Abstract
Carbon nanotubes hold enormous technological promise. It can only be harnessed if one controls their chirality, the feature of the tubular carbon topology that governs all the properties of nanotubes—electronic, optical, mechanical. Experiments in catalytic growth over the last decade have repeatedly revealed a puzzling strong preference towards minimally chiral (near-armchair) tubes, challenging any existing hypotheses and making chirality control ever more tantalizing, yet leaving its understanding elusive. Here we combine the nanotube/catalyst interface thermodynamics with the kinetic growth theory to show that the unusual near-armchair peaks emerge from the two antagonistic trends at the interface: energetic preference towards achiral versus the faster growth kinetics of chiral nanotubes. This narrow distribution is inherently related to the peaked behaviour of a simple function, xe−x.
Similar content being viewed by others
Introduction
The broad interest in carbon nanotubes (CNTs), unceasing since their first clear observation1, has been fuelled by possible technological applications derived from their unique fundamental properties2,3,4. All of the latter are in turn determined by the helical fashion of folding a tube, specified by the chiral angle χ between its circumference and the zigzag motif in the honeycomb lattice of atoms, with χ=0 and χ=30° for the achiral types, zigzag and armchair. Alternatively, a pair of chiral indices (n,m) is commonly used, the integer components of the circumference vector2. In spite of such a defining role of chirality, most synthetic methods yield a broad distribution with mixed properties. To achieve control of the CNT type remains a great challenge; what physical mechanisms determine the chirality distribution and even why the nanotubes grow chiral is still unsettled.
Although the discovery paper by Iijima1 has already suggested one key, connecting the tube ability to grow with the kinks at its end and the screw dislocation model, it took almost two decades until the first equation5 related the speed of growth and chirality, R~sin χ. It should further be useful to think, in hindsight, of the probable causes of such a delay. Besides the difficulties of determining chirality in experiments, in theory it was ambiguous as to whether the chiral angle must be measured from the zigzag (as χ) or perhaps from the armchair (as χ−≡ 30°−χ) direction. While each choice appears valid from a pure symmetry standpoint, their kinetic predictions are opposite (with one favouring armchair and the other, zigzag CNTs), and thus one stumbles upon an immediate contradiction. Another diversion was due to a simple thermodynamic argument that the lower energy of the tube edge, rather than its kinetic advantage of having kinks, must determine the dominant CNT type, pointing towards the armchair tubes, especially (10,10) broadly discussed by Smalley et al.6
This situation, together with recent advances in synthesis featuring, in several cases, very narrow chiral distributions7,8,9, poses a compelling question as to which factors—thermodynamic preference to lower energy, or kinetic preference of higher speed—play a major role in defining the distribution of CNT product. The true answer appears to be ‘both’, and the analysis below shows how the subtle interplay of these physical factors defines the more probable chirality choices. In particular, it explains why at lower temperatures on solid catalyst particles the yield peaks near armchair types (n,n–1), never exactly armchair, although quite close.
The evolution of chemical vapour deposition (CVD) techniques and chirality characterization methods10 has led to improvements in chiral selectivity7,8,9,11,12,13,14,15,16, eventually reaching >50% fraction for a single-CNT type and ~90% for semiconducting tubes9. More intuitive strategies such as post-growth selection17,18, rational synthesis19,20,21, or seeding22,23,24 do, in principle, offer great selectivity but lack the scalability of CVD. Further improvements of the latter direct synthesis techniques thus call for better understanding of the growth process. Yet, its mechanism and especially the causes behind its occasional success in chiral preference remain puzzling. In modelling efforts25, direct molecular dynamics (MD) or Monte Carlo simulations, although invaluable in many respects, generally fail to produce nondefective CNT structures of a well-defined diameter and recognizable chirality, regardless of the precision of interatomic potentials—from classical26,27,28,29 to tight-binding30,31 and density functional theory (DFT)32. This is caused by the short accessible timescale due to sheer computational limitations. Alternating MD with Monte Carlo relaxation has produced ‘definable’33 but occasionally changing34 chirality. It is clear that a physical theory bridging the gap between atomistic dynamics and macroscopic scales is needed to interpret both the experiments and simulations.
In the present work, we propose a theory that reconciles the kinetic and thermodynamic arguments within a unified model. We show how the rigid state of the catalyst causes the two factors to exhibit opposite trends, where nucleation energetics penalizes chiral CNTs while growth rate penalizes achiral tubes. These two trends find a ‘compromise’ in minimally chiral CNTs, which explains why these peculiar types, such as (6,5), are robustly observed in many experiments.
Results
CNT abundance
We treat the CNT abundance by type as the result of the action of two main factors. At proper conditions, the nanotubes nucleation occurs, with probability Nn,m of certain chiral type. It is followed by the steady carbon accretion by each tube, with its growth rate Rn,m. After some time, the fraction of the tubes of chirality (n,m), that is, their relative abundance An,m in the accumulated material, is determined by the product of both these factors35, as An,m=Nn,m · Rn,m (see Supplementary Note 1 for a more general analysis). Instead of the chiral indices, using the tube diameter d and chiral angle χ gives A(χ,d)=N(χ,d)·R(χ,d). Below we explore the physical mechanisms defining the right-hand side, in particular the case of a solid catalyst with a rigid shape, which yields sharp chiral angle selectivity in A(χ,d), as empirical evidence suggests.
Continuum model
Before discussing the details of atomistic study, it is useful to explore the key ideas in terms of simpler continuum model, which not only offers valuable insight but is even able to make accurate overall predictions. During nucleation, as carbon atoms attach to a nascent CNT nucleus, adding new hexagonal and pentagonal rings to it, the chirality of a CNT becomes permanently ‘locked in’ when the final 6th pentagon is added to the hemispherical cap (barring further changes of CNT type if pentagon–heptagon defects form at the edge, which is generally a rare event36). From this six-pentagon nucleus a cylindrical CNT structure can further grow by adding only hexagons, in a periodic fashion. The free energy of the critical nucleus contains two contributions, G*=Gcap+Γ. The first one, Gcap, originates from the ‘elastic’ energy of cap per se and does not depend on χ (ref. 37). The second term Γ represents the contact interface between the sp2-carbon lattice edge and the metal catalyst, and does contain chirality dependence since the edge energy γ(χ) varies with the crystallographic orientation and Γ(χ,d) ≡ πdγ(χ). Whereas previous studies on edge energetics38 assumed either vacuum or a liquid-like catalyst that fully adapts to the edge profile, chiral-selective CVD growth is usually reported at comparatively low temperatures8,9 when the catalyst particle is solid9. Accordingly, the metal side of the interface is rather a rigid atomic plane, and the structure of this interface affects both the energy of the nucleus and the subsequent insertion of new C atoms during growth.
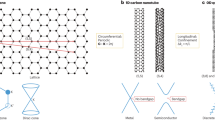
In Fig. 1 inset, the CNT is in contact with the catalyst which is represented locally as a continuous plane corresponding to an atomic terrace in a solid particle. The catalyst particle surface does not have to be planar. Since the important region is the narrow ring of CNT–catalyst contact, even a single atomic terrace running around the particle circumference will act essentially equivalent to a plane. The CNT is also continuous, but the kinks along its edge are retained according to the tube chirality, Fig. 1a–c. These kinks cause the gaps between the substrate and the CNT, shown in Fig. 1b,c for (n,3) and (n,1) tubes, with an associated energy penalty, relative to the tight contact in case of achiral tube in Fig. 1a. For the hexagonal lattice of CNT, the two fundamental achiral edges—armchair (A) and zigzag (Z)—form tight low-energy contacts. The interface energy for chiral tubes is higher, roughly in proportion with the number of kinks, which increases linearly with χ for near-Z tubes, or with χ− for near-A tubes. In other words, γ(x)≈γ+γ′·x, where x is the angular deviation from the achiral direction: near the Z-type x=χ, γ=γZ ≡ γ(0), and γ′=∂γ/∂χ|χ=0, or near the A-type x=χ−, γ=γA ≡ γ(30°), and γ′=−∂γ/∂χ|χ=30°.
Schematic representation of: (a) achiral; (b) multiple-kink chiral; and (c) single-kink chiral CNTs on a flat substrate. Unrolled CNT–substrate interfaces for (d) two-kink and (e) single-kink nanotubes show the nanotube tilt off the vertical, reducing the edge-substrate gap; the white dot in c and e marks the contact point. The abundance distributions A(χ)~χ e−χ computed as the product of nucleation (dotted) and growth rate (dashed) terms are shown in f for near-Z (blue) and near-A (red) chiralities. The inset illustrates a nascent CNT of diameter d on a solid catalyst.
Since γ(χ) is largest in the intermediate range of χ≈15°, such tubes are unlikely to nucleate, and one should focus on just the neighbourhoods of Z and A chiralities. Then we write:

The essential result here is that the nucleation probability falls rapidly as e−β·x with chiral angle x, and β=πdγ′/kBT. A distinction for single-kink cylinders, representing the nanotubes (n,1) and (n,n–1), should be noted. Their symmetry allows them to tilt off the vertical axis, improving the interface contact. Figure 1d,e illustrates it by ‘unrolling’ the CNT–substrate interface area for two-kink and single-kink tubes. In the latter case, the effect of tilt leads to a reduction of the tube-substrate separation, appearing as a sinusoid along the circumference as shown in Fig. 1e, enabling a substantial closure of the gap between substrate and tube edge, recovering up to as much as 70% of the energy penalty—see Supplementary Fig. 1 and Supplementary Note 2.
For the growth rate term R(χ,d), we augment the screw dislocation model5 by including the kinks created by thermal fluctuations on A and Z edges39, and accounting for the energy penalty ~1/d2 from the wall curvature. In the liquid-catalyst model, when the metal adapts to the CNT edge with a one-to-one termination, calculations suggest that the cost EA to create a pair of kinks on an A edge is zero, and consequently5 R∝χ. However, on a solid surface, creating a pair of kinks destroys the perfect contact between the CNT and substrate, costing energy. Therefore EA has a noticeable magnitude, and the dependence becomes bimodal with minima at the A and Z ends of the chiral angle range, and a maximum at the 19.1° magic angle39,40 of (2m,m) tubes. The final expression, linearized near the A and Z bounds of chirality, reads as follows,

where C=3.9 eV Å2 per atom is the bending rigidity of graphene41. The term x in parentheses corresponds to the density of geometry-imposed kinks, proportional to the vicinal-edge angular deviation from the main achiral direction, and the term  (typically small) represents the additional fluctuational kinks. The free-energy barriers for the initiation of a new atomic row on A or Z edge are E=EZ near the Z-type where
(typically small) represents the additional fluctuational kinks. The free-energy barriers for the initiation of a new atomic row on A or Z edge are E=EZ near the Z-type where  , or E=EA near the A-type where x=χ−.
, or E=EA near the A-type where x=χ−.
Multiplying together the nucleation and growth terms presents the key to understanding the observed selectivity for near-A chiralities9. At a given diameter,

a function with a sharp peak near zero (or near 30°). This is the essential result of our continuum consideration. Figure 1f illustrates this peaked distribution character. The two distributions for near-Z (blue) and near-A (red) chiral angles are plotted assuming equal interface energies and growth barriers: γA=γZ, EA=EZ. However, if either A or Z has a lower energy, the opposite peak distribution is additionally penalized by  , with ΔΓ=ΓZ−ΓA on the order of eV, and then one would expect to observe only one side of the distribution. These continuum-model predictions turn out to be remarkably robust. To see this, below we present the atomistic calculations of the relevant quantities, and proceed to simulate example CNT-type distributions.
, with ΔΓ=ΓZ−ΓA on the order of eV, and then one would expect to observe only one side of the distribution. These continuum-model predictions turn out to be remarkably robust. To see this, below we present the atomistic calculations of the relevant quantities, and proceed to simulate example CNT-type distributions.
Atomistic model
Computations were performed using a flat Ni(111) slab to represent the solid catalyst. We used a classical force-field MD sampling complemented with static DFT computations (on Ni and also Co slabs), as detailed in Methods.
To investigate the chiral selectivity of nucleation, two sets of CNTs are chosen, with d≈0.8 and 1.2 nm, to include the prominent in experiments (6,5)9 and (9,8)8 CNTs. Figure 2a shows the calculated CNT–substrate interface energies. The atomistic structures for the (9,0), (6,5), and (6,6) CNTs are shown in Fig. 2b, where the tilting of the (6,5) and (9,1) CNT to reduce the interface energy is seen clearly. We found that for the smaller-diameter set, for near-Z edges the Klein structure, in which a normal zigzag edge has an extra C atom (blue in Fig. 2b) attached to each hexagon42, is favored over the standard closed-hexagons, whereas the larger-diameter set shows little preference either way. All data display the same qualitative behaviour conforming to the above discussion, and are generally in good quantitative agreement. Both bounds of the chiral angle range (achiral CNTs) are energy minima, and the energy is higher for Z than for A tubes. The curves show the fit of γ(χ) using the analytical expression from earlier work38 for solid- and liquid-like cases.
(a) Interface energies calculated with MD (circles and triangles) and fitted with analytical expression (solid line) for two CNT sets (inset; dashed arcs denote the range of diameter variation in each set). Static DFT calculations on Ni and Co are also shown. Open and filled symbols denote regular (hexagonal) and Klein Z-edge structures—see sample atomistic structures in b. The dash-dotted line, calculated as γ(χ)=2γA sin χ+2γZ sin (30°−χ),38 corresponds to liquid-catalyst case. In b, the blue and red atoms highlight the Z and A edges, respectively.
Our computations pertaining to the growth kinetics are summarized in Fig. 3. We build upon our earlier approach for graphene39, adapting it for the case of CNTs. Figure 3a shows the energy changes with the addition of a new row of carbon atoms, dimer by dimer, for (6,6), (9,9) and (9,0) CNTs (see also Supplementary Figs 2 and 3 and Supplementary Note 3 for activation barrier calculations, showing the same trends). All three curves depart from the ‘nucleation to kink-flow’ scenario of graphene, the reason being the constantly changing tilt angle of the CNTs. However, both A curves (red, orange) show essentially the same height for the first dimer addition and the same maximum height (closer to the end). The Z curve bears the same qualitative character, having an initial and a final maximum. The system cycles through the curve with every new full ring of hexagons, after which ΔG returns to 0. The maximum height of each curve determines the free-energy barrier that needs to be overcome for successful addition of each new row of hexagons, ΔGA≈1.67−1.86 eV and ΔGZ>3 eV (calculations for the (9,0) CNT yield multiple intermediate structures with topological defects of energies lower than perfect hexagonal structures, an additional complication for Z-CNT growth). These are the terms that penalize the pure A and Z tubes, compared with the chiral ones (green line in Fig. 3a), by an additional factor  and thus effectively remove them from the product distribution, despite their favourable cap-nucleation energies. The atomic configurations for the first dimer addition to the (6,6) and (9,0) CNTs are shown in Fig. 3b. Figure 3c shows the concentrations of different site types—Z, A and K (kink)—as a function of χ38,39,40. Thin black lines show the intrinsic, topologically required values. Finally, Fig. 3d shows the CNT growth speed. Higher temperatures favour kink formation and promote the growth of zigzag and armchair CNTs. However, under realistic conditions, when kBT≪ΔG, the growth rate of achiral CNTs is negligible. Among different diameters d, the curvature of the wall penalizes insertion of C atoms into small-diameter CNTs, as in equation (2).
and thus effectively remove them from the product distribution, despite their favourable cap-nucleation energies. The atomic configurations for the first dimer addition to the (6,6) and (9,0) CNTs are shown in Fig. 3b. Figure 3c shows the concentrations of different site types—Z, A and K (kink)—as a function of χ38,39,40. Thin black lines show the intrinsic, topologically required values. Finally, Fig. 3d shows the CNT growth speed. Higher temperatures favour kink formation and promote the growth of zigzag and armchair CNTs. However, under realistic conditions, when kBT≪ΔG, the growth rate of achiral CNTs is negligible. Among different diameters d, the curvature of the wall penalizes insertion of C atoms into small-diameter CNTs, as in equation (2).
(a) Free-energy profiles during the growth of a new ring of hexagons on (red, orange) A and (blue) Z edges as a function of number of added atoms N. The green line corresponds to barrierless chiral-edge growth. (b) The atomic configurations after first dimer addition, N=2. (c) Linear density of different site types on CNT edges as a function of chiral angle. (d) The resulting CNT growth rate as a function of chiral angle for several diameters (inset shows the effect of thermal kinks).
We now have all the ingredients to calculate the relative abundance of different CNT types from atomistic computations. The distributions for the two CNT data sets in Fig. 4a both show a strong predominance of (n,n–1) near-armchair CNTs. The selectivity of distributions is actually so strong that one has to increase the temperature to T=2700 K when plotting these distributions; at T=900 K, both show effectively single peaks for (6,5) or (9,8) CNTs. Further, one can use either data set to compute a general (n,m) distribution through interface energy fitting. An example is shown in Fig. 4b. The peak in diameter distribution results from the competition between the interface energy, favoring smaller d in equation (1), and a prefactor due to the configurational entropy of CNT caps37,43, favoring larger diameters2, which scales approximately44 as ~d8 (details in Supplementary Note 4). In a real CVD experiment, there will be additional constraints on d from the size of catalyst particles. Then the product distribution will be a slice of Fig. 4b with prominent (n,n–1) peaks, such as those shown in Fig. 4a.
(a) Distributions calculated directly based on atomistic computations for two CNT sets (d≈0.8 nm and 1.2 nm, each distribution is normalized separately). The empty bars show chirality distributions for liquid-catalyst case. (b) Full (n,m) distribution based on an analytical fit to MD interface energies. For all plots, the temperature was artificially set to about three times the typical experimental value to make the heights visible.
Discussion
By similar logic, our theory suggests a possibility to highly selectively achieve the near-Z (n,1) CNTs, if a catalyst favors Z interface over A. While (n,n–1) are always semiconducting, the (n,1) series contains all three CNT families (metallic and two semiconducting). Then, control of diameter would allow a selective synthesis of CNTs of either conductivity type. Moreover, when speculating on a possibility of catalyst–template exactly matching a certain (n,m) tube, we learn here that this would more likely favour the one-index-off tubes such as (n,m±1), to allow for rapid kinetics at the cost of somewhat higher energy of the contact and nucleation. If however, the tight contact is dominant—γ′ in equation (1) is large—the previously neglected  term of equation (2) permits a non-zero growth rate even for perfectly matching tube–catalyst combinations (x=0), for example, armchair on flat substrate. This way catalyst matching can offer direct chirality control, though perhaps at a significantly reduced rate/yield.
term of equation (2) permits a non-zero growth rate even for perfectly matching tube–catalyst combinations (x=0), for example, armchair on flat substrate. This way catalyst matching can offer direct chirality control, though perhaps at a significantly reduced rate/yield.
We can also compare the simulated distributions with a liquid-catalyst model (dash-dotted line in Fig. 2a and the kink formation energy5 EA=0). The results are shown in Fig. 4a as hollow bars and display much greater presence of armchair CNTs in the overall broader distribution. In reality, irregular and highly mobile structure of a liquid catalyst may flatten the energy landscape in Fig. 2a and thus further broaden the distribution, making the kinetic route of selection dominant. If EA>0, the fastest-growing tubes have χ=19.1° (Fig. 3d), which corresponds to (2m,m) CNTs. Finally, with χ-unbiased nucleation probability and EA→0, one recovers the proportionality result5, A∝χ. We also note that additional flexibility could potentially be gained by performing the nucleation and steady-state growth stages under different conditions such as catalyst state, for example, nucleation on solid followed by growth on liquid should favour achiral types.
In summary, we discovered that the origins of chirality can only be understood if the kinetic and thermodynamic aspects of CNT growth are considered concurrently. The growth kinetics is aided by the kinks at the tube edge and thus favours the chiral types, in proportion to their chiral angle. The thermodynamic nucleation barrier, on a solid catalyst, is lower for the kinkless edges of achiral tubes. In spite of complex and random variability of numerous atomic structures in the process, the overall product abundance can be summed up in a remarkably compact mathematical expression: xe−β·x. For lower temperatures and a solid catalyst, this function has a sharp maximum near zero, which explains the observations of near-armchair nanotubes in experiments. Higher T and a liquid catalyst make contact energies effectively equal (β→0) and nucleation of various types similarly probable, with the abundance then nearly proportional to chiral angle. This demonstrates that the approach is sufficiently comprehensive, being able to explain rather disparate facts accumulated over decades of experiments, from broader chiral distributions to very narrow, almost single-type peaks. Furthermore, we believe that the newly gained insight may enable finding ways to engineer chiral-selective nanotube production, thus advancing a variety of long-awaited applications, all pending availability of properly pure material.
Note added in proof: While this paper was under consideration a study was published showing extremely strong preference for a specific CNT type that its authors attribute to catalyst-matching45. In terms of equation (1) this translates into a large magnitude of γ′. The authors of an even more recent study46 were able to achieve single-type selectivity via seeded growth (effectively, Nn,m=δn,6 δm,6). In both cases, according to equations (1) and (2), the smallness of the factor which provides for the growth of catalyst-matching CNTs should result in slow growth rates/low yield.
Methods
MD simulations
MD sampling was performed using the canonical (NVT) ensemble with the ReaxFF force field47,48 as implemented in the LAMMPS49,50 simulation package. The basic structural properties and energetics of graphene and Ni bulk and low-index surfaces using this setup were validated in earlier work51. The catalyst/support is represented by a four-layer Ni(111) slab of approximately square shape and area of 90 surface unit cells (~2.3 × 2.3 nm2). In all runs, the atomic motion of the bottom Ni layer is constrained to the (111) plane. Initially, a 100-ps NPT trajectory is generated, employing a Nosé–Hoover thermostat and barostat to maintain temperature T=900 K (damping parameter τT=0.1 ps; constrained bottom-layer degrees of freedom excluded from T calculation/control), and lateral pressure tensor components P||=0 (τP=1 ps), preserving the lateral simulation box shape. The average box sizes determined from this trajectory are used in all subsequent canonical (NVT) trajectories at the same temperature. All calculations use 0.25 fs timestep and ensemble averaging is obtained from 1.2 ns trajectories.
DFT calculations
The DFT calculations were performed with the local spin density approximation using the QUANTUM ESPRESSO package52. Vanderbilt ultrasoft pseudopotentials53 were used with a plane-wave basis set cutoff of 50 Ry. The model systems consisted of a (6 × 6) Ni(111) (or Co(111)) slab supercell with three atomic layers of Ni (or Co). The positions of the bottom atomic layer were frozen during geometry optimization. The CNTs were represented by fragments two hexagons tall (60–72 C atoms) with the top edge passivated by hydrogen atoms.
Correction to the ReaxFF-calculated interface energies
In our previous research, we found that classical potentials including the ReaxFF parameter set employed herein yield the ratio of Z- to A-edge energies in vacuum different from the DFT result38,54. Since using ab initio MD to compute all quantities necessary for this study would be orders of magnitude beyond the available computational power, we instead utilized a ‘DFT correction’ scheme for ReaxFF. First, we computed the interface energies for the small-diameter CNT set with DFT. The relaxed structures were then used as input for ReaxFF optimization. Then we used ReaxFF to search for the global-minimum adsorbed configuration by taking a CNT fragment and sampling the rotations about the vertical axis in the 0–180° interval in steps of 5°. For each initial geometry, a low-temperature annealing is performed for 5 ps. The lowest-energy configurations were then again optimized with DFT. Thus, we obtain two data points for the DFT–ReaxFF energy difference for each CNT. This set was fitted with a πd(A−Bχ) model, with A and B fitting parameters, and the resulting fit used to ‘reconstruct’ the interface energies from ReaxFF simulations. The mean and median residuals of the fit were only 0.1 eV Å−1 and 0.06 eV Å−1, respectively. It should be stressed that these differences correspond not to the same instantaneous configurations, but rather, to the closest local minima between the methods.
Additional information
How to cite this article: Artyukhov, V. I. et al. Why nanotubes grow chiral. Nat. Commun. 5:4892 doi: 10.1038/ncomms5892 (2014).
Change history
03 October 2014
The HTML version of this Article previously published contained an error in the punctuation of the author name Vasilii I. Artyukhov, which was incorrectly given as Vasilii I/Artyukhov. This has now been corrected in the HTML; the PDF version of the paper was correct from the time of publication.
References
Iijima, S. Helical microtubules of graphitic carbon. Nature 354, 56–58 (1991).
Dresselhaus, M. S., Dresselhaus, G. & Eklund, P. C. Science of Fullerenes and Carbon Nanotubes: Their Properties and Applications Academic Press (1996).
Avouris, P., Chen, Z. & Perebeinos, V. Carbon-based electronics. Nat. Nanotech. 2, 605–615 (2007).
De Volder, M. F. L., Tawfick, S. H., Baughman, R. H. & Hart, A. J. Carbon Nanotubes: present and future commercial applications. Science 339, 535–539 (2013).
Ding, F., Harutyunyan, A. R. & Yakobson, B. I. Dislocation theory of chirality-controlled nanotube growth. Proc. Natl Acad. Sci. USA 106, 2506–2509 (2009).
Thess, A. et al. Crystalline ropes of metallic carbon nanotubes. Science 273, 483–487 (1996).
Fouquet, M. et al. Highly chiral-selective growth of single-walled carbon nanotubes with a simple monometallic Co catalyst. Phys. Rev. B 85, 235411 (2012).
Wang, H. et al. Chiral-selective CoSO4/SiO2 catalyst for (9,8) single-walled carbon nanotube growth. ACS Nano 7, 614–626 (2013).
He, M. et al. Chiral-selective growth of single-walled carbon nanotubes on lattice-mismatched epitaxial cobalt nanoparticles. Sci. Rep. 3, 1460 (2013).
Bachilo, S. M. et al. Structure-assigned optical spectra of single-walled carbon nanotubes. Science 298, 2361–2366 (2002).
Bachilo, S. M. et al. Narrow (n,m)-distribution of single-walled carbon nanotubes grown using a solid supported catalyst. J. Am. Chem. Soc. 125, 11186–11187 (2003).
Lolli, G. et al. Tailoring (n,m) structure of single-walled carbon nanotubes by modifying reaction conditions and the nature of the support of CoMo catalysts. J. Phys. Chem. B 110, 2108–2115 (2006).
Li, X. et al. Selective synthesis combined with chemical separation of single-walled carbon nanotubes for chirality selection. J. Am. Chem. Soc. 129, 15770–15771 (2007).
Harutyunyan, A. R. et al. Preferential growth of single-walled carbon nanotubes with metallic conductivity. Science 326, 116–120 (2009).
Chiang, W.-H. & Sankaran, R. M. Linking catalyst composition to chirality distributions of as-grown single-walled carbon nanotubes by tuning NixFe1–x nanoparticles. Nat. Mater. 8, 882–886 (2009).
Liu, B., Ren, W., Li, S., Liu, C. & Cheng, H.-M. High temperature selective growth of single-walled carbon nanotubes with a narrow chirality distribution from a CoPt bimetallic catalyst. Chem. Commun. 48, 2409–2411 (2012).
Zheng, M. et al. Structure-based carbon nanotube sorting by sequence-dependent DNA assembly. Science 302, 1545–1548 (2003).
Tu, X., Manohar, S., Jagota, A. & Zheng, M. DNA sequence motifs for structure-specific recognition and separation of carbon nanotubes. Nature 460, 250–253 (2009).
Scott, L. T. et al. A short, rigid, structurally pure carbon nanotube by stepwise chemical synthesis. J. Am. Chem. Soc. 134, 107–110 (2012).
Mueller, A. & Amsharov, K. Y. Synthesis of precursors for large-diameter hemispherical buckybowls and precursors for short carbon nanotubes. Eur. J. Org. Chem. 2012, 6155–6164 (2012).
Omachi, H., Nakayama, T., Takahashi, E., Segawa, Y. & Itami, K. Initiation of carbon nanotube growth by well-defined carbon nanorings. Nat. Chem. 5, 572–576 (2013).
Ogrin, D. et al. Amplification of single-walled carbon nanotubes from designed seeds: separation of nucleation and growth. J. Phys. Chem. C 111, 17804–17806 (2007).
Orbaek, A. W., Owens, A. C. & Barron, A. R. Increasing the efficiency of single walled carbon nanotube amplification by Fe–Co catalysts through the optimization of CH4/H2 partial pressures. Nano Lett. 11, 2871–2874 (2011).
Liu, J. et al. Chirality-controlled synthesis of single-wall carbon nanotubes using vapour-phase epitaxy. Nat. Commun. 3, 1199 (2012).
Elliott, J. A., Shibuta, Y., Amara, H., Bichara, C. & Neyts, E. C. Atomistic modelling of CVD synthesis of carbon nanotubes and graphene. Nanoscale 5, 6662–6676 (2013).
Shibuta, Y. & Maruyama, S. Molecular dynamics simulation of generation process of SWNTs. Phys. B Condens. Matter 323, 187–189 (2002).
Ding, F., Bolton, K. & Rosén, A. Nucleation and growth of single-walled carbon nanotubes: a molecular dynamics study. J. Phys. Chem. B 108, 17369–17377 (2004).
Zhao, J., Martinez-Limia, A. & Balbuena, P. B. Understanding catalysed growth of single-wall carbon nanotubes. Nanotechnology 16, S575 (2005).
Ribas, M. A., Ding, F., Balbuena, P. B. & Yakobson, B. I. Nanotube nucleation versus carbon-catalyst adhesion–probed by molecular dynamics simulations. J. Chem. Phys. 131, 224501 (2009).
Amara, H., Bichara, C. & Ducastelle, F. Understanding the nucleation mechanisms of carbon nanotubes in catalytic chemical vapor deposition. Phys. Rev. Lett. 100, 056105 (2008).
Page, A. J., Ohta, Y., Irle, S. & Morokuma, K. Mechanisms of single-walled carbon nanotube nucleation, growth, and healing determined using QM/MD methods. Acc. Chem. Res. 43, 1375–1385 (2010).
Raty, J.-Y., Gygi, F. & Galli, G. Growth of carbon nanotubes on metal nanoparticles: a microscopic mechanism from ab initio molecular dynamics simulations. Phys. Rev. Lett. 95, 096103 (2005).
Neyts, E. C., Shibuta, Y., van Duin, A. C. T. & Bogaerts, A. Catalyzed growth of carbon nanotube with definable chirality by hybrid molecular dynamics−force biased Monte Carlo simulations. ACS Nano 4, 6665–6672 (2010).
Neyts, E. C., van Duin, A. C. T. & Bogaerts, A. Changing chirality during single-walled carbon nanotube growth: a reactive molecular dynamics/Monte Carlo study. J. Am. Chem. Soc. 133, 17225–17231 (2011).
Penev, E. S., Artyukhov, V. I., Ding, F. & Yakobson, B. I. Unfolding the fullerene: nanotubes, graphene and poly-elemental varieties by simulations. Adv. Mater. 24, 4956–4976 (2012).
Yuan, Q., Xu, Z., Yakobson, B. I. & Ding, F. Efficient defect healing in catalytic carbon nanotube growth. Phys. Rev. Lett. 108, 245505 (2012).
Penev, E. S., Artyukhov, V. I. & Yakobson, B. I. Extensive energy landscape sampling of nanotube end-caps reveals no chiral-angle bias for their nucleation. ACS Nano 8, 1899–1906 (2014).
Liu, Y., Dobrinsky, A. & Yakobson, B. I. Graphene edge from armchair to zigzag: the origins of nanotube chirality? Phys. Rev. Lett. 105, 235502 (2010).
Artyukhov, V. I., Liu, Y. & Yakobson, B. I. Equilibrium at the edge and atomistic mechanisms of graphene growth. Proc. Natl Acad. Sci. USA 109, 15136–15140 (2012).
Rao, R., Liptak, D., Cherukuri, T., Yakobson, B. I. & Maruyama, B. In situ evidence for chirality-dependent growth rates of individual carbon nanotubes. Nat. Mater. 11, 213–216 (2012).
Kudin, K. N., Scuseria, G. E. & Yakobson, B. I. C2F, BN, and C nanoshell elasticity from ab initio computations. Phys. Rev. B 64, 235406 (2001).
Klein, D. J. Graphitic polymer strips with edge states. Chem. Phys. Lett. 217, 261–265 (1994).
Brinkmann, G., Fowler, P. W., Manolopoulos, D. E. & Palser, A. H. R. A census of nanotube caps. Chem. Phys. Lett. 315, 335–347 (1999).
Reich, S., Li, L. & Robertson, J. Structure and formation energy of carbon nanotube caps. Phys. Rev. B 72, 165423 (2005).
Yang, F. et al. Chirality-specific growth of single-walled carbon nanotubes on solid alloy catalysts. Nature 510, 522–524 (2014).
Sanchez-Valencia, J. R. et al. Controlled synthesis of single-chirality carbon nanotubes. Nature 512, 61–64 (2014).
Van Duin, A. C. T., Dasgupta, S., Lorant, F. & Goddard, W. A. ReaxFF: a reactive force field for hydrocarbons. J. Phys. Chem. A 105, 9396–9409 (2001).
Mueller, J. E., van Duin, A. C. T. & Goddard, W. A. Development and validation of ReaxFF reactive force field for hydrocarbon chemistry catalyzed by nickel. J. Phys. Chem. C 114, 4939–4949 (2010).
Plimpton, S. Fast parallel algorithms for short-range molecular dynamics. J. Comput. Phys. 117, 1–19 (1995).
Aktulga, H. M., Fogarty, J. C., Pandit, S. A. & Grama, A. Y. Parallel reactive molecular dynamics: numerical methods and algorithmic techniques. Parallel Comput. 38, 245–259 (2012).
Pigos, E. et al. Carbon nanotube nucleation driven by catalyst morphology dynamics. ACS Nano 5, 10096–10101 (2011).
Giannozzi, P. et al. QUANTUM ESPRESSO: a modular and open-source software project for quantum simulations of materials. J. Phys. Condens. Matter 21, 395502 (2009).
Vanderbilt, D. Soft self-consistent pseudopotentials in a generalized eigenvalue formalism. Phys. Rev. B 41, 7892–7895 (1990).
Kim, K. et al. Ripping graphene: preferred directions. Nano Lett. 12, 293–297 (2011).
Acknowledgements
We thank R.B. Weisman and R.H. Hauge for their comments on the manuscript, and acknowledge the inspiring environment during the VI Guadalupe Workshop on Nanotube Growth (April 2013 in Bandera, Texas). The work was supported by the Air Force Office of Scientific Research (FA9550-14-1-0107), by the Office of Naval Research (N00014-11-1077) and by the National Science Foundation (CBET-0731246). Computer resources were provided by National Energy Research Scientific Computing Center, which is supported by the Office of Science of the U.S. Department of Energy under Contract No. DE-AC02-05CH11231; XSEDE, which is supported by NSF grant OCI-1053575, under allocation TG-DMR100029; and the DAVinCI cluster acquired with funds from NSF grant OCI-0959097.
Author information
Authors and Affiliations
Contributions
B.I.Y. suggested the xe–x-hypothesis and the project direction. All authors designed the research. V.I.A. and E.S.P. performed the calculations. All authors analysed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-3, Supplementary Notes 1-4 and Supplementary References (PDF 5111 kb)
Rights and permissions
About this article
Cite this article
Artyukhov, V., Penev, E. & Yakobson, B. Why nanotubes grow chiral. Nat Commun 5, 4892 (2014). https://doi.org/10.1038/ncomms5892
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms5892
This article is cited by
-
Molecular dynamics simulation of carbon nanotube growth under a tensile strain
Scientific Reports (2024)
-
Bulk growth and separation of single-walled carbon nanotubes from rhenium catalyst
Nano Research (2022)
-
Synthetic control over the binding configuration of luminescent sp3-defects in single-walled carbon nanotubes
Nature Communications (2021)
-
Multidimensional synthetic chiral-tube lattices via nonlinear frequency conversion
Light: Science & Applications (2020)
-
Carbon nanotube: Controlled synthesis determines its future
Science China Materials (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.