Abstract
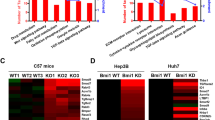
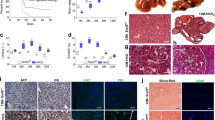
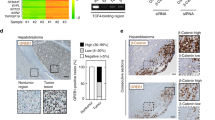
Hepatocellular carcinoma (HCC) is a major cause of death worldwide. Here, we provide evidence that the ligand-dependent nuclear receptor co-regulator Trim24 (also known as Tif1α) functions in mice as a liver-specific tumor suppressor. In Trim24-null mice, hepatocytes fail to execute proper cell cycle withdrawal during the neonatal-to-adult transition and continue to cycle in adult livers, becoming prone to a continuum of cellular alterations that progress toward metastatic HCC. Using pharmacological approaches, we show that inhibition of retinoic acid signaling markedly reduces hepatocyte proliferation in Trim24−/− mice. We further show that deletion of a single retinoic acid receptor alpha (Rara) allele in a Trim24-null background suppresses HCC development and restores wild-type expression of retinoic acid–responsive genes in the liver, thus demonstrating that in this genetic background Rara expresses an oncogenic activity correlating with a dysregulation of the retinoic acid signaling pathway. Our results not only provide genetic evidence that Trim24 and Rara co-regulate hepatocarcinogenesis in an antagonistic manner but also suggest that aberrant activation of Rara is deleterious to liver homeostasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Soprano, D.R., Quin, P. & Soprano, K.J. Retinoic acid receptors and cancers. Annu. Rev. Nutr. 24, 201–221 (2004).
Mark, M., Ghyselinck, N.B. & Chambon, P. Function of retinoid nuclear receptors: lessons from genetic and pharmacological dissections of the retinoic acid signaling pathway during mouse embryogenesis. Annu. Rev. Pharmacol. Toxicol. 46, 451–480 (2006).
Rosenfeld, M.G. & Glass, C.K. Coregulator codes of transcriptional regulation by nuclear receptors. J. Biol. Chem. 276, 36865–36868 (2001).
Le Douarin, B., Zechel, C., Garnier, J.-M., Lutz, Y., Tora, L., Pierrat, B., Heery, D., Gronemeyer, H., Chambon, P. & Losson, R. The N-terminal part of TIF1, a putative mediator of the ligand-dependent activation function (AF-2) of nuclear receptors, is fused to B-raf in the oncogenic protein T18. EMBO J. 14, 2020–2033 (1995).
Zhong, S. et al. A RA-dependent, tumor-growth suppressive transcription complex is the target of the PML-RARα and T18 oncoproteins. Nat. Genet. 23, 287–295 (1999).
Teyssier, C., Ou, C-Y., Khetchoumian, K., Losson, R. & Stallcup, M.R. TIF1α mediates physical interaction and functional synergy between the CARM1 and GRIP1 nuclear receptor coactivators. Mol. Endocrinol. 20, 1276–1286 (2006).
Venturini, L. et al. TIF1γ, a novel member of the transcriptional intermediary factor 1 family. Oncogene 18, 1209–1217 (1999).
Le Douarin, B. et al. A possible involvement of TIF1α and TIF1β in the epigenetic control of transcription by nuclear receptors. EMBO J. 15, 6701–6715 (1996).
Fausto, N. Mouse liver tumorigenesis: models, mechanisms, and relevance to human disease. Semin. Liver Dis. 19, 243–252 (1999).
Wu, H. et al. Targeted in vivo expression of the cyclin-dependent kinase inhibitor p21 halts hepatocyte cell-cycle progression, postnatal liver development, and regenration. Genes Dev. 10, 245–260 (1996).
Scholzen, T. & Gerdes, J. The Ki-67 protein: from the known and the unknown. J. Cell. Physiol. 182, 311–322 (2000).
Ledda-Columbano, G.M. et al. Induction of hepatocyte proliferation by retinoic acid. Carcinogenesis 25, 2061–2066 (2004).
Di Bisceglie, A.M., Osmack, P. & Brunt, E.M. Chemoprevention of hepatocellular carcinoma: use of tamoxifen in an animal model of hepatocarcinogenesis. J. Lab. Clin. Med. 145, 134–138 (2005).
Sano, K., Takayama, T., Murakami, K., Saiki, I. & Makuuchi, M. Overexpression of retinoic acid receptor a in hepatocellular carcinoma. Clin. Cancer Res. 9, 3679–3683 (2003).
Postic, C. et al. Dual roles for glucokinase in glucose homeostasis as determined by liver and pancreatic beta cell-specific gene knock-outs using Cre recombinase. J. Biol. Chem. 274, 305–315 (1999).
Germain, P. et al. International Union of Pharmacology. LX. Retinoic acid receptors. Pharmacol. Rev. 58, 712–725 (2006).
Balmer, J.E. & Blomhoff, R. Gene expression regulation by retinoic acid. J. Lipid Res. 43, 1773–1808 (2002).
Sun, S-Y. & Lotan, R. Retinoids and their receptors in cancer development and chemoprevention. Crit. Rev. Oncol. Hematol. 41, 41–55 (2002).
Lufkin, T. et al. High postnatal lethality and testis degeneration in retinoic acid receptor alpha mutant mice. Proc. Natl. Acad. Sci. USA 90, 7225–7229 (1993).
Chapellier, B. et al. A conditional floxed (loxP-flanked) allele for the retinoic acid receptor alpha (RARalpha) gene. Genesis 32, 87–90 (2002).
Frith, C.H., Ward, J.M. & Turosov, V.S. in Pathology of Tumors in Laboratory Animals Vol. 2 (eds. Turosov, V. & Mohr, U.) 223–270 (IARC Scientific, Lyon, France, 1994).
Khetchoumian, K. et al. TIF1δ, a novel HP1-interacting member of the transcriptional intermediary factor 1 (TIF1) family expressed by elongating spermatids. J. Biol. Chem. 279, 48329–48341 (2004).
Acknowledgements
This letter is dedicated to the memory of Bertrand Le Douarin on the ninth anniversary of his premature death. He discovered and carried out the first molecular characterization of TIF1α. We thank M. Cerviño, X. Belin, J.-V. Fougerolle, L. Walsh and E. Alexandre for technical help. We also thank all the common facilities of the IGBMC and, in particular, C. Ebel and J. Barths for assistance in FACS analysis, D. Dembélé for help with statistical analyses, N. Ghyselinck for providing Rara mutant mice, C. Suzi and P. Reczek (Bristol-Myers Squibb) for the gift of BMS493, M. Oulad-Abdelghani for the gift of antibody to β-tubulin 1TUB1A2 and P. Kastner and P. Germain for helpful discussions. This work was supported by the French Centre National de la Recherche Scientifique, Institut National de la Santé et de la Recherche Médicale, and Agence Nationale de la Recherche, the Association pour la Recherche sur le Cancer (ARC) and the Collège de France. Special thanks to R.P. Vashakidze from Volzhsky Orgsynthese for the financial support of K.K. K.K. was also supported by the French Ministère de la Recherche et des Technologies, the Ligue Nationale contre le Cancer and the Armenian Foundation Boghos Noubar Pacha (Belgium). M.I. was supported by a Charcot fellowship from the French Ministère des Affaires étrangères.
Author information
Authors and Affiliations
Contributions
K.K. created and analyzed all knock-out mice. M.T. and M.I. performed the quantification and histopathological assessment of mouse tumor and proliferation phenotypes under supervision of M.M. J.T. carried out microarray, qRT-PCR and ChIP experiments. B.H. worked on the analysis of Trim24 on cell models under supervision of K.K. J.Z.-R. contributed to Trim24 expression analysis. T.L. and C.T. provided technical assistance with plasmid contruction and microarray work, respectively. P.C., F.C. and D.M. provided project supervision. R.L. provided the general concept, design of study, supervision and manuscript writing.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7, Supplementary Tables 1–3, Supplementary Methods (PDF 11955 kb)
Rights and permissions
About this article
Cite this article
Khetchoumian, K., Teletin, M., Tisserand, J. et al. Loss of Trim24 (Tif1α) gene function confers oncogenic activity to retinoic acid receptor alpha. Nat Genet 39, 1500–1506 (2007). https://doi.org/10.1038/ng.2007.15
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng.2007.15
This article is cited by
-
Inhibition of the TRIM24 bromodomain reactivates latent HIV-1
Scientific Reports (2023)
-
Readout of histone methylation by Trim24 locally restricts chromatin opening by p53
Nature Structural & Molecular Biology (2023)
-
Loss of TRIM24 promotes IL-10 expression via CBP/p300-dependent IFNβ1 transcription during macrophage activation
Inflammation Research (2023)
-
Targeting TRIM24 promotes neuroblastoma differentiation and decreases tumorigenicity via LSD1/CoREST complex
Cellular Oncology (2023)
-
TRIM proteins in hepatocellular carcinoma
Journal of Biomedical Science (2022)