Abstract
High resolution proteomics increasingly reveals that most native ion channels are assembled in macromolecular complexes. However, whether different partners have additive or cooperative functional effects, or whether some combinations of proteins may preclude assembly of others are largely unexplored topics. The large conductance Ca2+-and-voltage activated potassium channel (BK) is well-suited to discern nuanced differences in regulation arising from combinations of subunits. Here we examine whether assembly of two different classes of regulatory proteins, β and γ, in BK channels is exclusive or independent. Our results show that both γ1 and up to four β2-subunits can coexist in the same functional BK complex, with the gating shift caused by β2-subunits largely additive with that produced by the γ1-subunit(s). The multiplicity of β:γ combinations that can participate in a BK complex therefore allow a range of BK channels with distinct functional properties tuned by the specific stoichiometry of the contributing subunits.
Similar content being viewed by others
Introduction
Proteomic1,2 and functional2,3,4,5,6 studies have revealed many partners that interact with BK channels, some of which are known to confer distinct tissue-specific functional properties on the BK pore-forming subunit. BK channels are dually activated by membrane depolarization and increases in intracellular [Ca2+] (refs 7, 8, 9). The minimal functional unit of a BK channel is a homotetramer of pore-forming α-subunits (Fig. 1a), each containing intrinsic voltage-sensing and Ca2+-sensing domains. However, BK channels may also contain any of two different families of regulatory proteins, β and γ, which help define tissue-specific functional properties of a BK complex. The four members of the β family (β1–β4) and the four members of the recently discovered γ family (γ1–γ4) differentially regulate BK function, influencing Ca2+-dependence of activation2,5,6,10,11, current inactivation4,5,12,13 and even pharmacology5,14.
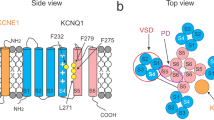
(a–b) Cartoons of the predicted topology of α-, β2- and γ1-subunits. The basic topology of β2- and γ1-subunits are shared with other members of their respective subunit families. (c) Schematic of deduced position of α- and β1-subunit TM segments in a BK channel complex viewed at the extracellular face17. Grey, pink and blue ellipses highlight voltage sensor domain (VSD), pore domain (PD) and β-subunit TMs, respectively. (d–g) Typical currents arising from (co)expression of BK subunits in oocytes: α alone, α+γ1, α+β2, or α+β2+γ1, respectively, at 0 (left) or 10 μM intracellular [Ca2+] (right); left and right currents were from the same patch using the same voltage protocol (depicted for each case). (h–i) Comparison among activation curves (G–V) at 0 Ca2+ or 10 μM of Ca2+, respectively, generated from sets of experiments as in panels (d–g). Notice the overlap between activation curves at 0 Ca2+ of α+γ1 and α+β2+γ1. G–V fits to single Boltzmann distributions yielded the following parameters: At 0 Ca2+, α-alone (Vh=193±6 mV, z=0.7±0.02 e0, n=5); α+β2ΔNt (Vh=182±4 mV, z=0.7±0.03 e0, n=5) ; α+γ1 (Vh=29.0±4.7 mV, z=1.3±0.08 e0, n=7); α+β2+γ1 (Vh=28.9±3.4 mV, z=0.9±0.04 e0, n=9). At 10 μM of Ca2+, α-alone (Vh =22.6±3 mV, z=1.4±0.12 e0, n=5); α+β2ΔNt (Vh=−9.2±4.4 mV, z=1.6±0.87 e0, n=5); α+γ1 (Vh=−95.1±3.9 mV, z=1.4±0.05 e0, n=8). (j) Averages of τinact at voltages up to +180 for α+β2+γ1 at 0 Ca2+ (n=9) and α+β2 at 10 Ca2+ (n=6), respectively. All errors are s.e.m.
β- and γ-subunits are unrelated proteins with very different predicted structural topology. β-subunits contain two transmembrane (TM) segments linked by an extracellular loop bridged by multiple disulfide linkages and intracellular C and N termini15; γ-subunits contain a single TM segment, a cytosolic C terminus, and an extracellular N terminus with a large leucine-rich repeat-containing motif6,16 (Fig. 1b). Specific structural information for β- and γ-subunits is not available. For β-subunits, potential positions of TM segments in relation to α-subunit TM segments have been proposed17,18,19 (Fig. 1c). Individual BK channels can contain 0–4 β-subunits, with each subunit contributing in an energetically independent fashion to shift BK gating20. For γ-subunits, both the position in the channel complex and the α:γ stoichiometry remain unknown. In heterologously expressed α+γ1 channels, the γ1-induced gating shift occurs in an all-or-none manner, consistent with an elementary functional unit of γ1 (for example, monomer, dimer and tetramer) being sufficient to produce the full effect21. Here to determine whether different types of regulatory subunits coassemble in the same BK channels, we take advantage of the distinctive functional properties conferred on BK channels by β2- and γ1-subunits. Using ensemble and single molecule approaches we report that γ1 and 1–4 β2-subunits can be simultaneously present in the same BK channel and independently contribute to modulation of BK function.
Results
β2- and γ1-subunits coassemble in the same BK channel
The γ1-subunit produces a remarkable negative shift of 120–140 mV in the voltage-range of BK channel activation either in the presence or absence of Ca2+(2) (Fig. 1d,e,h,i). While BK channels composed of α alone require at least 10 μM Ca2+ to show appreciable open probability over physiologically relevant voltages, (Fig. 1h,i), α+γ1 channels show a similar probability in the total absence of intracellular Ca2+ (Fig. 1e,h). The ability of γ1 to shift BK gating in 0 Ca2+ contrasts with the absence of a gating shift produced by β2 (Fig. 1h) under the same conditions22.
The most readily identifiable effect of the β2-subunit is essentially complete inactivation that occurs following channel activation (Fig. 1f). Inactivation arises from the cytosolic N terminus of the β2-subunit4,5,23. Since BK channels can contain 1–4 β2-subunits, each acting independently, single α+β2 channels exhibit one of four distinct inactivation rates20. The inactivation time constant (τinact) therefore provides a measure of β2-subunit stoichioimetry within either a channel population or individual channels20. Although β2 produces little gating shift at 0 Ca2+(23) (Fig. 1h), gating of α+β2 channels at 10 μM Ca2+ is shifted about 40 mV leftward compared with α channels3,5,20 which is more clearly observed using a non-inactivating β2 variant (β2ΔNt) (Fig. 1i). Thus, for α+β2 channels, voltage steps up to +180 mV only weakly activate BK currents at 0 Ca2+ (Fig. 1f, left), while 10 μM Ca2+ produces robust activation of an inactivating current (Fig. 1f, right), whose τinact approaches a limiting value of ∼20 ms (Fig. 1j), indicating an average of 4 β2-subunits/channel in the population20. Another physiologically relevant property of inactivating channels, the voltage dependence of steady-state inactivation (SS-inactivation), is also an indicator of the presence of β2-subunits. During exposure of α+β2 channels to 10 μM Ca2+, the availability of non-inactivated channels to open is very low at voltages above −50 mV (Fig. 2a,c) with half-channel availability (Vh) at about −110 mV. Together, the distinctive effects of β2- and γ1-subunits provide useful signatures to verify the presence of β2 and γ1 in BK channels resulting from the coexpression of α+β2+γ1.
(a–b) Examples of the fractional availability of channels as function of the conditioning potential from oocytes injected with α+β2 (10 μM Ca2+) or α+β2+γ1 (0 Ca2+), respectively. Currents were evoked by the indicated protocols using conditioning potentials of 100 ms duration. (c–d) Curves of activation (open symbols) and fractional availability (filled symbols) as function of voltage obtained at 10 μM of Ca2+ for α+β2 or 0 Ca2+ for α+β2+γ1 channels, respectively, are plotted together. G–V fits to single Boltzmann distributions yielded the following parameters: α+β2 SS-inactivation (Vh=−110.8±2.3 mV, z=1.81±0.11 e0, n=7) α+β2-activation (Vh=−6.7±2.4 mV, z=0.96±0.31 e0, n=6); α+β2+γ1-SS-inactivation (Vh=−113.5±5.3 mV, z=1.56±0.12 e0, n=4), α+β2+γ1-activation (also shown in Fig. 1h). All errors are s.e.m.
We therefore coexpressed γ1+β2 subunits with α at relative mole fractions of message that would produce full effects of either γ1 or β2 alone20,21. The coexpression of α+β2+γ1 subunits results in a prominent inactivating outward current even with 0 Ca2+ (Fig. 1g, left), similar to currents resulting from the expression of α+β2 subunits when activated by 10 μM Ca2+ (Fig. 1f). The complete inactivation of currents obtained after coexpression of α+β2+γ1 subunits indicates that essentially all channels contain β2-subunits. Furthermore, that in such patches the voltage dependence of activation at 0 Ca2+ is shifted more than −120 mV (Fig. 1h) is diagnostic for the presence of γ1. When the same patch is activated with 10 μM Ca2+, essentially no current is observed (Fig. 1g, right) which suggests that α+β2+γ1 channels are constitutively inactivated with 10 μM Ca2+ at −160 mV. Indeed, the fractional availability of α+β2+γ1 currents at 0 Ca2+ (Fig. 2b) exhibits a voltage dependence very similar to that observed with 10 μM Ca2+ for α+β2 currents (Fig. 2c–d). The markedly leftward-shifted steady-state inactivation curve of α+β2+γ1 channels also confirms that both β2- and γ1-subunits can coassemble in the same BK channels.
β2 and γ1 occupy distinct positions in the BK channel complex
We next wondered whether channels containing both β2+γ1 subunits can contain a full set of four β2-subunits. We imagined two kinds of assembly scenarios: (1) an independent model (Fig. 3a), where the presence of γ1-subunits does not hinder the ability of four β2-subunits to fully populate a BK channel (β2 and γ1 occupy different positions), or (2) an occlusive model (Fig. 3b), where the presence of γ1 excludes the assembly of β2-subunits (β2 and γ1 occupy overlapping positions). These two models can be tested by examination of the τinact arising from a set of single α+β2+γ1 channels obtained under conditions in which relative β2 subunit expression varies20. If the independent model is valid, γ1-containing inactivating single channels should exhibit four distinct τinact (ref. 20), while the finding of less than four inactivation behaviours supports the occlusive model. By keeping both α and γ1 constant but decreasing β2 in the RNA injection mix, we obtained a set of single channels that exhibit inactivation consistent with the presence of β2, but gating shifts consistent with the presence of γ1 (Supplementary Fig. 1). The τinact observed from 31 channels appeared to group into four behaviours (Fig. 3c–f). For channels that might potentially contain either three or four β2-subunits, the expected differences in mean inactivation time constant are not great. We therefore examined in detail the ability of three- and four-component Gaussian functions to fit the distribution. In evaluating fits of the distribution, we considered the impact of two criteria. First, since inactivation arises from independent inactivation domains20, the faster components should bear a simple arithmetic relationship to the slowest component. Second, the s.d. for any component, although perhaps not well-constrained by the data, should be smaller than for any slower components, such that s1≥s2≥s3≥s4. Various considerations in the fitting process are given in the Methods and in Supplementary Fig. 2. For the case that the s.d. are constrained as mentioned or where the mean values of each component are defined by the slowest component, the τinact distribution is better fit by a four-component than by a three-component Gaussian distribution (Fig. 3g,h, Supplementary Fig. 2), indicating that channels containing the γ1-induced effect can also contain one, two, three or four β2-subunits. In the case of the three-component Gaussian, if the constraint on s.d. is relaxed, the best fit is reached with the fastest component having the largest s.d. of any component, suggestive that this fast component in fact arises from two populations. The superior fit of the four-component Gaussian supports the existence of individual channels containing 4 β2 and at least one γ1-subunit indicating that both types of auxiliary subunits occupy independent positions in the channel complex (Fig. 3a).
(a–b) Cartoons representing two distinctive assembly models: where α, white; β2, blue; γ1, red. (c–f) Representative single channels obtained from various α:β2:γ1 injection ratios (given above each set of traces). For each example, five consecutive traces of current recorded at 0 Ca2+ are shown together with the ensemble current average from 100 total traces recorded in the same conditions. Red lines: fit of a single exponential with the indicated τinact. (g–h) Grey bars represent the distribution of τinact from 31 single-channels (bin size: 2 ms). Red lines represent fits of the binned data to four- or three-component Gaussian distributions, respectively, both with the s.d. (sn) constrained such that s1≥ s2≥ s3…sn. Mean values (τn) and sum of the squares of the errors (SSQ) obtained from each fit are indicated. The other parameters of each Gaussian component, amplitude (An) and standard deviation, of the distributions are: four-component distribution (A1=2.1, s1=3.0; A2=3.5, s2=1.8; A3=5.6, s3=1.2; A4=4.3, s4=1.2) and three-component distribution (A1=2.05, s1=3.1; A2=2.4, s2=3.1; A3=3.6, s3=3.1).
β2 and γ1 independently contribute to BK gating shifts
Both β2 and γ1 produce leftward shifts in BK gating, but their effects are likely mediated by different mechanisms. Whereas the γ1-effect can be explained by stronger coupling between the voltage-sensor movement and channel activation2, the effects of β2 appear more complex22,24,25,26. We asked whether γ1 and β2 effects might be additive or occlusive. For better elucidation of the β2 effects, we compared the gating shift resulting from the coexpression of γ1+β2ΔNt versus that produced by each construct separately when coexpressed with BK-α-subunits (Fig. 4). The Vh arising from the simultaneous presence of γ1 and β2ΔNt approximately reflects the sum of the independent effects of γ1 and β2ΔNt alone. These results indicate that, whatever the underlying molecular mechanism of the Vh shift produced by the γ1-subunit, it is predominantly energetically independent of that produced by the β2-subunit. Furthermore, there is no inhibitory allosteric coupling between the auxiliary subunits themselves.
(a–b) Currents arising from coexpression of α+β2-inactivation removed (β2ΔNt) (a) or α+β2ΔNt+γ1 (b) with 10 μM Ca2+. (c) Comparison of the gating shift obtained by β2 with or without the γ1 expression. β2 produces an approximately 30–35 mV shift with or without γ1. With 10 μM Ca2+, Vh for (α alone)=+22.6±3.4 mV, (α+γ1)=−95.1±3.9 mV, (α+β2ΔNt)=−9.2±4.4 mV and (α+β2ΔNt+γ1)=−134.7±8.8 mV (n=5); ΔV(β2)=31.8, ΔV(γ1)=117.7, ΔV(β2ΔNt+γ1)=157.3. All errors are s.e.m.
Other β-subunits also coassemble with γ1 in BK channels
Can other β-subunits also coassemble with γ1 in BK channels? An earlier report suggested that the presence of β1-subunits may occlude the ability of γ1 to produce its gating shift2. Since the overexpression of one subunit might influence the successful expression of another, we used proportions of RNA for each subunit similar to those used in testing γ1+β2 coassembly with α-subunits. Using a β1 construct in which its N terminus was replaced by the β2-N terminus (β1/β2Nt) so that inactivation reports the presence of β1, we found that all BK channels resulting from coexpression of α+β1/β2Nt +γ1 simultaneously contain both types of regulatory subunits (Supplementary Fig. 3).
Discussion
The present work unambiguously shows that two different types of non-pore-forming regulatory subunits, β2 and γ1, can coassemble in the same functional BK channel and independently regulate channel function. The α+β2+γ1 combination generates a BK channel with novel functional properties, which subtly change depending on the stoichiometry of β2 in the multimeric complex. In a normal cellular environment, the simultaneous presence of β2+γ1 in BK channels might effectively remove them from availability for activation especially when intracellular calcium is increased. However, at 0 Ca2+, such channels would not be fully inactivated (Fig. 2d): the window of overlap between the fractional availability curve and the activation curve spans approximately −60 to −30 mV, a range corresponding to normal resting potentials in many types of cells, including neurons. This essentially defines a range of voltage over which any contributions of α+β2+γ1 channels might be dynamically regulated by inactivation. Given the large conductance of the BK channel, only a small fraction of the total BK population would have to undergo cycles of inactivation, recovery from inactivation and activation to have some influence on cell excitability. Although it is unknown whether β2+γ1 subunits are simultaneously present in any cell-type, reported message levels of both β2 and γ1-subunits in some tissues11,27 support the possibility that β- and γ-subunits will copartner in at least some cells. For any cells which may express both a β2- and a γ1-subunit, our results establish that these two distinct regulatory partners of BK channels can simultaneously and independently contribute to modulation of BK function and do not appear to hinder the assemble of each other into a channel. The possibility of coassembly would also likely apply to other members of the β and γ families, as supported by our results with β1+γ1 subunits. Our findings highlight not only the critical importance of defining the identity of protein partners in native multimeric complexes within a cell or a specific cell location, but also the importance of understanding how individual contributions of distinct regulatory components and their stoichiometry can define fundamental properties of the complex.
Methods
Constructs
Primary constructs were mouse α (SLO1) (ref. 9), human LRRC26 (γ1) (ref. 11) and human β2 (ref. 23). In some experiments β2ΔNt (the first 33 amino acids were removed from the β2-N-terminal) or β1/β2ΔNt (first 43 amino acids from the β2-N terminus replaced the first 11 amino acids of the β1-N terminus) were used.
Oocyte expression
Stage IV Xenopus laevis oocytes were used for channel expression. The complementary RNAs (cRNAs) of all constructs were prepared at approximately 1 μg μl−1. For macroscopic recordings, cRNA mixes containing (molar ratio): mSLO1 alone, mSLO1+hβ2 (1:6.5), mSLO1+hLRRC26 (1:4) or mSLO1+hβ2+hLRRC26 (1:6.5:4), were diluted 1:5 before injection. For single channel recordings, mSLO1:hβ2:hLRRC26 (1: 0.65:4) and (1:0.16:4) were diluted 1:20–1:100 before injection. β2ΔNt or β1/β2ΔNt cRNAs were used at the same molar ratio as β2 in some experiments. Oocytes were used 2–5 days after injection, except for single channel recordings in which they were used 1–2 days after injection. Maintenance of frogs and isolation of oocytes following procedures approved by the Washington University in St Louis Institutional Animal Care and Use Committee.
Electrophysiology
Borosilicate glass capillaries (1B150F-4, World Precision Instruments) were pulled to diameters resulting in resistances of 1–2 or 5–6 MΩ for macroscopic and single channel recordings, respectively. Pipettes were coated with Sylgard 184 (Dow Chemical Corp.) and fire-polished. Currents were recorded in the inside-out patch configuration using an Axopatch 200B amplifier (Molecular Devices) and the Clampex program from the pClamp software package (Molecular Devices). Gigaohm seals were formed in frog Ringer (in mM, 115 NaCl, 2.5 KCl, 1.8 CaCl2, 10 HEPES, pH 7.4) and, after patch excision, moved into flowing test solutions. The pipette/extracellular solution was (in mM): 140 K-methanesulfonate, 20 KOH, 10 HEPES, 2 MgCl2, pH 7.0. Test solutions of different Ca2+ contained28 140 mM methanesulfonate, 20 mM KOH, 10 mM HEPES with pH adjusted to 7.0 (ref. 28). N-(2-hydroxyethyl)ethylenediamine-triacetic acid (HEDTA) was used for 10 μM Ca2+ and 5 mM ethylene glycol-bis(2-aminoethylether)-N,N,N′,N′-tetraacetic acid (EGTA) for 0 μM Ca2+solutions. The 10 μM Ca2+ solution was titrated to appropriate pCa with Ca-MES and calibrated against solutions of defined Ca2+ concentrations (World Precision Instruments) using a Ca2+-sensitive electrode. Test solutions were applied directly to patches via a large bore pipette tip containing multiple independent solution lines. All experiments were at room temperature (∼22–25 °C).
Data analysis
Analysis were made using Clampfit (Molecular Devices). For non-inactivating currents, conductance values (G) were obtained from the tail currents, while for inactivating currents the peak current was used. G–V data sets were fitted with a single Boltzmann function:  , where Vh represents the voltage of half activation and z is the valence of the voltage dependence. Notice that the α+γ1 injection ratio used (1:4 molar ratio) results in around 90% of BK channels expressed containing the full γ1-induced effect21, in which case the fit to a single Boltzmann distribution provides a good estimation of Vh and z for α+γ1 currents. Macroscopic inactivation time constants (τinact) were obtained by fitting each current decay to a single exponential function.
, where Vh represents the voltage of half activation and z is the valence of the voltage dependence. Notice that the α+γ1 injection ratio used (1:4 molar ratio) results in around 90% of BK channels expressed containing the full γ1-induced effect21, in which case the fit to a single Boltzmann distribution provides a good estimation of Vh and z for α+γ1 currents. Macroscopic inactivation time constants (τinact) were obtained by fitting each current decay to a single exponential function.
All error estimates are SEM.
Single-channel traces were first processed using digital subtraction of leak and capacitive current defined from traces lacking any channel opening. τinact for each single channel was estimated from the ensemble current average of 50–100 identical sweeps recorded at 0 Ca2+. The histogram of τinact was generated using a bin size of 2 ms and the distribution was fitted to the sum of three or four components Gaussian function:

where An, τn and sn represent the amplitude, mean-τinact and s.d. of each component, respectively, with n=1 for the parameters of the slowest inactivating component, n=2 for the second slower and so on. Since each measured τinact represents the mean of an exponentially distributed population, the s.d. for the average of those grouped in the slowest component of the τinact-distribution should be larger than for faster components, such that s1≥s2≥s3≥s4. Fitting to the sum of three or four Gaussian functions was first made allowing free variation of all fitting parameters. However, in both cases the rank of s.d. do not follow the expected criteria mentioned before: the four-component fit yields a very narrow third-component (very small s3), while the three-component fit results with the fastest component having the largest s.d. (s3) among all components (Supplementary Fig. 2a–b). Two factors may contribute to such deviations: first, that the numbers of entries in the histogram are insufficient to accurate define all aspects of each component and, second, that some components may actually arise from multiple components. We then constrained the s.d. in both cases to fulfil the expected criteria (s1≥s2≥…sn) (see results in Fig. 3g–h). Furthermore, we took advantage of the idea that inactivation results from the independent movement of each inactivation domain to the central cavity of the pore20. Based on this latter consideration, in some cases we also constrained all mean values of each component to be dependent on the slowest component (Supplementary Fig. 2c–d). All cases yield better fits using four than three-component Gaussian distributions.
Chemicals
Salts and buffers were obtained from Sigma.
Additional information
How to cite this article: Gonzalez-Perez, V. et al. Two classes of regulatory subunits coassemble in the same BK channel and independently regulate gating. Nat. Commun. 6:8341 doi: 10.1038/ncomms9341 (2015).
References
Berkefeld, H. et al. BKCa-Cav channel complexes mediate rapid and localized Ca2+-activated K+ signaling. Science 314, 615–620 (2006).
Yan, J. & Aldrich, R. W. LRRC26 auxiliary protein allows BK channel activation at resting voltage without calcium. Nature 466, 513–516 (2010).
Brenner, R., Jegla, T. J., Wickenden, A., Liu, Y. & Aldrich, R. W. Cloning and functional characterization of novel large conductance calcium-activated potassium channel beta subunits, hKCNMB3 and hKCNMB4. J. Biol. Chem. 275, 6453–6461 (2000).
Wallner, M., Meera, P. & Toro, L. Molecular basis of fast inactivation in voltage and Ca2+-activated K+ channels: a transmembrane beta-subunit homolog. Proc. Natl Acad. Sci. USA 96, 4137–4142 (1999).
Xia, X. M., Ding, J. P. & Lingle, C. J. Molecular basis for the inactivation of Ca2+- and voltage-dependent BK channels in adrenal chromaffin cells and rat insulinoma tumor cells. J. Neurosci. 19, 5255–5264 (1999).
Yan, J. & Aldrich, R. W. BK potassium channel modulation by leucine-rich repeat-containing proteins. Proc. Natl Acad. Sci. USA 109, 7917–7922 (2012).
Barrett, J. N., Magleby, K. L. & Pallotta, B. S. Properties of single calcium-activated potassium channels in cultured rat muscle. J. Gen. Physiol. 331, 211–230 (1982).
Latorre, R., Vergara, C. & Hidalgo, C. Reconstitution in planar lipid bilayers of a Ca2+-dependent K+ channel from transverse tubule membranes isolated from rabbit skeletal muscle. Proc. Natl Acad. Sci. USA 79, 805–809 (1982).
Xia, X. M., Zeng, X. & Lingle, C. J. Multiple regulatory sites in large-conductance calcium-activated potassium channels. Nature 418, 880–884 (2002).
McManus, O. B. et al. Functional role of the beta subunit of high conductance calcium-activated potassium channels. Neuron 14, 645–650 (1995).
Yang, C., Zeng, X. H., Zhou, Y., Xia, X. M. & Lingle, C. J. LRRC52 (leucine-rich-repeat-containing protein 52), a testis-specific auxiliary subunit of the alkalization-activated Slo3 channel. Proc. Natl Acad. Sci. USA 108, 19419–19424 (2011).
Xia, X. M., Ding, J. P., Zeng, X. H., Duan, K. L. & Lingle, C. J. Rectification and rapid activation at low Ca2+ of Ca2+-activated, voltage-dependent BK currents: consequences of rapid inactivation by a novel beta subunit. J. Neurosci. 20, 4890–4903 (2000).
Uebele, V. N. et al. Cloning and functional expression of two families of beta-subunits of the large conductance calcium-activated K+ channel. J. Biol. Chem. 275, 23211–23218 (2000).
Meera, P., Wallner, M. & Toro, L. A neuronal beta subunit (KCNMB4) makes the large conductance, voltage- and Ca2+-activated K+ channel resistant to charybdotoxin and iberiotoxin. Proc. Natl Acad. Sci. USA 97, 5562–5567 (2000).
Knaus, H. G., Eberhart, A., Kaczorowski, G. J. & Garcia, M. L. Covalent attachment of charybdotoxin to the beta-subunit of the high conductance Ca(2+)-activated K+ channel. Identification of the site of incorporation and implications for channel topology. J. Biol. Chem. 269, 23336–23341 (1994).
Dolan, J. et al. The extracellular leucine-rich repeat superfamily; a comparative survey and analysis of evolutionary relationships and expression patterns. BMC Genomics 8, 320–343 (2007).
Liu, G. et al. Location of modulatory beta subunits in BK potassium channels. J. Gen. Physiol. 135, 449–459 (2010).
Wu, R. S. et al. Location of the beta 4 transmembrane helices in the BK potassium channel. J. Neurosci. 29, 8321–8328 (2009).
Liu, G. et al. Locations of the beta1 transmembrane helices in the BK potassium channel. Proc. Natl Acad. Sci. USA 105, 10727–10732 (2008).
Wang, Y. W., Ding, J. P., Xia, X. M. & Lingle, C. J. Consequences of the stoichiometry of Slo1 alpha and auxiliary beta subunits on functional properties of large-conductance Ca2+-activated K+ channels. J. Neurosci. 22, 1550–1561 (2002).
Gonzalez-Perez, V., Xia, X. M. & Lingle, C. J. Functional regulation of BK potassium channels by gamma1 auxiliary subunits. Proc. Natl Acad. Sci. USA 111, 4868–4873 (2014).
Orio, P. & Latorre, R. Differential effects of beta 1 and beta 2 subunits on BK channel activity. J. Gen. Physiol. 125, 395–411 (2005).
Xia, X. M., Ding, J. P. & Lingle, C. J. Inactivation of BK channels by the NH2 terminus of the beta2 auxiliary subunit: an essential role of a terminal peptide segment of three hydrophobic residues. J. Gen. Physiol. 121, 125–148 (2003).
Hoshi, T., Pantazis, A. & Olcese, R. Transduction of voltage and Ca2+ signals by Slo1 BK channels. Physiology (Bethesda) 28, 172–189 (2013).
Sun, X. et al. The interface between membrane-spanning and cytosolic domains in Ca(2)+-dependent K+ channels is involved in beta subunit modulation of gating. J. Neurosci. 33, 11253–11261 (2013).
Contreras, G. F., Neely, A., Alvarez, O., Gonzalez, C. & Latorre, R. Modulation of BK channel voltage gating by different auxiliary beta subunits. Proc. Natl Acad. Sci. USA 109, 18991–18996 (2012).
Martinez-Espinosa, P. L., Yang, C., Gonzalez-Perez, V., Xia, X. M. & Lingle, C. J. Knockout of the BK beta2 subunit abolishes inactivation of BK currents in mouse adrenal chromaffin cells and results in slow-wave burst activity. J. Gen. Physiol. 144, 275–295 (2014).
Zhang, Z., Zhou, Y., Ding, J. P., Xia, X. M. & Lingle, C. J. A limited access compartment between the pore domain and cytosolic domain of the BK channel. J. Neurosci. 26, 11833–11843 (2006).
Acknowledgements
This work was supported by GM-081748 to CJL.
Author information
Authors and Affiliations
Contributions
V.G.-P. and C.J.L. designed research, analysed data and prepared the manuscript. V.G.-P collected the data. X.-M.X. contributed new reagents/analytical tools.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-3 (PDF 279 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Gonzalez-Perez, V., Xia, XM. & Lingle, C. Two classes of regulatory subunits coassemble in the same BK channel and independently regulate gating. Nat Commun 6, 8341 (2015). https://doi.org/10.1038/ncomms9341
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms9341
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.