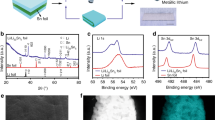
Abstract
Lithium metal is a highly desirable anode for lithium rechargeable batteries, having the highest theoretical specific capacity and lowest electrochemical potential of all material candidates. Its most notable problem is dendritic growth upon Li plating, which is a major safety concern and exacerbates reactivity with the electrolyte. Here we report that Li-rich composite alloy films synthesized in situ on lithium by a simple and low-cost methodology effectively prevent dendrite growth. This is attributed to the synergy of fast lithium ion migration through Li-rich ion conductive alloys coupled with an electronically insulating surface component. The protected lithium is stabilized to sustain electrodeposition over 700 cycles (1,400 h) of repeated plating/stripping at a practical current density of 2 mA cm−2 and a 1,500 cycle-life is realized for a cell paired with a Li4Ti5O12 positive electrode. These findings open up a promising avenue to stabilize lithium metal with surface layers having targeted properties.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Armand, M. & Tarascon, J. M. Building better batteries. Nature 451, 652–657 (2008).
Bruce, P. G., Freunberger, S. A., Hardwick, L. J. & Tarascon, J. M. Li–O2 and Li–S batteries with high energy storage. Nat. Mater. 11, 19–29 (2012).
Chen, Y., Freunberger, S. A., Peng, Z., Fontaine, O. & Bruce, P. G. Charging a Li–O2 battery using a redox mediator. Nat. Chem. 5, 489–494 (2013).
Lu, Y. C. et al. Lithium–oxygen batteries: bridging mechanistic understanding and battery performance. Energy Environ. Sci. 6, 750–768 (2013).
Pang, Q., Liang, X., Kwok, C. Y. & Nazar, L. F. Advances in lithium–sulfur batteries based on multifunctional cathodes and electrolytes. Nat. Energy 1, 16132 (2016).
Xu, W. et al. Lithium metal anodes for rechargeable batteries. Energy Environ. Sci. 7, 513–537 (2014).
Busche, M. R. et al. Dynamic formation of a solid–liquid electrolyte interphase and its consequences for hybrid-battery concepts. Nat. Chem. 8, 426–434 (2016).
Gallagher, K. G. et al. Quantifying the promise of lithium-air batteries for electric vehicles. Energy Environ. Sci. 7, 1555–1563 (2014).
Harry, K. J., Hallinan, D. T., Parkinson, D. Y., MacDowell, A. A. & Balsara, N. P. Detection of subsurface structures underneath dendrites formed on cycled lithium metal electrodes. Nat. Mater. 13, 69–73 (2014).
Aurbach, D. et al. Attempts to improve the behavior of Li electrodes in rechargeable lithium batteries. J. Electrochem. Soc. 149, A1267–A1277 (2002).
Qian, J. et al. High rate and stable cycling of lithium metal anode. Nat. Commun. 6, 6362 (2015).
Wang, H. et al. A reversible dendrite-free high-areal-capacity lithium metal electrode. Nat. Commun. 8, 15106 (2017).
Ding, F. et al. Dendrite-free lithium deposition via self-healing electrostatic shield mechanism. J. Am. Chem. Soc. 135, 4450–4456 (2013).
Gofer, Y., Ben-Zion, M. & Aurbach, D. Solutions of LiAsF in 1,3-dioxolane for secondary lithium batteries. J. Power Sources 39, 163–178 (1992).
Lu, Y., Tu, Z. & Archer, L. A. Stable lithium electrodeposition in liquid and nanoporous solid electrolytes. Nat. Mater. 13, 961–969 (2014).
Li, W. et al. The synergetic effect of lithium polysulfide and lithium nitrate to prevent lithium dendrite growth. Nat. Commun. 6, 7436 (2015).
Suo, L., Hu, Y. S., Li, H., Armand, M. & Chen, L. A new class of solvent-in-salt electrolyte for high-energy rechargeable metallic lithium batteries. Nat. Commun. 4, 2513 (2013).
Zheng, G. et al. Interconnected hollow carbon nanospheres for stable lithium metal anodes. Nat. Nanotech. 9, 618–623 (2014).
Kozen, A. C. et al. Next-generation lithium metal anode engineering via atomic layer deposition. ACS Nano 9, 5884–5892 (2015).
Bucur, C. B., Lita, A., Osada, N. & Muldoon, J. A. Soft, multilayered lithium–electrolyte interface. Energy Environ. Sci. 9, 112–118 (2016).
Umeda, G. et al. Protection of lithium metal surfaces using tetraethoxysilane. J. Mater. Chem. 21, 1593–1599 (2011).
Yang, C., Yin, Y. X., Zhang, S. F., Liu, N. W. & Guo, Y. Accommodating lithium into 3D current collectors with a submicron skeleton towards long-life lithium metal anodes. Nat. Commun. 6, 8058 (2015).
Zhang, R. et al. Conductive nanostructured scaffolds render low local current density to inhibit lithium dendrite growth. Adv. Mater. 28, 2155–2162 (2015).
Lin, D. et al. Layered reduced graphene oxide with nanoscale interlayer gaps as a stable host for lithium metal anodes. Nat. Nanotech. 11, 626–632 (2016).
Armand, M. B., Duclot, M. J. & Rigaud, P. Polymer solid electrolytes: stability domain. Solid State Ion. 3–4, 429–430 (1981).
Zhou, W. et al. Plating a dendrite-free lithium anode with a polymer/ceramic/polymer sandwich electrolyte. J. Am. Chem. Soc. 138, 9385–9388 (2016).
Khurana, R., Schaefer, J. L., Archer, L. A. & Coates, G. W. Suppression of lithium dendrite growth using cross-linked polyethylene/poly(ethylene oxide) electrolytes: a new approach for practical lithium-metal polymer batteries. J. Am. Chem. Soc. 136, 7395–7402 (2014).
Wenzel, S. et al. Direct observation of the interfacial instability of the fast ionic conductor Li10GeP2S12 at the lithium metal anode. Nat. Chem. 28, 2400–2407 (2016).
Ren, Y., Shen, Y., Lin, Y. & Nan, C. Direct observation of lithium dendrites inside garnet-type lithium-ion solid electrolyte. Electrochem. Commun. 57, 27–30 (2015).
Richardson, T. J. & Chen, G. Solid solution lithium alloy cermet anodes. J. Power Sources 174, 810–812 (2007).
Stark, J. K., Ding, Y. & Kohl, P. A. Dendrite-free electrodeposition and reoxidation of lithium–sodium alloy for metal-anode battery. J. Electrochem. Soc. 158, A1100–A1105 (2011).
Shi, Z., Liu, M. & Gole, J. L. Electrochemical properties of Li–Zn alloy electrodes prepared by kinetically controlled vapor deposition for lithium batteries. Electrochem. Sol. State Lett. 3, 312–315 (2000).
Hiratani, M., Miyauchi, K. & Kudo, T. Effect of a lithium alloy layer inserted between a lithium anode and a solid electrolyte. Solid State Ion. 28–30, 1406–1410 (1988).
Hiratani, M. et al. Solid state lithium battery. US patent 4,645,726 (1987).
Zheng, G. et al. Interconnected hollow carbon nanospheres for stable lithium metal anodes. Nat. Nanotech. 9, 618–623 (2014).
Kim, J. S., Kim, D. W., Jung, H. T. & Choi, J. W. Controlled lithium dendrite growth by a synergistic effect of multilayered graphene coating and an electrolyte additive. Chem. Mater. 27, 2780–2787 (2015).
Yan, K. et al. Ultrathin two-dimensional atomic crystals as stable interfacial layer for improvement of lithium metal anode. Nano Lett. 14, 6016–6022 (2014).
Web, S. A., Baggetto, L., Bridges, C. A. & Veith, G. M. The electrochemical reactions of pure indium with Li and Na: anomalous electrolyte decomposition, benefits of FEC additive, phase transitions and electrode performance. J. Power Sources 248, 1105–1117 (2014).
Hewitt, R. W. & Winograd, N. Oxidation of polycrystalline indium studied by X-ray photoelectron spectroscopy and static secondary ion mass spectroscopy. J. Appl. Phys. 51, 2620–2624 (1980).
Kanamura, K., Tamura, H., Shiraishi, S. & Takehara, Z. XPS analysis of lithium surfaces following immersion in various solvents containing LiBF4 . J. Electrochem. Soc. 142, 340–347 (1995).
Dologlou, E. Self diffusion in solid lithium. Glass Phys. Chem. 36, 570–574 (2010).
Park, M., Zhang, X., Chung, M., Less, G. B. & Sastry, A. M. A review of conduction phenomena in Li-ion batteries. J. Power Sources 195, 7904–7929 (2010).
Huggins, R. A. Polyphase alloys as rechargeable electrodes in advanced battery systems. J. Power Sources 22, 341–350 (1988).
Aurbach, D., Zinigrad, E., Cohen, Y. & Teller, H. A short review of failure mechanisms of lithium metal and lithiated graphite anodes in liquid electrolyte solutions. Solid State Ion. 148, 405–416 (2002).
Ohzuku, T., Ueda, A. & Yamamoto, N. Zero-strain insertion material of Li[Li1∕3Ti5∕3]O4 for rechargeable lithium cells. J. Electrochem. Soc. 142, 1431–1435 (1995).
Acknowledgements
This research was supported by the BASF International Scientific Network for Electrochemistry and Batteries. L.F.N. also thanks NSERC for generous support via their Canada Research Chair, and Discovery Grant programs. We greatly appreciate helpful discussions with K. Zavadil, P. Bruce and J. Janek.
Author information
Authors and Affiliations
Contributions
X.L. and L.F.N. designed the experimental work. X.L. performed all the physical measurements on the films and the electrochemistry on symmetric and full cells. I.R.K. and Q.P. carried out the resistivity measurements of the protective layers. M.S.S., H.H. and X.S. participated in the discussion of the data. X.L., I.R.K. and L.F.N. wrote the manuscript. L.F.N. directed the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–12. (PDF 1327 kb)
Rights and permissions
About this article
Cite this article
Liang, X., Pang, Q., Kochetkov, I. et al. A facile surface chemistry route to a stabilized lithium metal anode. Nat Energy 2, 17119 (2017). https://doi.org/10.1038/nenergy.2017.119
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nenergy.2017.119
This article is cited by
-
An extended substrate screening strategy enabling a low lattice mismatch for highly reversible zinc anodes
Nature Communications (2024)
-
Transition metals for stabilizing lithium metal anode: advances and perspectives
Tungsten (2024)
-
Constructing a fluorinated interface layer enriched with Ge nanoparticles and Li-Ge alloy for stable lithium metal anodes
Nano Research (2024)
-
Post lithium-sulfur battery era: challenges and opportunities towards practical application
Science China Chemistry (2024)
-
Machine-learning-assisted design of a binary descriptor to decipher electronic and structural effects on sulfur reduction kinetics
Nature Catalysis (2023)