Society for Clinical Electrophysiology of Vision. ISCEV standard for full-field clinical electroretinography (2008 update). Doc. Ophthalmol. 118, 69–77 (2009).
Hood, D.C. et al. International Society For Clinical Electrophysiology of Vision. ISCEV standard for clinical multifocal electroretinography (mfERG) (2011 edition). Doc. Ophthalmol. 124, 1–13 (2012).
Park, S.P. et al. Disruption of the human cone photoreceptor mosaic from a defect in NR2E3 transcription factor function in young adults. Graefes Arch. Clin. Exp. Ophthalmol. 251, 2299–2309 (2013).
Pyo Park, S., Hwan Hong, I., Tsang, S.H. & Chang, S. Cellular imaging demonstrates genetic mosaicism in heterozygous carriers of an X-linked ciliopathy gene. Eur. J. Hum. Genet. 21, 1240–1248 (2013).
Park, S.P. et al. Early structural anomalies observed by high-resolution imaging in two related cases of autosomal-dominant retinitis pigmentosa. Ophthalmic Surg. Lasers Imaging Retina 45, 469–473 (2014).
Hong, I.H. et al. Cone photoreceptor abnormalities correlate with vision loss in a case of acute posterior multifocal placoid pigment epitheliopathy. Ophthalmic Surg. Lasers Imaging Retina 45, 74–78 (2014).
Hoffmann, K. & Lindner, T.H. easyLINKAGE Plus—automated linkage analyses using large-scale SNP data. Bioinformatics 21, 3565–3567 (2005).
Seelow, D., Schuelke, M., Hildebrandt, F. & Nürnberg, P. HomozygosityMapper—an interactive approach to homozygosity mapping. Nucleic Acids Res. 37, W593–W599 (2009).
Fischer, M.D. et al. Noninvasive, in vivo assessment of mouse retinal structure using optical coherence tomography. PLoS ONE 4, e7507 (2009).
Acknowledgements
We want to thank C.W. Seok for data analysis. These studies were supported by various grants to the different authors and institutions: Bundesministerium für Bildung und Forschung (BMBF) grant 01GM1108A to B.W. and S.K.; US National Institutes of Health grants EY001919 and EY020846 to J.H.L. and DK042394, DK088227 and HL052173 to R.J.K. and a post-doctoral Foundation Fighting Blindness fellowship to W.-C.C.; National Institute for Health Research, Biomedical Research Centre at Moorfields Eye Hospital, National Health Service (NHS) Foundation Trust and University College London Institute of Ophthalmology, Fight For Sight, Moorfields Eye Hospital Special Trustees, Retinitis Pigmentosa Fighting Blindness and the Foundation Fighting Blindness (US) all to A.T.M., M.M. and A.R.W.; and the Wellcome Trust (099173/Z/12/Z) to M.M. and A.R.W. M.M. is supported by a Foundation Fighting Blindness Career Development Award; Mira Godard Research fund to E.H.; the imaging facilities at the Barbara and Donald Jonas Laboratory of Stem Cells and Regenerative Medicine and the Bernard and Shirlee Brown Glaucoma Laboratory are supported by Cannon, US National Institutes of Health Core grant 5P30EY019007, National Cancer Institute Core grant 5P30CA013696 and unrestricted funds from Research to Prevent Blindness (RPB), a Columbia University, New York RPB Physician-Scientist Award, the Schneeweiss Stem Cell Fund, New York State (N09G-302 and N13G-275) and the Gebroe Family Foundation, grant R01EY018213 to S.H.T.; Foundation Fighting Blindness (US) grants BR-GE-0510-0489-RAD to A.I.d.H. and C-GE-0811-0545-RAD01 to F.P.M.C., the Prof. Dr. H.J. Flieringa Foundation Stichting Wetenschappelijk Onderzoek het Oogziekenhuis (SWOO) and the Rotterdam Eye Hospital to F.P.M.C. and A.I.d.H. E.Z. is supported by Center for Integrative Neuroscience–DFG Center of Excellence EXC 307, University of Tübingen, Germany. R.K.K. is supported by the Foundation Fighting Blindness (Canada) and the CIHR (Canadian Institutes for Health Research).
Author information
Authors and Affiliations
Contributions
S.K., B.W., J.H.L. and R.J.K. conceived and designed the project and analyzed and interpreted data. D.Z., F.S., F.B., F.I., E.H., A.V., J.B., G.R., A.T.M., A.W., M.M., R.K.K., E.Z. and S.H.T. provided clinical data collection and interpretation. N.W., J.S., W.-C.C., S.R., A.I.d.H., F.P.M.C., I.L. and H.R. designed and performed experiments and analyzed and interpreted data. Specifically, N.W. performed cDNA analysis and haplotyping. J.S. performed all candidate gene sequencing. I.G.M. performed mouse retinal histology. T.M.S. was responsible for exome sequencing. S.C. and S.H.T. provided the AOSLO data. S.C.B., M.G.G., V.S. and M.W.S. provided the in vivo morphological and functional analyses of the mouse model, data generation and analysis, and writing of the manuscript. S.K., J.H.L. and D.Z. drafted the manuscript. M.M., R.K.K., E.H., A.V., A.T.M., A.W., M.M. and R.K.K. critically revised the manuscript for intellectual content. All authors discussed the results and commented on the manuscript. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Electropherograms of all identified mutations in ATF6.
Mutant sequence (top) compared to wild-type sequence (bottom). Nucleotide and protein sequence (one-letter code) are presented beneath the electropherogram. Exonic sequence is given in uppercase letters, and intronic sequence is given in lowercase letters. Arrows indicate the mutation.
Supplementary Figure 2 Comparative sequence analysis showing conservation of ATF6A at the protein level.
Comparative sequence analysis showing conservation of ATF6A at the protein level according to HomoloGene. In addition, alignment with human ATF6B is shown on the last line for comparison. Top, protein alignment for the p.Arg324Cys missense mutation. Bottom, protein alignment for the p.Tyr567Asn mutation.
Supplementary Figure 3 Atf6−/− mouse whole-mount and biochemistry data.
(a) Whole-mount retina preparations from 1-year-old Atf6+/– and Atf6−/− mice were stained with FITC-PNA (left immunofluorescent images), and the numbers of FITC-PNA–positive cone cells were quantified with Keyence BZ image analysis software for eight different eyes. Scale bar, 500 µm. (b–d) Whole retinas were collected and lysed from 90-d-old wild-type, Atf6+/– and Atf6−/− mice (n = 4 per genotype). (b) Cone-specific proteins (M opsin, S opsin and glycogen phosphorylase), (c) rod-specific proteins (rhodopsin and Gαt1 / rod transducin) and (d) ER stress-induced proteins (PDI, calreticulin and BiP (Grp78)) were detected by immunoblotting. HSP90 served as a protein loading control. WT, wild type; FITC, fluorescein isothiocyanate; PNA, peanut agglutinine.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Tables 1–3. (PDF 3096 kb)
Rights and permissions
About this article
Cite this article
Kohl, S., Zobor, D., Chiang, WC. et al. Mutations in the unfolded protein response regulator ATF6 cause the cone dysfunction disorder achromatopsia. Nat Genet 47, 757–765 (2015). https://doi.org/10.1038/ng.3319
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.3319
This article is cited by
-
Imbalanced unfolded protein response signaling contributes to 1-deoxysphingolipid retinal toxicity
Nature Communications (2023)
-
Endoplasmic reticulum stress: molecular mechanism and therapeutic targets
Signal Transduction and Targeted Therapy (2023)
-
Cellular stress signaling and the unfolded protein response in retinal degeneration: mechanisms and therapeutic implications
Molecular Neurodegeneration (2022)
-
Achromatopsia: Genetics and Gene Therapy
Molecular Diagnosis & Therapy (2022)
-
ATF6 is required for efficient rhodopsin clearance and retinal homeostasis in the P23H rho retinitis pigmentosa mouse model
Scientific Reports (2021)