Abstract
Increasing atmospheric CO2 concentrations are causing ocean acidification (OA), altering carbonate chemistry with consequences for marine organisms. Here we show that OA increases by 46–212% the production of phenolic compounds in phytoplankton grown under the elevated CO2 concentrations projected for the end of this century, compared with the ambient CO2 level. At the same time, mitochondrial respiration rate is enhanced under elevated CO2 concentrations by 130–160% in a single species or mixed phytoplankton assemblage. When fed with phytoplankton cells grown under OA, zooplankton assemblages have significantly higher phenolic compound content, by about 28–48%. The functional consequences of the increased accumulation of toxic phenolic compounds in primary and secondary producers have the potential to have profound consequences for marine ecosystem and seafood quality, with the possibility that fishery industries could be influenced as a result of progressive ocean changes.
Similar content being viewed by others
Introduction
Increasing atmospheric CO2 levels are continuing to cause global warming, with increasing oceanic CO2 uptake playing an important role in the mediation of the extent of this increase. However, the rapid CO2 dissolution into seawater is also causing ocean acidification (OA), which progressively alters marine chemical environments, with consequences for many organisms. While physiological and ecological effects of ocean climate change on primary producers have been examined broadly1,2, little is known about the molecular aspects and/or metabolic pathways underlying the responses of phytoplankton to OA3. Moreover, there are growing concerns about the need to couple the data obtained from monospecific laboratory studies with that from natural communities, as well as under multiple stressor conditions4. In addition, the consequences of OA for energy transfer, food quality and the food web remain poorly understood.
To address this issue, we here employ a proteomics approach to investigate the responses of a coccolithophorid, Emiliania huxleyi (CCMP 1516) to elevated CO2 at the molecular level. On the basis of the findings of the proteomics study, we hypothesize that OA could enhance some metabolic pathways, leading to enhanced production of phenolic compounds. To test this, we measured the levels of phenolic compounds and mitochondrial respiration rates in phytoplankton in monospecific laboratory cultures and in mixed phytoplankton assemblages, grown under different levels of CO2. Subsequently, zooplankton assemblages were fed with phytoplankton cells grown under the elevated CO2 concentration to examine possible food chain effects. Our results show that OA increases the levels of phenolic compounds in phytoplankton by enhancing β-oxidation, Krebs cycle and mitochondrial respiration, and the accumulated phenolic compounds are transferred to higher trophic levels (zooplankton).
Results
Proteomic responses of phytoplankton to OA
When the coccolithophore, E. huxleyi (CCMP 1516), was grown monospecifically under low (LC, 395 μatm) pCO2 or, high (HC, 1,000 μatm) pCO2, contrasting differences were found in the proteomics (Supplementary Fig. 1). The enzymes methane/phenol/toluene hydroxylase, which plays key roles in phenolic compound biodegradation, propionyl CoA synthase and enoyl CoA hydratase that function in β-oxidation, adenylate kinase (ADK), which is a key enzyme in energy metabolism, and chloroplastic GADPH, were all upregulated in the HC-grown cells (Supplementary Table 1). However, another key enzyme in energy metabolism, nucleoside diphosphate kinase, transferring phosphoryl groups between the adenine and guanine pools (GDP+ATP to GTP+ADP)5, was downregulated by about 50% in the HC-grown cells compared with the LC-grown cells (Supplementary Table 1), suggesting that relative ATP loss in cells under HC was slowed down. On the basis of these differentially expressed proteins and their functions, we hypothesized that elevated CO2 enhanced the metabolic pathway, described below, leading to enhanced production of phenolic compounds and their metabolism (Fig. 1).
Metabolic pathways in the coccolithophorid Emiliania huxleyi altered under ocean acidification (HC, CO2 1,000 μatm; pHNBS 7.81) based on proteomic, physiological and biochemical analyses. More phenolic compounds were biosynthesized, subsequently biodegraded and then metabolized via β-oxidation and Krebs cycle, generating more ATP in the HC-grown cells by mitochondrial respiration. This extra energy can be used to counter the effects of high CO2/low pH stress from the environment. Adenylate kinase (ADK) was upregulated, and glycolysis was accelerated in the HC-grown cells. On the other hand, nucleoside diphosphate kinase (NDPK) was downregulated, reflecting the slower loss of ATP loss in the HC-grown cells. The novel pathway including β-oxidation and the Krebs cycle becomes enhanced under ocean acidification to meet the extra energy requirement for maintaining homoeostasis. The red and green symbols represent up- and downregulated proteins or processes, respectively.
The high CO2-induced changes in seawater carbonate chemistry might enhance the cellular production of phenolic compounds, but may also accelerate their biodegradation. As a consequence, enzymes such as phenol hydroxylase would be required for their biodegradation. The resulting products, cis, cis, muconic acid or 2-hydroxymuconic semialdehyde, are further metabolized via β-oxidation and, subsequently, expression of propionyl CoA synthase and enoyl CoA hydratase would be enhanced. The resulting product, acetyl CoA, is a key precursor compound for the Krebs cycle, which functions in oxidizing acetyl CoA to CO2 and drives the synthesis of ATP. In such circumstances, the generation of ATP could thus be stimulated in the HC-grown cells (Fig. 1, Supplementary Table 1). Moreover, the enzyme chloroplast GADPH associated with glycolysis was upregulated in the HC-grown cells, indicating that energy demand under the elevated CO2 level was higher to operate cellular essential metabolism (Fig. 1, Supplementary Table 1). Together with this, ADK, a key enzyme in energy metabolism, catalysing a reversible transphosphorylation reaction interconverting ADP to ATP and AMP showed significantly higher expression in the HC-grown cells (Fig. 1, Supplementary Table 1). Hence, at least two lines of evidence have to be provided to support the above hypothesis: namely, a higher phenolic compound content and increased mitochondrial respiration rates in E. huxleyi grown under high CO2 conditions.
Biochemical and physiological tests
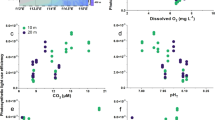
To test the above hypothesis, we measured the content of phenolic compounds and mitochondrial respiration rates in E. huxleyi cells grown under HC and LC conditions. The phenolic compounds were about 56% higher in the HC-acclimated cells than in the LC-acclimated cells (20 generations; analysis of variance (ANOVA) 1, F1,3=119.53, P<0.001) (Fig. 2a, statistical details in Supplementary Table 2). When the LC-acclimated cells were transferred to HC conditions, the phenolic compound content significantly increased by 24% after a short period of growth (∼10 generations; ANOVA 1, F1,3=74.33, P=0.01; Fig. 2a, statistical details in Supplementary Table 2). Mitochondrial respiration rates were about 130% higher in the HC-grown cells than in the LC-grown cells (ANOVA 1, F1,3=532.66, P<0.001; Fig. 2b, statistical details in Supplementary Table 2).
Contents of phenolic compounds and mitochondrial respiration rates under high CO2 (HC, 1,000 μatm, pHNBS 7.81) (black column), low pCO2 (LC, 395 μatm, pHNBS 8.16; white column) and LC–HC conditions (inset, grey column) in different test systems. (a,b) Laboratory cultures: contents of phenolic compounds of Emiliania huxleyi, μg (μg chlorophyll a; chl a)−1 (a); Mitochondrial respiration rates of E. huxleyi, fmol O2 per cell per h (b); (c,d) Microcosm and mesocosm tests: Phenolic contents of natural phytoplankton assemblages grown in 30 L microcosms, μg (μg chl a)−1 (c); Phenolic contents of phytoplankton assemblages grown in 4,000 L mesocosms, μg (μg chl a)−1 (d); (e): Respiratory carbon loss rates of natural phytoplankton assemblages grown in the mesocosms, μg C (μg chl a)−1 d−1). LC–HC (a, inset) represents LC-acclimated cells (20 generations) that were transferred to HC conditions for 7 days (∼10 generations). Three independent replicate systems (laboratory cultures, microcosms or mesocosms) were run for each treatment (details for statistics are shown in Supplementary Table 2). Vertical lines represent s.d. of the means. * indicate significance at the P<0.05 level.
In an attempt to further test our hypothesis at a community level, we conducted a microcosm-level experiment (30 L) with natural coastal phytoplankton assemblages (dominated by the diatoms Skeletonema costatum and Chaetoceros sp.; details in Supplementary Note 2). We found that phytoplankton assemblages under the high (HC, 1,000 μatm) pCO2 treatment showed significantly higher contents of phenolic compounds (by 45.7%) when compared with that of the low pCO2 assemblages (ANOVA 2, model: F5,6=4.28, P=0.048; CO2: F1,6=18.84, P=0.005) (Fig. 2c, statistical details in Supplementary Table 2). During a mesocosm experiment (4,000 L; details in Supplementary Note 2) using The Facility for Ocean Acidification Impacts Study of Xiamen University (24.52°N, 117.18°N, Wuyuan Bay, Xiamen, China, http://mel.xmu.edu.cn/facility.asp?id=33), the phytoplankton assemblages (dominated mainly by diatoms and coccolithophores) also showed elevated, by about 212%, contents of phenolic compounds under high (HC, 1,000 μatm) pCO2 (ANOVA 2, model: F5,12=10.91, P<0.001; CO2: F1,12=54.48, P<0.001; Fig. 2d, statistical details in Supplementary Table 2), compared with that of the low pCO2 assemblages. Although the mean values of mitochondrial respiration were higher by 160% in the high CO2 mesocosms than in the low CO2 treatments, the difference was not significant due to a large variation in the data (ANOVA 1, F1,3=2.78, P=0.171; Fig. 2e, statistical details in Supplementary Table 2).
Transfer of phenolics to higher trophic level
Since biochemical compositions altered by OA in phytoplankton may have profound impacts on trophic energy transfer6,7, we therefore tested whether zooplankton fed with phytoplankton containing higher levels of phenolic compounds would also have higher concentrations of these toxic compounds in their bodies. When the zooplankton (dominated by calanoid copepods (95%), of which Acartia pacifica accounted for up to ∼60%, Supplementary Fig. 2) was fed with HC- and LC-grown phytoplankton cells under the high (1,000 μatm) and low (395 μatm) pCO2 levels, higher contents of phenolic compounds were detected in their bodies at elevated pCO2, which was true either in the microcosms (47.5%, ANOVA 2, model: F5,6=7.19, P=0.016; CO2: F1,6=33.24, P=0.001; Fig. 3a) or when fed with phytoplankton cells from the mesocosms (27.8%, ANOVA 2, model: F5,6=8.15, P=0.012; CO2: F1,6=29.46, P=0.002; Fig. 3b, statistic details in Supplementary Table 2).
Content of phenolic compounds (μg per individual) in zooplankton assemblages (body size >112 μm) that were fed on high CO2 (HC, 1,000 μatm, pHNBS 7.81) or low pCO2 grown (LC, 395 μatm, pHNBS 8.16) phytoplankton cells collected from the microcosms (a, triplicate) or mesocosms (b, triplicate). The feeding tests were carried out in in situ seawater pre-equilibrated with the high (1,000 μatm) or low pCO2 (395 μatm) levels, that is, HC-grown zooplankton fed with HC-grown phytoplankton (black column) or LC-grown zooplankton fed with LC-grown phytoplankton (white column). Details of statistics are shown in Supplementary Table 2. Vertical lines represent the s.d. of the means. * indicates significance at the P<0.05 level.
Discussion
On the basis of the outcomes of proteomics in the monospecific study with E. huxleyi and the physiological results from the monospecific study, microcosm and mesocosm tests with mixed phytoplankton species or natural phytoplankton assemblages, the present work suggests that a novel phenolic compound metabolism pathway, involving β-oxidation and the Krebs cycle, was enhanced by OA. While there are controversial findings on the effects of OA on mitochondrial respiration8,9,10, rising pCO2 and decreasing pH in seawater perturb the cytoplasmic acid–base balance of phytoplankton11, so that extra energy would be required to maintain the cell’s homoeostasis or positive H+ efflux12. Enhanced photorespiratory carbon loss in high CO2 grown cells9,13 consumes additional energy for photoprotection. Consequently, an extra energy requirement for maintaining homoeostasis when phytoplankton cells are perturbed by changed seawater chemistry can be expected. In this study, we demonstrated that β-oxidation and the Krebs cycle were enhanced under OA and could thus meet any extra energetic demand to allow phytoplankton to tolerate acidic stress. For coccolithophores, increased phenolic compounds may not only reflect a way to endure with any extra energetic demand under OA but may also act as repellents to protect them from grazers as the cells calcify less under OA1.
It is a well-known phenomenon in higher plants that they increase the production of phenolic compounds to deter grazers. Our finding that phytoplankton increased the production of these compounds under high CO2 thus has implications for grazers, though the mechanism by which phytoplankton species or assemblages upregulate phenolic biosynthesis in response to increased pCO2 is not immediately clear. In contrast to our results, a recent study showed decreased content of these compounds in seagrasses exposed to high CO2/low pH condition near a natural CO2 vent14. OA has been shown to significantly alter fatty acid content and composition in diatoms6. The increased cellular phenolic compounds (shown in this work, Fig. 2a,c), also linked to fatty acid metabolism via β-oxidation, would further decrease the nutritional value of these organisms. Since phenolic compounds are highly toxic and are found in marine systems15,16, an increase in their content in primary producers would undoubtedly lead to significant consequences for food webs and carbon cycles. In addition, as phenolics are known to possess antimicrobial properties17, biogeochemical cycles in the oceans may be affected as well.
The present work demonstrated that accumulation of phenolic compounds increased in phytoplankton under OA and that they were transferred to higher trophic levels (zooplankton). Consequently, accumulation of phenolic compounds in seafood could be a factor that affects the quality of seafood18 when the organisms are exposed to OA, which was recently shown to affect the taste of shrimps19. Different taxa are known to show differential sensitivities to OA20, which can be altered or amplified under other forcing from ocean changes, such as warming21 and ultraviolet-B irradiance22. At the same time, phytoplankton species can exhibit evolutionary responses to OA23, and the changes in the profile of phenolic compounds remain unknown for long-term adaptation to OA. While altered biochemistry of the diatom Thalassiosira pseudonana grown under OA conditions can decrease egg production of a copepod6, exposure to phenol can also decrease egg, faecal pellet production and survival in copepods24,25 and toxic compounds can be transferred to higher trophic levels (this work). Therefore, an increase in the content of phenolic compounds in plankton could have far-reaching impacts on seafood qualitative and quantitative values and on species interactions as well as community structures, with consequences for ecosystem functioning and fishery industries.
Methods
Species and laboratory cultures
E. huxleyi (CCMP 1516), acquired from the Provasoli-Guillard National Center for Culture of Marine Phytoplankton (CCMP), was grown semi-continuously in high (HC, 1,000 μatm, pHNBS 7.81) or low (LC, 395 μatm, pHNBS 8.16) pCO2 (with the HC representing CO2 levels projected for the end of this century26) pre-equilibrated artificial seawater enriched with Aquil culture medium. The HC and LC cultures (triplicate independent cultures for each treatment) were maintained in exponential growth phase by continual dilution (every 24 h) for 20 generations before being used in the experiments, and the seawater carbonate system parameters were maintained at stable levels (daily variation in pHNBS <0.06, seawater carbonate system parameters see Supplementary Note 1 and Supplementary Table 3) by using freshly prepared medium equilibrated with the target CO2 levels and by sustaining cell concentration within a range of 2.0–4.5 × 104 cells per ml. The target CO2 levels of HC and LC medium were achieved by using a CO2 Enrichlor (CE-100B, Wuhan Ruihua Instrument & Equipment Ltd, China) and bubbling with ambient air, respectively. The cells were grown under a photon flux density of 100 μmol photons per m2 s−1 (12:12 light: dark cycle) in a plant growth chamber (GXZ, Ruihua, Wuhan, China) at 20 °C.
Proteomics analysis
After acclimation for 20 generations, HC- and LC-grown E. huxleyi cells were collected for protein extractions, and then analysed for proteomics by applying a two-dimensional electrophoresis (2-DE) gel and MALDI-TOF-TOF mass spectrometry (MS) approach27 to identify the differentially expressed proteins between HC and LC treatments.
Specifically, after the acclimation of E. huxleyi cultures at the relevant treatments for 20 generations, 3 L samples were collected onto PC filters (Millipore, pore size 0.4 μm), re-suspended in pre-prepared medium equilibrated with the target CO2 level (pHNBS 7.81 and 8.16 for the HC- and LC-grown cells, respectively) and then re-collected by centrifugation at 10,000 × g for 30 min at 4 °C for protein extraction. The cell pellets were rinsed twice with precooled sterilized seawater to avoid any carry-over of culture medium and external proteins. Trizol reagent (1 ml) was added to the cell pellet followed by sonication (a total of 2 min with short pulses of 3–5 s) on ice. Cell lysis was confirmed using light microscopy. Subsequently, 200 μl of chloroform was added to the cell lysate before shaking vigorously for 15 s. The mixture was allowed to stand for 5 min at room temperature before being centrifuged at 12,000 × g for 15 min at 4 °C. The top pale yellow or colourless layer was removed, and then 300 μl of ethanol was added to re-suspend the reddish bottom layer, and the mixture centrifuged at 2,000 × g for 5 min at 4 °C. The supernatant was then transferred to a new tube, and 2 ml of isopropanol was added. The mixture was allowed to stand for at least 1 h for precipitation of proteins at −20 °C. It was then centrifuged at 14,000 × g for 30 min at 4 °C. Subsequently, the pellet was washed with 95% ethanol before being air-dried. To solubilize the protein pellet, 30 μl of rehydration buffer (7 M urea, 2 M thiourea, 4% w/v 3-[(3-cholamidopropyl) dimethyl-ammonio]-1-propanesulfonate (CHAPS), 1% dithiothreitol (DTT) and 0.5% v/v immobilized pH gradient (IPG)) were added. The resulting solution was centrifuged at 20,000 × g for 30 min at 4 °C and the supernatant was collected for 2-DE analysis. The protein content was quantified using a 2-D Quant kit (GE Healthcare, San Francisco, USA).
Exactly 100 μg of protein sample (duplicates for each CO2 treatment) was mixed with a rehydration buffer (7 M urea, 2 M thiourea, 4% w/v 3-[(3-cholamidopropyl) dimethyl-ammonio]-1-propanesulfonate, 1% DTT, and 0.5% v/v IPG) before being loaded onto IPG strips with a linear pH gradient of 4–7 (Immobiline Drystrip, pH 4–7, GE Healthcare Life Science, Piscataway, USA). The sample was subjected to isoelectric focusing using an IPGphor III system with 24 cm IPG strips in the following manner: 6 h at 40 V (active rehydration), 6 h at 100 V; 0.5 h at 500 V; 1 h at 1,000 V; 1 h at 2,000 V; 1.5 h at 10,000 V; and 60,000 Vh at 10,000 V. The minimal Vh applied was at least 60,000 units. Subsequently, the immobilized pH gradient strips were equilibrated for 15 min in 10 ml of equilibration buffer containing 6 M urea, 2% SDS, 50 mM Tris-Cl (pH 8.8), 30% glycerol and 1% DTT, followed by equilibration for 15 min in alkylation buffer containing 6 M urea, 2% SDS, 50 mM Tris-Cl (pH 8.8), 30% glycerol and 2.5% iodoacetamide. Two-dimensional SDS–PAGE (2-DE) gels (12.5%) were run in an EttanDalt system (GE Healthcare) at 1 w per gel for 30 min and then at 15 w per gel for 6 h. The 2-DE gels were visualized using Coomassie Blue staining and digitized using a gel documentation system on a GS-670 Imaging Densitometer from Bio-Rad (USA) with 2-DE electrophoretogram-matching software. Image analysis was performed using DeCyder version 7.0 software (GE Healthcare) following the manufacturer’s instructions.
MS analyses were conducted using an AB SCIEX MALDI TOF-TOF 5800 Analyser (AB SCIEX, Shanghai, China) equipped with a neodymium: yttrium-aluminum-garnet laser (laser wavelength was 349 nm), in reflection positive-ion mode. Protein identification was conducted according to the previously described method27. Briefly, the MS and MS/MS spectra of each protein spot obtained from MALDI-TOF-TOF MS were searched against the NCBI non-redundant protein database using the BLASTX algorithm. If the total ion score confidence interval was above 95% and the E value was below e−20 at the amino acid sequence level, the sequence similarities were considered to be significant. The details of NCBI ID number, theoretical pI value, theoretical molecular weight, protein score, protein score confidence interval %, as well as the average relative change are listed in Supplementary Table 1.
Microcosm test
Microcosms of 30 L (water-jacketed for temperature control) were run from December 2014 to January 2015 at Wuyuan Bay (Xiamen, China) on the mesocosm facility platform (24.52°N, 117.18°N. Surface seawater (0–1 m) was collected at midday, filtered (200 μm) to remove large grazers and dispensed into the microcosms (triplicate microcosms were run for each CO2 treatment). The microcosms were made of polymethyl methacrylate, which allowed 91% photosynthetically active radiation, 63% ultraviolet-A (315–400 nm) and 6% ultraviolet-B (280–315 nm) transmissions under the incident solar radiation. The temperature within the microcosms was controlled to the sea surface temperature (13.0–15.0 °C) by circulating in situ seawater through the jacket. The seawater carbonate system in the microcosms was maintained stable by aerating with air of high (HC, 1,000 μatm) or low (LC, 395 μatm) pCO2 (see CO2 manipulation method in ref. 9). Samples of 100 ml from each microcosm were collected for the determination of phenolic compounds on day 5, while the microcosms were exposed to solar irradiance and run until the end of exponential growth phase of the phytoplankton growth (9 days).
Mesocosm test
The Facility for Ocean Acidification Impacts Study of XMU consists of eight independently operated mesocosm units, located at 24.52°N, 117.18°N, Wuyuan Bay (Xiamen, China), of which three HC (HC, 1,000 μatm CO2) and three LC (LC, 395 μatm CO2) units were randomly chosen (for details see in Supplementary Note 2). Four species of phytoplankton, Phaeodactylum tricornutum (CCMA 106), T. weissflogii (CCMP 1335), E. huxleyi (CS-369) and Gephyrocapsa oceanica (NIES-1318) (details about the species are given in Supplementary Note 2) were then inoculated into each mesocosm at equivalent chlorophyll a concentrations to give a total final concentration of 50 cells per L on 15 June 2013 (day 0). The pCO2 in the mesocosms was controlled by bubbling air of high (HC, 1,000 μatm) or low (LC, 395 μatm) pCO2 (details for pCO2 manipulation are provided in the Supplementary Note 2).
Estimation of phenolic compounds
Phenolic content was determined according to ref. 28. Briefly, the cell pellets of phytoplankton or ∼300 zooplankton individuals were placed in 2.5 ml of 95% ethanol for a period of 48 h at 47 °C. The cells were sonicated and the supernatant was separated by centrifugation (4,500 × g) for 10 min, 1.0 ml of which was transferred to glass test tubes along with 1.0 ml 95% ethanol, 5.0 ml distilled water and 0.5 ml of 50% Folin–Ciocalteu reagent (Sigma Chemical, USA). The solution was allowed to react for 5 min, then 1.0 ml of 5% Na2CO3 was added, and the mixture was vortexed and placed in the darkness for 1 h. Absorbance was determined with a scanning spectrophotometer (DU800, Beckman, Fullerton, CA, USA) at 725 nm and plotted against a standard curve obtained from gallic acid.
Measurement of respiration
Mitochondrial respiration rates of laboratory cultures were determined by a Clark-type oxygen electrode in darkness. The respiratory carbon loss in phytoplankton assemblages from the mesocosms over 12 h was calculated as the difference in the amount of fixed carbon, using 14C tracer methods, between the two time spans (carbon fixation 12 h–carbon fixation 24 h; for details, see the Supplementary Note 3).
Feeding experiments
Zooplankton individuals were obtained at night through horizontal hauling with a medium plankton net (mesh diameter, 112 μm) from surface water in Wuyuan Bay. Collected zooplankton samples were nursed in culture dishes (400 ml) in in situ seawater pre-equilibrated with target high (1,000 μatm) or low pCO2 (395 μatm) levels for 12 h before using them. Zooplankton in each culture under HC or LC treatment (three independent replicates for each CO2 treatment) were fed with either HC- or LC-grown phytoplankton cells collected from the microcosms or mesocosms at about 15 μg chlorophyll a per L every 12 h. The cultures were maintained under solar radiation by covering with five neutral density screens (providing 6% of incident solar radiation). After the feeding procedure (24 h), zooplankton samples were collected for measurements of phenolic content and sub-samples were collected for microscopic enumeration.
Additional information
How to cite this article: Jin, P. et al. Ocean acidification increases the accumulation of toxic phenolic compounds across trophic levels. Nat. Commun. 6:8714 doi: 10.1038/ncomms9714 (2015).
References
Riebesell, U. & Tortell, P. D. in Ocean Acidification eds Gattuso J. P., Hansson L. 99–121Oxford Univ. Press (2011).
Beardall, J., Stojkovic, S. & Gao, K. Interactive effects of nutrient supply and other environmental factors on the sensitivity of marine primary producers to ultraviolet radiation: implications for the impacts of global change. Aquat. Biol. 22, 5–23 (2014).
Benner, I. et al. Emiliania huxleyi increases calcification but not expression of calcification-related genes in long-term exposure to elevated temperature and pCO2 . Philos. Trans. R Soc. Lond. B Biol. Sci. 368, 20130049 (2013).
Riebesell, U. & Gattuso, J.-P. Lessons learned from ocean acidification research. Nat Clim. Chang. 5, 12–14 (2015).
Anderca, M. I., Furuichi, T., Pinontoan, R. & Muto, S. Identification of a mitochondrial nucleoside diphosphate kinase from the green alga Dunaliella tertiolecta. Plant Cell Physiol. 43, 1276 (2002).
Rossoll, D. et al. Ocean acidification-induced food quality deterioration constrains trophic transfer. PLoS ONE 7, e34737 (2012).
Schoo, K. L., Malzahn, A. M., Krause, E. & Boersma, M. Increased carbon dioxide availability alters phytoplankton stoichiometry and affects carbon cycling and growth of a marine planktonic herbivore. Mar. Biol. 160, 2145–2155 (2013).
Wu, Y., Gao, K. & Riebesell, U. CO2-induced seawater acidification affects physiological performance of the marine diatom Phaeodactylum tricornutum. Biogeosciences 7, 2915–2923 (2010).
Gao, K. et al. Rising CO2 and increased light exposure synergistically reduce marine primary productivity. Nat Clim. Chang. 2, 519–523 (2012).
Hennon, G. M., Quay, P., Morales, R. L., Swanson, L. M. & Virginia Armbrust, E. Acclimation conditions modify physiological response of the diatom Thalassiosira pseudonana to elevated CO2 concentrations in a nitrate-limited chemostat. J. Phycol. 50, 243–253 (2014).
Taylor, A. R., Chrachri, A., Wheeler, G., Goddard, H. & Brownlee, C. A voltage-gated H+ channel underlying pH homeostasis in calcifying coccolithophores. PLoS Biol. 9, e1001085 (2011).
Raven, J. A., Beardall, J. & Giordano, M. Energy costs of carbon dioxide concentrating mechanisms in aquatic organisms. Photosynth. Res. 121, 111–124 (2014).
Xu, J. & Gao, K. Future CO2-induced ocean acidification mediates the physiological performance of a green tide alga. Plant Physiol. 160, 1762–1769 (2012).
Arnold, T. et al. Ocean acidification and the loss of phenolic substances in marine plants. PLoS ONE 7, e35107 (2012).
Boyd, T. J. & Carlucci, A. Degradation rates of substituted phenols by natural populations of marine bacteria. Aquat. Toxicol. 25, 71–82 (1993).
Ianora, A. et al. New trends in marine chemical ecology. Estuar. Coast. 29, 531–551 (2006).
Arnold, T. M. & Targett, N. M. Marine tannins: the importance of a mechanistic framework for predicting ecological roles. J. Chem. Ecol. 28, 1919–1934 (2002).
Maga, J. A. Simple phenol and phenolic compounds in food flavor. Crit. Rev. Food Sci. Nutr. 10, 323–372 (1978).
Dupont, S., Hall, E., Calosi, P. & Lundve, B. First evidence of altered sensory quality in a shellfish exposed to decreased pH relevant to Ocean Acidification. J. Shellfish Res. 33, 857–861 (2014).
Kroeker, K. J. et al. Impacts of ocean acidification on marine organisms: quantifying sensitivities and interaction with warming. Glob. Chang. Biol. 19, 1884–1896 (2013).
Pörtner, H. O. & Farrell, A. P. Physiology and climate change. Science 322, 690–692 (2008).
Gao, K., Helbling, E. W., Häder, D-P. & Hutchins, D. A. Responses of marine primary producers to interactions between ocean acidification, solar radiation and warming. Mar. Ecol. Prog. Ser. 470, 167–189 (2012).
Sunday, J. M. et al. Evolution in an acidifying ocean. Trends Ecol. Evol. 29, 117–125 (2014).
Buttino, I. The effect of low concentrations of phenol and ammonia on egg production rates, fecal pellet production and egg viability of the calanoid copepod Acartia clausi. Mar. Biol. 119, 629–634 (1994).
Bechmann, R. Effect of the endocrine disrupter nonylphenol on the marine copepod Tisbe battagliai. Sci. Total Environ. 233, 33–46 (1999).
IPCC. Climate Change 2013. The Physical Science Basis eds Stocker T. F.et al. Cambridge Univ. Press (2013).
Wang, D., Gao, Y., Lin, L. & Hong, H.-S. Comparative proteomic analysis reveals proteins putatively involved in toxin biosynthesis in the marine dinoflagellate Alexandrium catenella. Mar. Drugs 11, 213–232 (2013).
Shetty, K., Curtis, O. F., Levin, R. E., Witkowsky, R. & Ang, W. Prevention of vitrification associated with in vitro shoot culture of oregano (Origanum vulgare) by Pseudomonas spp. J. Plant Physiol. 147, 447–451 (1995).
Acknowledgements
This study was supported by National Natural Science Foundation (41430967; 41120164007), State Oceanic Administration (GASI-03-01-02-04), Joint project of National Natural Science Foundation of China and Shandong province (No. U1406403), Strategic Priority Research Program of Chinese Academy of Sciences (No. XDA11020302). Visits of S.D. and J.B. to Xiamen University were supported by ‘111’ project from Ministry of Education and the State Key Laboratory of Marine Environmental Science (MEL). We thank Yaping Wu for his leading roles in microcosm and mesocosm experiments, and X. Lin, X. Liu, L. Guo, Y. Li, W. Li, Y. Li, T. Xing, X. Cai, J. Ding, F. Li, S. Tong, X. Yi, D. Yan, H. Miao, Z. Li, R. Huang, W. Zhao and X. Zeng for their kind assistance in operations of the microcosms and mesocosms. We also thank D. Wang, H. Zhang and Z. Xie for their kind assistance in analysis of the proteomics.
Author information
Authors and Affiliations
Contributions
On the basis of an original idea from P.J. and K.G., the concept of this paper was developed in discussion between all authors. P.J., T.W. and N.L. performed the experiments under the supervision of K.G., S.D., J.B., P.W.B. and U.R. contributed to the data analysis and discussion and writing of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-2, Supplementary Tables 1-3, Supplementary Notes 1-4 and Supplementary References (PDF 661 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Jin, P., Wang, T., Liu, N. et al. Ocean acidification increases the accumulation of toxic phenolic compounds across trophic levels. Nat Commun 6, 8714 (2015). https://doi.org/10.1038/ncomms9714
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms9714
This article is cited by
-
Nano-ecotoxicology in a changing ocean
SN Applied Sciences (2022)
-
Approaches and involved principles to control pH/pCO2 stability in algal cultures
Journal of Applied Phycology (2021)
-
Production and bioaccessibility of Emiliania huxleyi biomass and bioactivity of its aqueous and ethanolic extracts
Journal of Applied Phycology (2021)
-
Different physiological and molecular responses of the green algae Chlorella variabilis to long-term and short-term elevated CO2
Journal of Applied Phycology (2020)
-
Impacts of ocean acidification under multiple stressors on typical organisms and ecological processes
Marine Life Science & Technology (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.