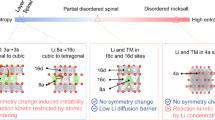
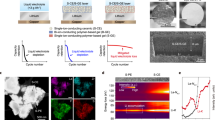
Abstract
Lithium-ion batteries based on intercalation compounds have dominated the advanced portable energy storage market. The positive electrode materials in these batteries belong to a material group of lithium-conducting crystals that contain redox-active transition metal and lithium. Materials without lithium-conducting paths or lithium-free compounds could be rarely used as positive electrodes due to the incapability of reversible lithium intercalation or the necessity of using metallic lithium as negative electrodes. These constraints have significantly limited the choice of materials and retarded the development of new positive electrodes in lithium-ion batteries. Here, we demonstrate that lithium-free transition metal monoxides that do not contain lithium-conducting paths in their crystal structure can be converted into high-capacity positive electrodes in the electrochemical cell by initially decorating the monoxide surface with nanosized lithium fluoride. This unusual electrochemical behaviour is attributed to a surface conversion reaction mechanism in contrast with the classic lithium intercalation reaction. Our findings will offer a potential new path in the design of positive electrode materials in lithium-ion batteries.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Larcher, D. & Tarascon, J. M. Towards greener and more sustainable batteries for electrical energy storage. Nat. Chem. 7, 19–29 (2015).
Sathiya, M. et al. Reversible anionic redox chemistry in high-capacity layered-oxide electrodes. Nat. Mater. 12, 827–835 (2013).
Lee, J. et al. Unlocking the potential of cation-disordered oxides for rechargeable lithium batteries. Science 343, 519–522 (2014).
Kim, S.-W. et al. Energy storage in composites of a redox couple host and a lithium ion host. Nano Today 7, 168–173 (2012).
Tarascon, J. M. & Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 414, 359–367 (2001).
Tarascon, J. M., Wang, E., Shokoohi, F. K., McKinnon, W. R. & Colson, S. The spinel phase of LiMn2O4 as a cathode in secondary lithium cells. J. Electrochem. Soc. 138, 2859–2864 (1991).
Padhi, A. K., Nanjundaswamy, K. S. & Goodenough, J. B. Phospho-olivines as positive-electrode materials for rechargeable lithium batteries. J. Electrochem. Soc. 144, 1188–1194 (1997).
Mizushima, K., Jones, P. C., Wiseman, P. J. & Goodenough, J. B. LixCoO2 (0 < x < −1): a new cathode material for batteries of high energy density. Mater. Res. Bull. 15, 783–789 (1980).
Poizot, P., Laruelle, S., Grugeon, S., Dupont, L. & Tarascon, J. M. Nano-sized transition-metal oxides as negative-electrode materials for lithium-ion batteries. Nature 407, 496–499 (2000).
de Faria, D. L. A., Venâncio Silva, S. & de Oliveira, M. T. Raman microspectroscopy of some iron oxides and oxyhydroxides. J. Raman Spectrosc. 28, 873–878 (1997).
Fang, X. et al. Electrode reactions of manganese oxides for secondary lithium batteries. Electrochem. Commun. 12, 1520–1523 (2010).
Sun, B., Chen, Z., Kim, H.-S., Ahn, H. & Wang, G. MnO/C core–shell nanorods as high capacity anode materials for lithium-ion batteries. J. Power Sources 196, 3346–3349 (2011).
Gao, M. et al. FeO/C anode materials of high capacity and cycle stability for lithium-ion batteries synthesized by carbothermal reduction. J. Alloys Compd. 565, 97–103 (2013).
Dimov, N., Kitajou, A., Hori, H., Kobayashi, E. & Okada, S. Electrochemical splitting of LiF: a new approach to lithium-ion battery materials. ECS Trans. 58, 87–99 (2014).
Augustyn, V. et al. High-rate electrochemical energy storage through Li+ intercalation pseudocapacitance. Nat. Mater. 12, 518–522 (2013).
Wang, J., Polleux, J., Lim, J. & Dunn, B. Pseudocapacitive contributions to electrochemical energy storage in TiO2 (anatase) nanoparticles. J. Phys. Chem. C 111, 14925–14931 (2007).
Brezesinski, T., Wang, J., Polleux, J., Dunn, B. & Tolbert, S. H. Templated nanocrystal-based porous TiO2 films for next-generation electrochemical capacitors. J. Am. Chem. Soc. 131, 1802–1809 (2009).
Dunn, B., Kamath, H. & Tarascon, J.-M. Electrical energy storage for the grid: a battery of choices. Science 334, 928–935 (2011).
Gilbert, B. et al. Multiple scattering calculations of bonding and X-ray absorption spectroscopy of manganese oxides. J. Phys. Chem. A 107, 2839–2847 (2003).
Qiao, R., Chin, T., Harris, S. J., Yan, S. & Yang, W. Spectroscopic fingerprints of valence and spin states in manganese oxides and fluorides. Curr. Appl. Phys. 13, 544–548 (2013).
Augustyn, V., Simon, P. & Dunn, B. Pseudocapacitive oxide materials for high-rate electrochemical energy storage. Energy Environ. Sci. 7, 1597–1614 (2014).
Kim, S.-W. et al. Structure stabilization by mixed anions in oxyfluoride cathodes for high-energy lithium batteries. ACS Nano 9, 10076–10084 (2015).
Wang, X.-L. et al. Amorphous hierarchical porous GeOx as high-capacity anodes for Li ion batteries with very long cycling life. J. Am. Chem. Soc. 133, 20692–20695 (2011).
Yamakawa, N., Jiang, M., Key, B. & Grey, C. P. Identifying the local structures formed during lithiation of the conversion material, iron fluoride, in a Li ion battery: a solid-state NMR, X-ray diffraction, and pair distribution function analysis study. J. Am. Chem. Soc. 131, 10525–10536 (2009).
Pereira, N., Badway, F., Wartelsky, M., Gunn, S. & Amatucci, G. G. Iron oxyfluorides as high capacity cathode materials for lithium batteries. J. Electrochem. Soc. 156, A407–A416 (2009).
Wiaderek, K. M. et al. Comprehensive insights into the structural and chemical changes in mixed-anion FeOF electrodes by using Operando PDF and NMR spectroscopy. J. Am. Chem. Soc. 135, 4070–4078 (2013).
Choi, S. & Manthiram, A. Factors influencing the layered to spinel-like phase transition in layered oxide cathodes. J. Electrochem. Soc. 149, A1157–A1163 (2002).
Kim, H. et al. Multicomponent effects on the crystal structures and electrochemical properties of spinel-structured M3O4 (M = Fe, Mn, Co) anodes in lithium rechargeable batteries. Chem. Mater. 24, 720–725 (2012).
Sina, M., Pereira, N., Amatucci, G. G. & Cosandey, F. Microstructural evolution of iron oxyfluoride/carbon nanocomposites upon electrochemical cycling. J. Phys. Chem. C 120, 13375–13383 (2016).
Oh, M. H. et al. Galvanic replacement reactions in metal oxide nanocrystals. Science 340, 964–968 (2013).
Vlasse, M., Massies, J. C. & Demazeau, G. The refinement of the crystal structure of iron oxyfluoride, FeOF. J. Solid State Chem. 8, 109–113 (1973).
Dong, A. et al. A generalized ligand-exchange strategy enabling sequential surface functionalization of colloidal nanocrystals. J. Am. Chem. Soc. 133, 998–1006 (2011).
Kelly, S., Hesterberg, D. & Ravel, B. Analysis of soils and minerals using X-ray absorption spectroscopy. Methods Soil Anal. 5, 387–463 (2008).
Ravel, B. & Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 12, 537–541 (2005).
Rehr, J. J. & Albers, R. Theoretical approaches to X-ray absorption fine structure. Rev. Mod. Phys. 72, 621–654 (2000).
Lee, S.-Y. et al. Unveiling origin of additional capacity of SnO2 anode in lithium-ion batteries by realistic ex situ TEM analysis. Nano Energy 19, 234–245 (2016).
Acknowledgements
This work was supported by Samsung Research Funding Centre of Samsung Electronics under Project Number SRFC-TA1403-03. Also, this work was supported by Project Code (IBS-R006-G1). S.-K.J., H.K., K.-Y.P. and K.K. are grateful for the financial support from IBS.
Author information
Authors and Affiliations
Contributions
S.-K.J. conceived the research and carried out the synthesis, electrochemical test, and characterization. H.K. performed the overall X-ray absorption spectroscopy experiment and analyses. M.G.C. synthesized the Mn3O4 nanoparticle and performed TEM work for the LiF–C/Mn3O4 system. S.-P.C. conducted TEM and STEM–EELS measurements for the LiF–MnO nanocomposite. B.L. and G.Y. conducted the DFT calculations and developed the theoretical model. W.-S.Y. and K.K. supervised the overall research. All authors discussed the experiments and final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–35, Supplementary Tables 1–3, Supplementary Discussion, Supplementary Methods, Supplementary References (PDF 3825 kb)
Rights and permissions
About this article
Cite this article
Jung, SK., Kim, H., Cho, M. et al. Lithium-free transition metal monoxides for positive electrodes in lithium-ion batteries. Nat Energy 2, 16208 (2017). https://doi.org/10.1038/nenergy.2016.208
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nenergy.2016.208
This article is cited by
-
Enhanced Electrochemical Performances of Ni Doped Cr8O21 Cathode Materials for Lithium-ion Batteries
Journal of Wuhan University of Technology-Mater. Sci. Ed. (2023)
-
Building Better Full Manganese-Based Cathode Materials for Next-Generation Lithium-Ion Batteries
Electrochemical Energy Reviews (2023)
-
Amorphous iron fluorosulfate as a high-capacity cathode utilizing combined intercalation and conversion reactions with unexpectedly high reversibility
Nature Energy (2022)
-
Inorganic-organic competitive coating strategy derived uniform hollow gradient-structured ferroferric oxide-carbon nanospheres for ultra-fast and long-term lithium-ion battery
Nature Communications (2021)
-
Atomic-scale structural evolution of electrode materials in Li-ion batteries: a review
Rare Metals (2020)