Abstract
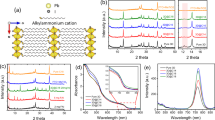
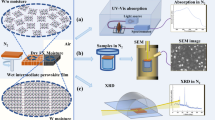
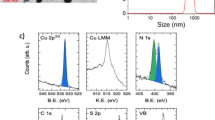
Organic–inorganic hybrid perovskites are particularly suited as light-harvesting materials in photovoltaic devices. The power conversion efficiency of perovskite solar cells has reached certified values of over 20% in just a few years. However, one of the major hindrances for application of these materials in real-world devices is the performance degradation in humid conditions, leading to a rapid loss of photovoltaic response. Here, we demonstrate that hydrophobic tertiary and quaternary alkyl ammonium cations can be successfully assembled on the perovskite surface as efficient water-resisting layers via a facile surface functionalization technique. Such layers can protect the perovskite film under high relative humidity (90 ± 5%) over 30 days. More importantly, devices based on such films can retain the photovoltaic capacities of bulk perovskites, with power conversion efficiencies over 15%. Improving the humidity tolerance of perovskite materials is a necessary step towards large-scale production of high-performance perovskite-based devices under ambient humidity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kojima, A., Teshima, K., Shirai, Y. & Miyasaka, T. Organometal halide perovskites as visible-light sensitizers for photovoltaic cells. J. Am. Chem. Soc. 131, 6050–6051 (2009).
Im, J.-H., Lee, C.-R., Lee, J.-W., Park, S.-W. & Park, N.-G. 6.5% efficient perovskite quantum-dot-sensitized solar cell. Nanoscale 3, 4088–4093 (2011).
Kim, H.-S. et al. Lead iodide perovskite sensitized all-solid-state submicron thin film mesoscopic solar cell with efficiency exceeding 9%. Sci. Rep. 2, 591 (2012).
Lee, M. M., Teuscher, J., Miyasaka, T., Murakami, T. N. & Snaith, H. J. Efficient hybrid solar cells based on meso-superstructured organometal halide perovskites. Science 338, 643–647 (2012).
Zhou, H. et al. Interface engineering of highly efficient perovskite solar cells. Science 345, 542–546 (2014).
Jeon, N. J. et al. Compositional engineering of perovskite materials for high-performance solar cells. Nature 517, 476–480 (2015).
Hagfeldt, A., Boschloo, G., Sun, L., Kloo, L. & Pettersson, H. Dye-sensitized solar cells. Chem. Rev. 110, 6595–6663 (2010).
Niu, G. et al. Study on the stability of CH3NH3PbI3 films and the effect of post-modification by aluminum oxide in all-solid-state hybrid solar cells. J. Mater. Chem. A 2, 705–710 (2014).
Snaith, H. J. Perovskites: the emergence of a new era for low-cost, high-efficiency solar cells. J. Phys. Chem. Lett. 4, 3623–3630 (2013).
Park, N.-G. Organometal perovskite light absorbers toward a 20% efficiency low-cost solid-state mesoscopic solar cell. J. Phys. Chem. Lett. 4, 2423–2429 (2013).
Vincent, B. R., Robertson, K. N., Cameron, T. S. & Knop, O. Alkylammonium lead halides. Part 1. isolated PbI6 4− ions in (CH3NH3)4PbI6⋅2H2O. Can. J. Chem. 65, 1042–1046 (1987).
Ko, H.-S., Lee, J.-W. & Park, N.-G. 15.76% efficiency perovskite solar cells prepared under high relative humidity: importance of PbI2 morphology in two-step deposition of CH3NH3PbI3 . J. Mater. Chem. A 3, 8808–8815 (2015).
Luo, P. et al. Uniform, stable, and efficient planar-heterojunction perovskite solar cells by facile low-pressure chemical vapor deposition under fully open-air conditions. ACS Appl. Mater. Int. 7, 2708–2714 (2015).
Heo, J. H., Song, D. H. & Im, S. H. Planar CH3NH3PbBr3 hybrid solar cells with 10.4% power conversion efficiency, fabricated by controlled crystallization in the spin-coating process. Adv. Mater. 26, 8179–8183 (2014).
Heo, J. H., Han, H. J., Kim, D., Ahn, T. K. & Im, S. H. Hysteresis-less inverted CH3NH3PbI3 planar perovskite hybrid solar cells with 18.1% power conversion efficiency. Energ. Environ. Sci. 8, 1602–1608 (2015).
Smith, I. C., Hoke, E. T., Solis-Ibarra, D., McGehee, M. D. & Karunadasa, H. I. A layered hybrid perovskite solar-cell absorber with enhanced moisture stability. Angew. Chem. Int. Ed. 126, 11414–11417 (2014).
Jiang, Q. et al. Pseudohalide-induced moisture tolerance in perovskite CH3NH3Pb(SCN)2I thin films. Angew. Chem. Int. Ed. 127, 7727–7730 (2015).
Zhang, J. et al. Bifunctional alkyl chain barriers for efficient perovskite solar cells. Chem. Commun. 51, 7047–7050 (2015).
Li, X. et al. Improved performance and stability of perovskite solar cells by crystal crosslinking with alkylphosphonic acid ω-ammonium chlorides. Nature Chem. 7, 703–711 (2015).
Mitzi, D. B. Solution-processed inorganic semiconductors. J. Mater. Chem. 14, 2355–2365 (2004).
Saba, M. et al. Correlated electron–hole plasma in organometal perovskites. Nature Commun. 5, 6049 (2014).
Mitzi, D. B. Templating and structural engineering in organic–inorganic perovskites. J. Chem. Soc. Dalton Trans. 2001, 1–12 (2001).
Mosconi, E., Azpiroz, J. M. & DeAngelis, F. Ab initio molecular dynamics simulations of methylammonium lead iodide perovskite degradation by water. Chem. Mater. 27, 4885–4892 (2015).
Wang, Y. et al. Density functional studies of stoichiometric surfaces of orthorhombic hybrid perovskite CH3NH3PbI3 . J. Phys. Chem. C 119, 1136–1145 (2014).
Baikie, T. et al. Synthesis and crystal chemistry of the hybrid perovskite (CH3NH3)PbI3 for solid-state sensitised solar cell applications. J. Mater. Chem. A 1, 5628–5641 (2013).
Kollek, T. et al. Porous and shape-anisotropic single crystals of the semiconductor perovskite CH3NH3PbI3 from a single-source precursor. Angew. Chem. Int. Ed. 54, 1341–1346 (2015).
Liang, K., Mitzi, D. B. & Prikas, M. T. Synthesis and characterization of organic–inorganic perovskite thin films prepared using a versatile two-step dipping technique. Chem. Mater. 10, 403–411 (1998).
Tang, J. et al. Colloidal-quantum-dot photovoltaics using atomic-ligand passivation. Nature Mater. 10, 765–771 (2011).
Chuang, C.-H. M., Brown, P. R., Bulović, V. & Bawendi, M. G. Improved performance and stability in quantum dot solar cells through band alignment engineering. Nature Mater. 13, 796–801 (2014).
Yang, S. et al. Formation mechanism of freestanding CH3NH3PbI3 functional crystals: in situ transformation vs dissolution–crystallization. Chem. Mater. 26, 6705–6710 (2014).
Fu, Y. et al. Solution growth of single crystal methylammonium lead halide perovskite nanostructures for optoelectronic and photovoltaic applications. J. Am. Chem. Soc. 137, 5810–5818 (2015).
Kim, H.-S. & Park, N.-G. Parameters affecting I–V hysteresis of CH3NH3PbI3 perovskite solar cells: effects of perovskite crystal size and mesoporous TiO2 layer. J. Phys. Chem. Lett. 5, 2927–2934 (2014).
Snaith, H. J. et al. Anomalous hysteresis in perovskite solar cells. J. Phys. Chem. Lett. 5, 1511–1515 (2014).
Shao, Y., Xiao, Z., Bi, C., Yuan, Y. & Huang, J. Origin and elimination of photocurrent hysteresis by fullerene passivation in CH3NH3PbI3 planar heterojunction solar cells. Nature Commun. 5, 5784 (2014).
Matsumoto, F., Vorpahl, S. M., Banks, J. Q., Sengupta, E. & Ginger, D. S. Photodecomposition and morphology evolution of organometal halide perovskite solar cells. J. Phys. Chem. C 119, 20810–20816 (2015).
Niu, G., Guo, X. & Wang, L. Review of recent progress in chemical stability of perovskite solar cells. J. Mater. Chem. A 3, 8970–8980 (2015).
Haller, I. Covalently attached organic monolayers on semiconductor surfaces. J. Am. Chem. Soc. 100, 8050–8055 (1978).
Feng, X. J. & Jiang, L. Design and creation of superwetting/antiwetting surfaces. Adv. Mater. 18, 3063–3078 (2006).
Jiang, L., Zhao, Y. & Zhai, J. A lotus-leaf-like superhydrophobic surface: a porous microsphere/nanofiber composite film prepared by electrohydrodynamics. Angew. Chem. Int. Ed. 116, 4438–4441 (2004).
Christians, J. A., Miranda Herrera, P. A. & Kamat, P. V. Transformation of the excited state and photovoltaic efficiency of CH3NH3PbI3 perovskite upon controlled exposure to humidified air. J. Am. Chem. Soc. 137, 1530–1538 (2015).
Leguy, A. M. A. et al. Reversible hydration of CH3NH3PbI3 in films, single crystals, and solar cells. Chem. Mater. 27, 3397–3407 (2015).
Forney, C. F. & Brandl, D. G. Control of humidity in small controlled-environment chambers using glycerol–water solutions. HortTechnology 2, 52–54 (1992).
Kresse, G. & Hafner, J. Ab initio molecular-dynamics for liquid-metals. Phys. Rev. B 47, 558–561 (1993).
Kresse, G. & Furthmuller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Vanderbilt, D. Soft self-consistent pseudopotentials in a generalized eigenvalue formalism. Phys. Rev. B 41, 7892–7895 (1990).
Kresse, G. & Hafner, J. Norm-conserving and ultrasoft pseudopotentials for first-row and transition-elements. J. Phys. Condens. Matter 6, 8245–8257 (1994).
Wang, Y. et al. Density functional theory analysis of structural and electronic properties of orthorhombic perovskite CH3NH3PbI3 . Phys. Chem. Chem. Phys. 16, 1424–1429 (2014).
Dion, M., Rydberg, H., Schroder, E., Langreth, D. C. & Lundqvist, B. I. Van der Waals density functional for general geometries. Phys. Rev. Lett. 92, 246401 (2004).
Klimes, J., Bowler, D. R. & Michaelides, A. Van der Waals density functionals applied to solids. Phys. Rev. B 83, 195131 (2011).
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (21373083) and Fundamental Research Funds for the Central Universities (WD1313009). This research was undertaken in the NCI National Facility in Canberra, Australia, which is supported by the Australian Commonwealth Government.
Author information
Authors and Affiliations
Contributions
H.G.Y. and H.J.Z. conceived the project and contributed to the design of the experiments and analysis of the data. S.Y. performed the material preparation, device fabrication and characterizations. Y.W. and P.L. conducted DFT calculations and wrote part of the paper (calculations). Y.-B.C. performed data analyses of photovoltaic properties. S.Y. prepared the manuscript with the help of H.G.Y. and H.J.Z. All the authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-16, Supplementary Tables 1-3, Supplementary References. (PDF 1678 kb)
Rights and permissions
About this article
Cite this article
Yang, S., Wang, Y., Liu, P. et al. Functionalization of perovskite thin films with moisture-tolerant molecules. Nat Energy 1, 15016 (2016). https://doi.org/10.1038/nenergy.2015.16
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nenergy.2015.16
This article is cited by
-
Bioinspired “cage traps” for closed-loop lead management of perovskite solar cells under real-world contamination assessment
Nature Communications (2023)
-
Tailoring passivators for highly efficient and stable perovskite solar cells
Nature Reviews Chemistry (2023)
-
Ionic Liquid Assisted Imprint for Efficient and Stable Quasi-2D Perovskite Solar Cells with Controlled Phase Distribution
Nano-Micro Letters (2023)
-
Tough, stable and self-healing luminescent perovskite-polymer matrix applicable to all harsh aquatic environments
Nature Communications (2022)
-
Impeded degradation of perovskite solar cells via the dual interfacial modification of siloxane
Science China Chemistry (2022)