Abstract
Low estimated lunar volatile contents, compared with Earth, are a fundamental observation for Earth–Moon system formation and lunar evolution. Here we present zinc isotope and abundance data for lunar crustal rocks to constrain the abundance of volatiles during the final stages of lunar differentiation. We find that ferroan anorthosites are isotopically heterogeneous, with some samples exhibiting high δ66Zn, along with alkali and magnesian suite samples. Since the plutonic samples were formed in the lunar crust, they were not subjected to degassing into vacuum. Instead, their compositions are consistent with enrichment of the silicate portions of the Moon in the heavier Zn isotopes. Because of the difference in δ66Zn between bulk silicate Earth and lunar basalts and crustal rocks, the volatile loss likely occurred in two stages: during the proto-lunar disk stage, where a fraction of lunar volatiles accreted onto Earth, and from degassing of a differentiating lunar magma ocean, implying the possibility of isolated, volatile-rich regions in the Moon’s interior.
Similar content being viewed by others
Introduction
The present-day inventory of volatile elements in inner Solar System terrestrial bodies is a function of initial delivery and subsequent volatile depletion histories. Unravelling this history can, therefore, elucidate mechanisms of planetary growth and differentiation. In the case of the Earth–Moon system, the contrasting fate of volatiles, between a relatively volatile-poor Moon and a water-rich Earth, provides important evidence for their formation and evolution. Despite their clear importance, however, the fate of the volatile elements and the origin of the volatile depletion of the Moon is not well understood1,2. An important observation of Apollo lunar samples was their strong depletion in volatile elements compared with terrestrial samples3. More recent studies of the volatile content in the Moon have shown that isotopes of moderately volatile elements, such as Cl and Zn, are highly depleted, compared with bulk silicate Earth4,5,6. In addition, some lunar rocks are enriched in the heavier isotopes of Fe compared with bulk silicate Earth7. This observation was originally interpreted in terms of Fe volatilization during the giant impact7; however, igneous processes provide a more robust interpretation for these results8,9. In contrast, studies of OH contents and D/H in apatites from mare basalts, as well as Apollo pyroclastic glass beads, have argued for elevated volatile contents in portions of the lunar interior10,11,12,13. Reconciling these seemingly contrasting observations remains critical to examining models of lunar formation (for example, ref. 14) and of lunar mantle differentiation15, since a volatile-rich Moon is not a logical consequence of large-scale collisional events, or magma ocean differentiation4. To examine the origin of lunar volatiles, we employ high-precision Zn isotopes and abundances. Zinc is a moderately volatile element with a 50% condensation temperature of 726 K under solar nebula conditions16. Isotopic fractionation during magmatic processes has a negligible effect (<0.1‰ for δ66Zn; per mil deviation of 66Zn/64Zn from the JMC-Lyon standard)17. However, Zn isotopes are highly fractionated during high-temperature volatilization in planetary rocks and variations in δ66Zn larger than 1‰ in terrestrial igneous rocks have only been observed in association with evaporation events18,19. The absence of isotopic fractionation during igneous processes makes Zn isotopes powerful tracers of high-temperature evaporation during volatile depletion episodes in planets. Recent work on lunar samples has focused largely on using Zn isotopes to study mare basalts, regolith and pyroclastic glass beads6,18,20. These studies have shown that mare basalts are consistently enriched in the heavier Zn isotopes compared with terrestrial basalts, which has been interpreted as a consequence of a whole-scale evaporation event on the Moon6, or during magma ocean differentiation4. It has also been suggested that similar isotopic fractionation could possibly have occurred during eruptive degassing of volatiles from mare basalt lavas into vacuum5.
To obtain the most comprehensive understanding of the Zn isotopic composition of lunar samples, we investigated the Zn isotopic compositions and abundances of lunar crustal rocks, which include magnesian and alkali suite samples (collectively termed here as MGS) and ferroan anorthosite (FAN) plutonic rocks. MGS are interpreted to have formed within the FAN-dominated lunar crust21 and, therefore, have not experienced degassing in a vacuum. FAN samples were formed by crystallization of a plagioclase floatation crust above a magma ocean after >80% differentiation21. FANs have highly variable volatile element abundances, with Zn ranging from ∼0.1 to>50 p.p.m. (for example, ref. 22), possibly consistent with transport and redistribution of Zn at the lunar surface. New data are also presented for the pyroclastic green glass 15426 to further examine whether pyroclastic glasses are enriched in the light isotopes of Zn due to recondensation of isotopically light vapour18. Additional data are also reported for three lunar regolith samples and several mare basalt samples to confirm previously observed heavy Zn isotope enrichment in these lithologies6,18,20.
Results
Lunar basalts
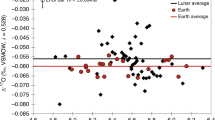
New data (Supplementary Table 1) for four high-Ti basalts (δ66Zn=+1.5±0.6‰, 2 s.d.) and three low-Ti basalts (δ66Zn=+1.5±0.4‰, 2 s.d.) (Supplementary Note 1) fall within the range observed previously for mare basalts6,20, and confirm that there is a consistent enrichment in the heavier isotopes of Zn compared with terrestrial basaltic rocks (δ66Zn=+0.28±0.05‰ for the bulk silicate Earth17) (Fig. 1). Combining our new data together with literature data6,20 (Supplementary Table 2), we calculate a new average value for lunar basalts of δ66Zn=+1.4±0.5‰ (2 s.d., n=26). A few exceptions (five samples out of 30) are isotopically lighter and are excluded from this average due to secondary condensation effects (Supplementary Discussion).
The grey dashed box in a represents the magnified area shown in b. Lunar mare basalts, alkali and magnesium suite samples are enriched in the heavier isotopes of Zn in comparison with the terrestrial mantle composition (orange square). Ferroan anorthosites exhibit significant isotopic variability, to light and heavy Zn isotopic compositions. Isotopic data for lunar samples are from this study and refs 6, 18, 20. The bulk silicate Earth value is from ref. 17.
Lunar crust and interior
In contrast to the relative homogeneity of mare basalts, FAN samples show large Zn isotopic variability (δ66Zn between −11.4 and +4.2‰) and the light isotope enrichment is correlated with the reciprocal Zn abundance (Fig. 2). In comparison, the two MGS rocks that we measured are both isotopically heavy (Supplementary Table 1).
Pyroclastic glass and regolith
The pyroclastic green glass (15426) is enriched in Zn compared with mare basalts (∼50 p.p.m. versus ∼2 p.p.m., which is in good agreement with previous studies22) and is isotopically lighter than the mare basalts (δ66Zn=−0.98‰). This result confirms consistent light isotopic enrichment of Zn in both high- and low-Ti pyroclastic glass beads18,20, with δ66Zn for the pyroclastic glasses ranging from −1 to −4.1‰. Three regolith samples are enriched in the heavier isotopes of Zn, with δ66Zn up to +6.4‰, confirming previous results18,20 that the lunar regolith is isotopically heavier than lunar basalts through impact gardening processes (Supplementary Discussion). The Zn abundances of the different samples fall within the range of previous measurements: mare basalts: ∼1–5 p.p.m.; FAN: 0.6–75 p.p.m.; MGS: ∼3 p.p.m.; and pyroclastic green glass: ∼50 p.p.m. (Supplementary Table 2).
Discussion
Enrichment in the lighter isotopes of Zn in Apollo 17 glasses was originally interpreted as the consequence of degassing of basaltic magma during a lava-fountaining event18. During magmatic ascent, the volatile elements are thought to have vapourized, leading to light isotope enrichment through kinetic isotopic fractionation. This vapour is then considered to have recondensed and coated the bead surfaces18. The new results on the Apollo 15 green glass (15426) offer the same interpretation for this pyroclastic deposit. The green glass 15426 is coarser (with a mean radius of the grains of ∼100 μm) than the two Apollo 17 glasses (mean radii of ∼50 μm)23. Therefore, a prediction, based on surface/volume, is for more limited light isotope enrichment of Apollo 15 glass beads due to surface/volume effects, as our results show (Apollo 15426 δ66Zn∼−1‰, [Zn]∼53 p.p.m. versus Apollo 74220 δ66Zn −3.4 to −4‰ and [Zn]∼230 p.p.m.).
Enrichment in the heavier isotopes of Zn in lunar mare basalts confirms that most lunar mare basalts have a homogeneous Zn isotopic composition, with 25 samples (out of 30) enriched by >1‰ (δ66Zn=+1.42±0.5‰; 2 s.d., n=26). It has previously been argued that the heavy isotope enrichment in mare basalts reflects a global phenomenon, where basalts representing four different Apollo mission sampling locations (Apollo 10, 12, 15 and 17), as well as unknown regions sampled by lunar meteorite LaPaz Icefield 02205, all show nearly identical enrichments6. The heavy Zn isotopic composition in the source of the mare basalts together, with the depletion in absolute abundances of moderately volatile elements, suggests that the lunar material suffered a whole-scale high-temperature evaporation process, with the preferential partitioning of the lighter isotopes of Zn in the vapour. This evaporation process most probably occurred during large-scale melting of the Moon, such as during the giant impact6.
Highland rocks represent the primary crust of the Moon and are the oldest known lunar samples (for example, ref. 24). The large range of Zn abundances in FAN samples suggests two possible scenarios: (1) the source of the FAN had a highly variable Zn content, which is reflected in the FAN samples, or (2) the Zn abundance represents secondary redistribution of Zn at the surface of the Moon during cataclasis of the FAN, as well as mixing with some chondritic components. Partial melting does not seem to extensively fractionate Fe/Zn due to the fact that these two elements share similar ionic radii25. The five different FAN samples analysed here have variable Fe/Zn (Supplementary Table 3), which, therefore, suggests that either the source of the anorthosites had variable Fe/Zn, that Fe and Zn were fractionated during plagioclase crystallization, or that the Zn elemental variations are a result of secondary evaporative processes. Since Zn isotopes are not fractionated by >0.2‰ during igneous processes17, the large Zn isotopic variations, which are correlated with the Zn abundance (Fig. 2), indicate that the Zn systematics of FAN are due to high-temperature evaporation effects. Thus, Zn-rich FAN contains a large fraction of an isotopically light vapour, while the most Zn-poor FAN (15415) retains primary magmatic characteristics.
Apollo sample 15415 is unbrecciated, pristine26 and is enriched in the heavier isotopes of Zn (δ66Zn=4.2‰). On the other hand, 65315, which is enriched in Zn (75 p.p.m.) is a brecciated cataclastic anorthosite with exogenously derived (meteoritic) highly siderophile element additions27. Similarly, 67955 is also a brecciated sample, formed from an impact melt, with a significant component of meteoritic siderophile elements28 and has disturbed Zn isotope systematics. These lines of evidence indicate that the brecciated samples incorporated isotopically light Zn-rich vapour during cataclasis or residence on the lunar surface. Transport of Zn at the surface of the Moon, during later impact bombardment of the crust and during the assimilation of exogenous Zn, implies that the Zn isotopic composition of cataclastic FAN samples do not represent the composition of their source reservoirs.
Significant enrichments in the heavy isotopes of Zn observed in the MGS, which solidified within the lunar crust, point towards a higher volatile depletion in their source than mare basalts. The origin of the MGS is generally interpreted as magmatic intrusions within the FAN crust (for example, ref. 21), formed by either a very high degree of partial melting29 or by assimilation of FAN crust30. Evaporation and condensation in a vacuum at the surface of the Moon has, therefore, played no role in the composition of these samples. Enriched heavier isotopes of Zn observed in the MGS points towards a source already depleted in volatile elements. In turn, this provides strong support to loss of Zn and moderately volatile elements during a whole-scale evaporation of the Moon early in its history either following the giant impact or during a lunar magma ocean phase4,6, and not locally during degassing, as has been proposed for some of the mare basalts (ref. 5). A third scenario would be that the impactor was already depleted in volatile elements before the giant impact (for example, refs 31, 32). The new data cannot constrain this scenario; however, a second process of volatile loss is required to explain the differences in Zn isotopic composition between mare basalts and pristine highland samples. A more likely scenario is, therefore, that volatile element of the Moon occurred during lunar formation and differentiation.
An outstanding question is how Zn and other moderately volatile elements were lost from the Moon, while water, which is far more volatile (for example, ref. 32), seems to have been retained in some samples (for example, refs 10, 11, 12, 13). It has been suggested that hydrodynamic escape after the giant impact was not a viable mechanism to explain the loss of water (or potassium) from the Earth–Moon system and that the volatile element content of the Moon was inherited from the precursor material33. However, this model fails to explain the extreme volatile depletion of the mare basalt source rocks, which are the most representative derivative rock type from the lunar mantle, or the later evolution of volatiles in the Moon revealed from crustal rocks. In a two-phase vapour-melt numerical model of the dynamical and thermodynamical evolution of the proto-lunar disk34, where the vapour phase is viscous due to magnetorotational instability, the volatile elements are predicted to accrete to Earth, leaving the material that formed the Moon enriched in refractory elements and correspondingly depleted in volatiles. Calculations of the chemical composition of the vapour of the lunar protoplanetary disk35 indicates that Zn enters the volatile phase of the disk at temperatures <3,000 K, leaving the Moon depleted in Zn and more volatile elements. Our new results support this mode of origin, but also suggest that continued volatile depletion in a lunar magma ocean is required4 to explain differences in δ66Zn between mare basalts and pristine lunar crustal rocks.
Methods
Sample preparation
One-gram chips of samples (Supplementary Note 1) were powdered with an agate mortar and pestle. Samples were then dissolved in a mixture of HF/HNO3 for several days in Teflon beakers at ∼140 °C. Zinc purification was achieved in a 0.25-ml column using AG-1X8 (200–400 mesh) anion-exchange resin. After applying the sample solution in HBr onto the resin, further HBr was added to remove elements causing matrix effects. Purified Zn was collected in a HNO3 solution following the procedure of ref. 18. The resulting Zn fraction was further purified in smaller 100-μl columns following the same ion-exchange chromatography procedure.
Sample analysis
Samples were analysed using a Thermo-Fisher Neptune Plus multicollector inductively coupled plasma mass spectrometer at the Department of Earth and Planetary Sciences, Washington University in St Louis. Masses 62, 63, 64, 65, 66, 67 and 68 were measured by Faraday cups positioned, respectively, and the intensity of 62Ni was measured to control and correct the isobaric interference of 64Ni (ref. 6). All samples fell on the mass-dependent fractionation line (Supplementary Fig. 1), and the typical analytical uncertainty (2 s.d.), is 0.04‰ for δ66Zn and 0.05‰ for δ67Zn and δ68Zn. Instrumental mass bias was corrected by bracketing of the standard and the sample and is further discussed in ref. 17. The Zn concentrations were also obtained from multicollector inductively coupled plasma mass spectrometer data by comparing the intensity of the sample signal with that of the standard, with an estimated precision of ±10%.
Additional information
How to cite this article: Kato, C. et al. Extensive volatile loss during formation and differentiation of the Moon. Nat. Commun. 6:7617 doi: 10.1038/ncomms8617 (2015).
References
Taylor, S. R. & McLennan, S. M. Planetary Crusts: Their Composition, Origin and Evolution Cambridge Univ. Press (2009).
Taylor, S. R. The Moon re-examined. Geochim. Cosmochim. Acta 141, 670–676 (2014).
Wolf, R. & Anders, E. Moon and Earth: compositional differences inferred from siderophiles, volatiles, and alkalis in basalts. Geochim. Cosmochim. Acta 44, 2111–2124 (1980).
Day, J. M. D. & Moynier, F. Evaporative fractionation of volatile stable isotopes and their bearing on the origin of the Moon. Philos. Trans. A Math. Phys. Eng. Sci. 372, 20130259 (2014).
Sharp, Z. D., Shearer, C. K., McKeegan, K. D., Barnes, J. D. & Wang, Y. Q. The chlorine isotope composition of the Moon and implications for an anhydrous mantle. Science 329, 1050–1053 (2010).
Paniello, R. C., Day, J. M. D. & Moynier, F. Zinc isotopic evidence for the origin of the Moon. Nature 490, 376–379 (2012).
Poitrasson, F., Halliday, A. N., Lee, D. C., Levasseur, S. & Teutsch, N. Iron isotope differences between Earth, Moon, Mars and Vesta as possible records of contrasted accretion mechanisms. Earth Planet. Sci. Lett. 223, 253–266 (2004).
Weyer, S. et al. Iron isotope fractionation during planetary differentiation. Earth Planet. Sci. Lett. 240, 251–264 (2005).
Wang, K., Jacobsen, S. B., Sedaghatpour, F., Chen, H. & Korotev, R. L. Lunar dunite reveals the same iron isotopic composition of the bulk silicate Earth and Moon. Lunar Planet. Sci. Conf. 46, 1980 (2015).
Saal, A. E., Hauri, E. H., Van Orman, J. A. & Rutherford, M. J. Hydrogen isotopes in lunar volcanic glasses and melt inclusions reveal a carbonaceous chondrite heritage. Science 340, 1317–1320 (2013).
McCubbin, F. M. et al. Nominally hydrous magmatism on the Moon. Proc. Natl Acad. Sci. USA 107, 11223–11228 (2010).
Saal, A. E. et al. Volatile content of lunar volcanic glasses and the presence of water in the Moon’s interior. Nature 454, 192–195 (2008).
Hauri, E. H., Saal, A. E., Rutherford, M. J. & Van Orman, J. A. Water in the Moon’s interior: Truth and consequences. Earth Planet. Sci. Lett. 409, 252–264 (2015).
Cameron, A. The origin of the Moon and the single impact hypothesis V. Icarus 126, 126–137 (1997).
Wood, J. A., Dicky, J. S. Jr., Marvin, U. B. & Powell, B. N. Lunar anorthosites and a geophysical model of the moon. Proc. Apollo 11 Lunar Sci. Conf. 1, 965–988 (1970).
Lodders, K. Solar System abundances and condensation temperatures of the elements. Astrophys. J. 591, 1220–1247 (2003).
Chen, H., Savage, P. S., Teng, F.-Z., Helz, R. T. & Moynier, F. Zinc isotopic fractionation during magmatic differentiation and the isotopic composition of the bulk Earth. Earth Planet. Sci. Lett. 369-370, 34–42 (2013).
Moynier, F., Albarède, F. & Herzog, G. F. Isotopic composition of zinc, copper, and iron in lunar samples. Geochim. Cosmochim. Acta 70, 6103–6117 (2006).
Moynier, F. et al. Nature of volatile depletion and genetic relationships in enstatite chondrites and aubrites inferred from Zn isotopes. Geochim. Cosmochim. Acta 75, 297–307 (2011).
Herzog, G. F., Moynier, F., Albarède, F. & Berezhnoy, A. A. Isotopic and elemental abundances of copper and zinc in lunar samples, Zagami, Pele's hairs, and a terrestrial basalt. Geochim. Cosmochim. Acta 73, 5884–5904 (2009).
Shearer, C. K. et al. Thermal and magmatic evolution of the Moon. Rev. Mineral. Geochem. 60, 365–518 (2006).
Ebihara, M., Wolf, R., Warren, P. H. & Anders, E. Trace elements in 59 mostly highland Moon rocks. Proc. Lunar Planet. Sci. Conf. 22, 417–426 (1992).
Morgan, J. W. & Wandless, G. A. Surface-correlated trace elements in 15426 lunar glasses. Proc. Lunar Planet. Sci. Conf. 15, 562–563 (1984).
Carlson, R. W. & Lugmair, G. W. The age of ferroan anorthosite 60025: oldest crust on a young Moon. Earth Planet. Sci. Lett. 90, 119–130 (1988).
Lee, C-T. A. et al. The redox state of arc mantle using Zn/Fe systematics. Nature 468, 681–685 (2010).
Ganapathy, R., Morgan, J. W., Krähenbühl, U. & Anders, E. Ancient meteoritic components in lunar highland rocks: clues from trace elements in Apollo 15 and 16 samples. Proc. Lunar Sci. Conf. 4, 1239–1261 (1973).
Day, J. M. D., Walker, R. J., James, O. B. & Puchtel, I. S. Osmium isotope and highly siderophile element systematics of the lunar crust. Earth Planet. Sci. Lett. 289, 595–605 (2010).
Norman, M. D. & Nemchin, A. A. A 4.2 billion year old impact basin on the Moon: U-Pb dating of zirconolite and apatite in lunar melt rock 67955. Earth Planet. Sci. Lett. 388, 387–398 (2014).
Longhi, J. Preliminary modeling of high pressure partial melting: Implications for early lunar differentiation. Proc. Lunar Planet. Sci. Conf. 12, 1001–1018 (1981).
Warren, P. H. Anorthosite assimilation and the origin of the Mg/Fe-related bimodality of pristine Moon rocks: Support for the magmasphere hypothesis. J. Geophys. Res. 91, D331–D343 (1986).
Albarède, F., Albalat, E. & Lee, C.-T. A. An intrinsic volatility scale relevant to the Earth and Moon and the status of water in the Moon. Meteorit. Planet. Sci. 50, 568–577 (2015).
Albarède, F. Volatile accretion history of the terrestrial planets and dynamic implications. Nature 461, 1227–1233 (2009).
Nakajima, M. & Stevenson, D. J. Hydrodynamic escape does not prevent the “wet” Moon formation. Lunar Planet. Sci. Conf. 45, 2770 (2014).
Charnoz, S. & Michaut, C. Evolution of the protolunar disk: dynamics, cooling timescale and implantation of volatiles into the Earth. Workshop on the Early Solar System Impact Bombardment III, 3002 (2015).
Visscher, C. & Fegley, B. Jr. Chemistry of impact-generated silicate melt-vapor debris disks. Astrophys. J. Lett. 767, L12 (2013).
Acknowledgements
We are grateful to CAPTEM and the NASA Johnson Space Center curatorial staff for provision of samples. This work was supported by funding from NASA (NNX12AH70G to F.M. and NNX11AG34G to J.M.D.D.). F.M. thanks the European Research Council for funding under the H2020 framework program/ERC grant agreement #637503 (Pristine), as well as the financial support of the UnivEarthS Labex program at Sorbonne Paris Cité (ANR-10-LABX-0023 and ANR-11-IDEX-0005-02), the ANR through a chaire d’excellence Sorbonne Paris Cité and the INSU through the PNP programme.
Author information
Authors and Affiliations
Contributions
C.K. collected the data and drafted the paper; F.M. and J.M.D.D. conceived the project and M.C.V. and J.K.D. assisted in the data collection. C.K., F.M. and J.M.D.D. wrote the paper and all authors contributed to discussion and interpretation of results.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figure 1, Supplementary Tables 1-3, Supplementary Note 1, Supplementary Discussion and Supplementary References (PDF 587 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Kato, C., Moynier, F., Valdes, M. et al. Extensive volatile loss during formation and differentiation of the Moon. Nat Commun 6, 7617 (2015). https://doi.org/10.1038/ncomms8617
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms8617
This article is cited by
-
Geochemical Constraints on the Origin of the Moon and Preservation of Ancient Terrestrial Heterogeneities
Space Science Reviews (2020)
-
An extremely heavy chlorine reservoir in the Moon: Insights from the apatite in lunar meteorites
Scientific Reports (2019)
-
Tin isotopes indicative of liquid–vapour equilibration and separation in the Moon-forming disk
Nature Geoscience (2019)
-
Tracing the formation and differentiation of the Earth by non-traditional stable isotopes
Science China Earth Sciences (2019)
-
Depletion of potassium and sodium in mantles of Mars, Moon and Vesta by core formation
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.