Abstract
Purpose
In this study, we described a large family presenting different manifestations of cone dystrophy at different ages associated with GUCY2D gene mutation.
Method
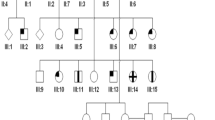
Sixty-three individuals of a single kindred, including 23 affected with cone dystrophies, were recruited and received ocular examinations, including best corrected visual acuity, intraocular pressure, slit-lamp biomicroscopy, color fundus photograph (CFP), fundus autofluorescence, optical coherence tomography, fluorescence fundus angiography, color vision testing, full-field electroretinography, and electro-oculogram. Whole exome sequencing (WES) and Sanger sequencing were performed for underlying mutations associated with cone dystrophy.
Result
There were 23 affected family members. Clinical analysis showed that the proband and other patients had impaired visual acuity ranging from 20/800 to 20/50 with impaired color vision. Fundus photograph showed retinal pigment epithelium (RPE) granular abnormalities with depressed macular reflex in young patients and macular or retinochoriodal atrophy in older patients. OCT examination confirmed the reduced outer retinal thickness or inner retinal thickness, absence of the ellipsoid zone (EZ) and retinal atrophy to varying degrees. Electroretinography revealed a reduced cone response combined with a relatively maintained rod response. WES and Sanger sequencing revealed a heterozygous variant c.2512C>T in the GUCY2D gene of the affected family members.
Conclusions
We reported cone dystrophy in 23 affected individuals in a five-generation family and demonstrated different macular abnormalities in OCT scans and CFP at different ages. The multimodal ocular records in our study provide physicians and ophthalmologists with a better understanding of cone dystrophy associated with GUCY2D mutation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 18 print issues and online access
$259.00 per year
only $14.39 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
References
Hamel CP. Cone rod dystrophies. Orphanet J Rare Dis. 2007;2:7.
Prokofyeva E, Troeger E, Bernd A, Zrenner E. Visual acuity changes in cone and cone-rod dystrophies. Ophthalmic Physiol Opt. 2012;32:53–59.
Boulanger-Scemama E, Mohand-Said S, El Shamieh S, Demontant V, Condroyer C, Antonio A, et al., Phenotype analysis of retinal dystrophies in light of the underlying genetic defects: application to cone and cone-rod dystrophies. Int J Mol Sci. 2019;20:4854.
Michaelides M, Hardcastle AJ, Hunt DM, Moore AT. Progressive cone and cone-rod dystrophies: phenotypes and underlying molecular genetic basis. Surv Ophthalmol. 2006;51:232–58.
Gill JS, Georgiou M, Kalitzeos A, Moore AT, Michaelides M. Progressive cone and cone-rod dystrophies: clinical features, molecular genetics and prospects for therapy. Br J Ophthalmol. 2019;103:711–20.
Toto L, Battaglia Parodi M, D’Aloisio R, Mercuri S, Senatore A, Di Antonio L, et al. Cone dystrophies: an optical coherence tomography angiography study. J Clin Med. 2020;9:1500.
Oishi M, Oishi A, Gotoh N, Ogino K, Higasa K, Iida K, et al. Next-generation sequencing-based comprehensive molecular analysis of 43 Japanese patients with cone and cone-rod dystrophies. Mol Vis. 2016;22:150–60.
Watson CM, El-Asrag M, Parry DA, Morgan JE, Logan CV, Carr IM, et al. Mutation screening of retinal dystrophy patients by targeted capture from tagged pooled DNAs and next generation sequencing. PloS one. 2014;9:e104281.
Kitiratschky VB, Wilke R, Renner AB, Kellner U, Vadala M, Birch DG, et al. Mutation analysis identifies GUCY2D as the major gene responsible for autosomal dominant progressive cone degeneration. Invest Ophthalmol Vis Sci. 2008;49:5015–23.
Dizhoor AM, Olshevskaya EV, Peshenko IV. The R838S mutation in retinal guanylyl cyclase 1 (RetGC1) alters calcium sensitivity of cGMP synthesis in the retina and causes blindness in transgenic mice. J Biol Chem. 2016;291:24504–16.
Sato S, Peshenko IV, Olshevskaya EV, Kefalov VJ, Dizhoor AM, Cone-Rod GUCY2D. Dystrophy-6 is a “phototransduction disease” triggered by abnormal calcium feedback on retinal membrane guanylyl cyclase 1. J Neurosci. 2018;38:2990–3000.
Hughes AE, Meng W, Lotery AJ, Bradley DT. A novel GUCY2D mutation, V933A, causes central areolar choroidal dystrophy. Invest Ophthalmol Vis Sci. 2012;53:4748–53.
Sun Z, Wu S, Zhu T, Li H, Wei X, Du H, et al. Variants at codon 838 in the GUCY2D gene result in different phenotypes of cone rod dystrophy. Ophthalmic Genet. 2020;41:548–55.
Sharon D, Wimberg H, Kinarty Y, Koch KW. Genotype-functional-phenotype correlations in photoreceptor guanylate cyclase (GC-E) encoded by GUCY2D. Prog retinal eye Res. 2018;63:69–91.
Hosono K, Harada Y, Kurata K, Hikoya A, Sato M, Minoshima S, et al. Novel GUCY2D gene mutations in japanese male twins with leber congenital amaurosis. J Ophthalmol. 2015;2015:693468.
Stunkel ML, Brodie SE, Cideciyan AV, Pfeifer WL, Kennedy EL, Stone EM, et al. Expanded retinal disease spectrum associated with autosomal recessive mutations in GUCY2D. Am J Ophthalmol. 2018;190:58–68.
Song J, Smaoui N, Ayyagari R, Stiles D, Benhamed S, MacDonald IM, et al. High-throughput retina-array for screening 93 genes involved in inherited retinal dystrophy. Invest Ophthalmol Vis Sci. 2011;52:9053–60.
Liu X, Tao T, Zhao L, Li G, Yang L. Molecular diagnosis based on comprehensive genetic testing in 800 Chinese families with non-syndromic inherited retinal dystrophies. Clin Exp Ophthalmol. 2021;49(1):46–59.
Collery RF, Cederlund ML, Kennedy BN. Transgenic zebrafish expressing mutant human RETGC-1 exhibit aberrant cone and rod morphology. Exp eye Res. 2013;108:120–8.
Wilkie SE, Newbold RJ, Deery E, Walker CE, Stinton I, Ramamurthy V, et al. Functional characterization of missense mutations at codon 838 in retinal guanylate cyclase correlates with disease severity in patients with autosomal dominant cone-rod dystrophy. Hum Mol Genet. 2000;9:3065–73.
Garcia-Hoyos M, Auz-Alexandre CL, Almoguera B, Cantalapiedra D, Riveiro-Alvarez R, Lopez-Martinez MA, et al. Mutation analysis at codon 838 of the Guanylate Cyclase 2D gene in Spanish families with autosomal dominant cone, cone-rod, and macular dystrophies. Mol Vis. 2011;17:1103–9.
Perrault I, Rozet JM, Gerber S, Kelsell RE, Souied E, Cabot A, et al. A retGC-1 mutation in autosomal dominant cone-rod dystrophy. Am J Hum Genet. 1998;63:651–4.
Payne AM, Morris AG, Downes SM, Johnson S, Bird AC, Moore AT, et al. Clustering and frequency of mutations in the retinal guanylate cyclase (GUCY2D) gene in patients with dominant cone-rod dystrophies. J Med Genet. 2001;38:611–4.
Smith M, Whittock N, Searle A, Croft M, Brewer C, Cole M. Phenotype of autosomal dominant cone-rod dystrophy due to the R838C mutation of the GUCY2D gene encoding retinal guanylate cyclase-1. Eye. 2007;21:1220–5.
Van Ghelue M, Eriksen HL, Ponjavic V, Fagerheim T, Andreasson S, Forsman-Semb K, et al. Autosomal dominant cone-rod dystrophy due to a missense mutation (R838C) in the guanylate cyclase 2D gene (GUCY2D) with preserved rod function in one branch of the family. Ophthalmic Genet. 2000;21:197–209.
Lima LH, Sallum JM, Spaide RF. Outer retina analysis by optical coherence tomography in cone-rod dystrophy patients. Retina. 2013;33:1877–80.
Acknowledgements
We wish to thank this family for participating in our study.
Funding
This work was financially supported by The Project of National Key Research and Development (No. 2018YFC1106103) to MZ and Post-Doctor Research Project, West China Hospital, Sichuan University (No.2021HXBH030), Natural Science Foundation of Sichuan Province (No. 2022NSFSC1285) to XR.
Author information
Authors and Affiliations
Contributions
YG, and MZ designed the study, directed the project, and interpreted the data. KL, LX, HL, ZZ, XW, RR, YT and YG performed the experiments. NY provided guidance for this project. XR wrote the paper, and YL, XF, NY, and MZ contributed to editing. All authors contributed to the paper and approved the submitted version.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gao, Y., Ren, X., Lin, H. et al. Phenotypic characterization of autosomal dominant progressive cone dystrophies associated with a heterozygous variant c.2512C>T of GUCY2D gene in a large kindred. Eye 37, 2461–2469 (2023). https://doi.org/10.1038/s41433-022-02355-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41433-022-02355-1