Abstract
Thermodynamics is traditionally concerned with systems comprised of a large number of particles. Here we present a framework for extending thermodynamics to individual quantum systems, including explicitly a thermal bath and work-storage device (essentially a ‘weight’ that can be raised or lowered). We prove that the second law of thermodynamics holds in our framework, and gives a simple protocol to extract the optimal amount of work from the system, equal to its change in free energy. Our results apply to any quantum system in an arbitrary initial state, in particular including non-equilibrium situations. The optimal protocol is essentially reversible, similar to classical Carnot cycles, and indeed, we show that it can be used to construct a quantum Carnot engine.
Similar content being viewed by others
Introduction
Thermodynamics forms part of the bedrock of our current understanding of the physical world. It has remained unchanged despite huge revolutions in physics, such as relativity and quantum theory, and few believe it will ever fail. Over time, it has been applied to situations well outside its original domain; from black holes1,2 to quantum engines comprised of only a few qubits3,4,5,6,7. Drawing inspiration, in part, from the resource theory paradigm in quantum information8,9,10,11,12,13, recently, there has been much renewed interest in the foundations of thermodynamics, with a number of very interesting results already obtained14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36. One of the overarching fundamental questions that these works are concerned with is of the applicability of thermodynamics to quantum systems; it is this question that we wish to address in this paper.
Thermodynamics was originally invented to deal with macroscopic thermal machines such as steam engines, long before microscopic particles, let alone the theory of quantum mechanics, were discovered. It is therefore plausible that significant differences exist in the quantum regime. Indeed, recent results call into question the role of free energy for individual quantum systems14. Classical thermodynamics tells us that the total amount of work we are able to extract from a system is given by its change in free energy, which was also supported by previous quantum results18,19,22,23,24,25,26,27,28,29,30. Yet in ref. 14, an alternative paradigm was presented in which it was shown that work equal to free energy can be extracted only if we collectively process many copies of the same system. When acting on each copy individually, the amount of work that can be extracted is generally significantly less than the free energy. Moreover, even more recent results show that considering catalysts16 further change the story. These results therefore suggest that the free energy is not the relevant quantity for individual systems.
Here we revisit the issue of work extraction and show that free energy is a significant quantity for individual systems. To do so, we present a paradigm for dealing with thermodynamic processes within quantum theory. Our paradigm is similar to that of ref. 14 but differs in two essential aspects. In ref. 14, they considered almost deterministic work extraction, from the ‘single-shot’ viewpoint that has received much attention lately16,25,31,32,33,34. Here, in contrast, we will consider average work extraction, and only require average energy conservation. In this context, we first prove the second law of thermodynamics holds, and second give a simple protocol that extracts work equal to the free energy change of an individual quantum system and show that this is optimal. We furthermore show that this protocol can be used to construct a quantum Carnot engine similar to the one in ref. 35, from which our optimality results imply the Carnot limit, an alternative formulation of the second law of thermodynamics. An alternative approach that also allows one to extract average work equal to the free energy change of a system was very recently proposed in ref. 36, where a key difference is that a reusable source of coherence is included in the framework.
Results
The paradigm
In this section, we more precisely describe our framework for quantum thermodynamics. In particular, we define the system, thermal bath and work-storage device, and give explicit definitions for thermodynamic quantities such as heat, work, free energy and entropy within our framework. In light of this, we consider the allowed transformations, and impose the first law of thermodynamics.
We consider any quantum system (of finite dimension) in an arbitrary initial state ρS and with arbitrary Hamiltonian HS. In accordance with statistical mechanics, we define the system’s internal energy as U=tr(ρSHS) (that is, its average energy), and its entropy as the von Neumann entropy S=−tr(ρS log ρS). Note that the system itself need not have a well-defined temperature, however, its free energy relative to a thermal bath at temperature T is given by F=U−TS.
To represent a thermal bath at temperature T, we assume that we have an unlimited supply of finite dimensional systems, each with any desired Hamiltonian HB, in the corresponding thermal state  , where
, where  is the partition function, and we set kB=1 throughout for convenience. When one has access to a thermal bath, any system in a thermal state is essentially a ‘free resource’15. Note that any physical protocol must involve a finite number of systems from the bath, which can be thought of as a single large thermal system. We define the heat flow Q out of the bath as the decrease in its average energy, that is, if the bath system is transformed into state σB, then Q=tr(HB(τB−σB)).
is the partition function, and we set kB=1 throughout for convenience. When one has access to a thermal bath, any system in a thermal state is essentially a ‘free resource’15. Note that any physical protocol must involve a finite number of systems from the bath, which can be thought of as a single large thermal system. We define the heat flow Q out of the bath as the decrease in its average energy, that is, if the bath system is transformed into state σB, then Q=tr(HB(τB−σB)).
In this work, we wish to explicitly include the physical device that stores the work we extract. The work-storage device we consider here is a suspended weight, which is raised or lowered when work is done on or by it. In particular, we consider a quantum system whose height is given by the position operator  , with Hamiltonian
, with Hamiltonian  representing its gravitational potential energy. For simplicity, we choose mg=1 J m−1, such that the value of
representing its gravitational potential energy. For simplicity, we choose mg=1 J m−1, such that the value of  directly denotes the work stored by the mass. Such a system has a long history of being used as a work-storage system in classical thermodynamics37. We define the work W extracted as the change in the average energy of the weight. Hence, if the weight is initially in the state ρW and is left in the state σW, then W=tr(HW(σW−ρW)). We do not place any constraints on the initial state of the weight, unlike in ref. 36. In fact, as we will see below, by construction, the explicit choice of initial state will play no role in this work.
directly denotes the work stored by the mass. Such a system has a long history of being used as a work-storage system in classical thermodynamics37. We define the work W extracted as the change in the average energy of the weight. Hence, if the weight is initially in the state ρW and is left in the state σW, then W=tr(HW(σW−ρW)). We do not place any constraints on the initial state of the weight, unlike in ref. 36. In fact, as we will see below, by construction, the explicit choice of initial state will play no role in this work.
It will be helpful to define the translation operator Γa, which acts on (un-normalized) position states of the weight as Γa|x›=|x+a›.
In previous work14, an alternative work-storage system was suggested—raising a qubit deterministically from its ground state to its excited state. This qubit was termed a wit, short for work bit. However, choosing the energy gap of the work bit requires advance knowledge of the work to be extracted, and so this model does not translate well to non-deterministic work extraction, which we will be interested in here. Furthermore, we would prefer to use a single work-storage system as a ‘battery’ capable of gaining and expending work in multiple thermodynamic processes.
We assume that the initial state is a product state of the system, bath and weight. We now consider the allowed transformations in our framework. The intention here is to remain as general as possible, while eliminating the possibility of ‘cheating’ by bringing in resources from outside the framework (such as external sources of work or free energy), or making use of objects within the framework for a purpose other than intended (for example, by using the work-storage device as a cold reservoir in a heat engine). Our first two assumptions are very general: the first is unitarity. The most general quantum transformation is a completely positive trace-preserving map. However, here we consider only unitary transformations of the system, bath and weight. This prevents us from using external ancillas in non-thermal states as a source of free energy. The second assumption is average energy conservation (the first law). We require that any particular protocol conserves the total average energy (for the particular initial state of the system and bath on which it is designed to operate, and on any initial state of the weight). In terms of the quantities defined earlier, this corresponds to the first law of thermodynamics, which with our chosen sign convention can be expressed as

This prevents us from using the transformation itself as a source of work (for example, by simply raising the weight). Note that this assumption differs from that made in previous works8,15 that the unitary evolution commutes with the total energy operator. We will comment more on this in the Discussion.
We also place two additional constraints on the allowed dynamics governing interactions with the weight: the first is weight-state independence, that the work extracted in an allowed protocol must be independent of the initial state of the weight. Intuitively, this is because we want the weight to play a ‘passive’ role, such that its sole purpose is to keep account of the extracted work. More importantly, this prevents us from ‘cheating’ by using the weight for purposes other than as a work-storage system (for example, as a cold reservoir or a source of coherence). Furthermore, this ensures that we can use the same work-storage system for multiple thermodynamics protocols (or on several copies of the same state) without having to worry how its initial state has been modified by earlier procedures. We prove that our protocol obeys this assumption in the Methods. The second constraint is weight-translation invariance. We require the allowed unitaries to commute with all translation operators on the weight. This reflects the translational symmetry of the weight system, and the fact that only displacements in its height are important.
The second law
We now show that the second law of thermodynamics holds in our framework, by proving that there is no protocol that extracts a positive quantity of work from a thermal bath while leaving the system unchanged (that is, that there is no way of turning heat into work)38. To show this, we will use proof by contradiction.
Consider a thermal bath at temperature T, an arbitrary quantum system (acting as a working system for the protocol) and a weight.
Let us first consider the energy changes during the protocol. As the final state of the system is the same as its initial state, its average energy cannot change. Suppose that we are able to extract average energy from the bath and store it in the weight, ΔEW>0. The average energy of the thermal bath must change by ΔEB=−ΔEW due to average energy conservation.
Now consider the entropy changes during the same protocol (in particular, the changes in von Neumann entropy S(ρ)=−tr(ρ log ρ)). As the system, bath and weight are initially uncorrelated, their initial entropy is simply the sum of their individual entropies. Unitary transformations conserve the total entropy, ΔStotal=0. However, as correlations can form during the protocol, the sum of the final entropies can be greater than the sum of their initial entropies (as the entropy is subadditive). This means that

As the final state of the system must be the same as its initial state ΔSS=0. Furthermore, given an initial thermal state for the bath (with positive temperature), any change of the state that reduces its average energy must also reduce its entropy (since the thermal state is the maximal entropy state with given average energy), ΔSB<0. However, within our framework, all allowed protocols are such that the work extracted is independent of the initial state of the weight; we are therefore free to choose any initial state of the weight we like. We show in Methods that the entropy change of the weight can be made as small as desired by taking its initial state to be a very broad wavepacket (with well-defined momentum). In particular, we can make ΔSW<|ΔSB|. This would result in violating equation (2). Hence, there is a contradiction, and thus there is no way to extract work from the bath.
The second law places an upper bound on the amount of work that can be extracted from a system. In the following section, we will show that we can come as close as desired for extracting this maximum amount of work, by presenting an explicit protocol.
Extracting work from an individual quantum system
Our protocol for extracting work from a quantum system proceeds in two stages. In the first stage, we transform the state of the system into a mixture of energy eigenstates, without using the thermal bath. In the second stage, we gradually transform the system into a thermal state in a sequence of steps, each of which involves a new qubit from the bath. Each step is essentially an infinitesimal Carnot cycle, similar to the one discussed in ref. 35. In both stages, we extract an amount of work arbitrarily close to the free energy change of the system. It follows from our proof of the second law that this protocol is optimal.
Stage 1 is to transform the system into a mixture of energy eigenstates. In this stage, we transform the system into a mixture of energy eigenstates without using the thermal bath, and extract work equal to its change in free energy. Consider a system and weight, initially in an arbitrary uncorrelated state, represented by the density operator ρS⊗ρW.
We can always expand ρS in terms of its eigenvalues pn and eigenvectors |ψn› as  , where we have ordered the eigenvalues such that pn+1≤pn. Denoting the energy eigenstates of the system by |En› (with corresponding eigenvalues En), we implement the unitary transformation
, where we have ordered the eigenvalues such that pn+1≤pn. Denoting the energy eigenstates of the system by |En› (with corresponding eigenvalues En), we implement the unitary transformation

where ͛n=‹ψn|HS|ψn›−En, such that V always conserves average energy. After the transformation, the final state is

with the reduced states σS=trW(σSW) and σW=trS(σSW). The work extracted is given by  . The change in average energy of the system is
. The change in average energy of the system is  , hence this protocol is in accordance with the first law of thermodynamics (that is, ΔU=−W). Futhermore, the entropy of the system remains unchanged, so the work extracted is precisely equal to the free energy lost by the system
, hence this protocol is in accordance with the first law of thermodynamics (that is, ΔU=−W). Futhermore, the entropy of the system remains unchanged, so the work extracted is precisely equal to the free energy lost by the system

Stage 2 of the protocol consists in extracting work from a mixture of energy eigenstates. In this stage, we show that it is possible to extract work equal to the free energy change when transforming a system between two states that are diagonal in the energy eigenbasis. By transforming the state σS obtained in the first stage into a thermal state, we extract the maximum amount of work from the system.
We begin by considering a small change in the occupation probabilities of two energy levels. In particular, suppose that we wish to transform the state  into a new state
into a new state  , in which q1=p1+δp, q0=p0−δp and qk=pk in all other cases (that is, for all k>1). We consider the situation in which |δp|<<p1≤p0. Note that this excludes the case in which p1=0, which introduces some additional subtleties that are detailed in Methods. Nevertheless, the protocol and conclusions presented below are unchanged.
, in which q1=p1+δp, q0=p0−δp and qk=pk in all other cases (that is, for all k>1). We consider the situation in which |δp|<<p1≤p0. Note that this excludes the case in which p1=0, which introduces some additional subtleties that are detailed in Methods. Nevertheless, the protocol and conclusions presented below are unchanged.
To achieve the above transformation, we take a qubit from the thermal bath with energy eigenstates |0›B and |1›B such that its state has the form

that is, such that the ratio of ground and excited state populations matches that of the corresponding states in  . Note that this fixes the energy spacing EB of the qubit, as
. Note that this fixes the energy spacing EB of the qubit, as  , hence
, hence  . We then apply the unitary transformation that swaps the bath qubit with the state of the system if the system is in the two-dimensional subspace spanned by
. We then apply the unitary transformation that swaps the bath qubit with the state of the system if the system is in the two-dimensional subspace spanned by  and
and  , and translates the weight to conserve the total energy. This transformation maps
, and translates the weight to conserve the total energy. This transformation maps

for all x, where ɛ=EB−(E1−E0), while leaving all other states invariant. This leaves the system in the state  (for more details see Methods). Note that this unitary commutes with the Hamiltonian H=HS+HB+HW, so it will obey the first law of thermodynamics.
(for more details see Methods). Note that this unitary commutes with the Hamiltonian H=HS+HB+HW, so it will obey the first law of thermodynamics.
As the weight is only shifted up or down by ɛ when the system and bath are in  or
or  , respectively, the work extracted is given by δW=ɛδp. The change in the free energy of the system is given by δF=δU−TδS. As we show in Methods, it is straightforward to see that δU=δp(E1−E0), and that the change in the entropy of the system is given by
, respectively, the work extracted is given by δW=ɛδp. The change in the free energy of the system is given by δF=δU−TδS. As we show in Methods, it is straightforward to see that δU=δp(E1−E0), and that the change in the entropy of the system is given by  . Hence to first order in δp, δF≈δp(E1−E0−EB)=−δpɛ=−W. This shows that we extract work equal to the reduction in free energy of the system, up to a deficit of (δp2).
. Hence to first order in δp, δF≈δp(E1−E0−EB)=−δpɛ=−W. This shows that we extract work equal to the reduction in free energy of the system, up to a deficit of (δp2).
To extract the maximal amount of work from a quantum state, we perform a sequence of N steps like the one above, interacting the system with a new thermal qubit in each step, until the system has been transformed into a thermal state at temperature T. In particular, for sufficiently large N, we can choose a sequence of N+1 states for the system in which subsequent states only differ by a transfer of probability  between a pair of energy levels, with the first and last states equal to the initial state of the system and its thermal state, respectively (for example, starting at the highest energy level, we could first shift probability from all energy levels with higher probability in σS than in τS to the
between a pair of energy levels, with the first and last states equal to the initial state of the system and its thermal state, respectively (for example, starting at the highest energy level, we could first shift probability from all energy levels with higher probability in σS than in τS to the  state, then move probability from
state, then move probability from  to the remaining levels, using N/(d−1) steps for each pair of levels). Applying a unitary of the form (7) in each step, the work extracted from this stage of the protocol will be
to the remaining levels, using N/(d−1) steps for each pair of levels). Applying a unitary of the form (7) in each step, the work extracted from this stage of the protocol will be

In the limit N→∞, the work extracted will equal the free energy change of the system, regardless of the precise choice of path. Note that in the limit of large N, the state of each thermal qubit is only changed slightly by the protocol.
Next, we move onto the question of reversibility and optimality of the protocol. By combining both stages of the protocol, and using a sufficiently large number of thermal qubits, it is clear that we can transform an arbitrary state ρS into a thermal state τS and extract an amount of work as close as we like to the free energy change of the system. The limiting amount of work we can achieve is therefore Wmax=F(ρS)−F(τS).
Interestingly, if ρS is full rank (that is, it has no zero eigenvalues), we can also implement the reverse process to create ρS from an initially thermal system taken from the bath. We can use the stage 2 protocol to move from τS to σS, and then apply the inverse of the stage 1 transformation. The work cost for this will be W=F(τS)−F(ρS). Note that it is not possible to use our protocol to create a state that is not full rank, as the final step would require the use of a thermal qubit with EB=∞. However, as there are always full-rank states arbitrarily close to every state, this is not a physically significant restriction. In this sense, all transitions between states can be implemented in a thermodynamically reversible way (we note, however, that if a state is thermalised and then recreated using our protocol, the fluctuations in the position of the weight will increase). This differs from the results of refs 14, 25, who show that such transitions are irreversible when considering (almost) deterministic work extraction, rather than average work. Similarly, an arbitrary transformation of the system from a state ρS to  can be achieved (when
can be achieved (when  is full rank) for a work cost as close as desired to the free energy change of the system; one way this could be achieved is to transform the system into a thermal state, and then transform the thermal state into the final state. We now show that our protocol is optimal, using our proof of the second law (see Methods). Suppose that there exists a different protocol that extracts work F(ρS)−Fβ(τS)+δ (where δ>0) when the system is transformed from ρS to τS, we can then use the above protocol to return the state from τS to ρS, extracting work F(τS)−Fβ(ρS)−ɛ, where we choose the number of thermal qubits such that ɛ is in the range 0<ɛ<δ/2. The net effect is that a positive amount of work ⩾δ/2 is extracted, and the system begins and ends the combined procedure in the same state ρS, in violation of the second law.
is full rank) for a work cost as close as desired to the free energy change of the system; one way this could be achieved is to transform the system into a thermal state, and then transform the thermal state into the final state. We now show that our protocol is optimal, using our proof of the second law (see Methods). Suppose that there exists a different protocol that extracts work F(ρS)−Fβ(τS)+δ (where δ>0) when the system is transformed from ρS to τS, we can then use the above protocol to return the state from τS to ρS, extracting work F(τS)−Fβ(ρS)−ɛ, where we choose the number of thermal qubits such that ɛ is in the range 0<ɛ<δ/2. The net effect is that a positive amount of work ⩾δ/2 is extracted, and the system begins and ends the combined procedure in the same state ρS, in violation of the second law.
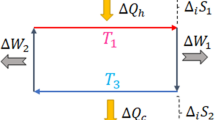
A quantum Carnot engine
In previous work35, a quantum Carnot engine was described, and an argument was made that essentially all Carnot engines are the same. Indeed, each infinitesimal step of stage 2 of our protocol is essentially the action of such a Carnot engine (although the situation is more complicated here, as the engine has to adapt between steps). Moreover, the frameworks are very different—Hamiltonian versus unitary, master equations versus extracting qubits from the bath and so on. It is therefore essential to verify that in our present framework we can implement a full Carnot engine. In this section, we show that this is indeed the case.
We must now consider two thermal baths, a hot bath with temperature TH and a cold bath with temperature TC<TH. As before, we also have a quantum system (used as a working system that links the two baths) and a weight. Imagine that the system is initially in the thermal state relative to the cold bath (with internal energy UC and entropy SC). Our Carnot cycle is as follows: first bring the system in contact with the hot bath and use the protocol given in the previous subsection to transform it into the thermal state at temperature TH (with internal energy UH and entropy SH). In the asymptotic limit, this allows us to extract work equal to the free energy change of the system with respect to the hot bath. Second, move the system back in contact with the cold bath and use the same protocol to transform it back into the thermal state at temperature TC, extracting work now equal to the free energy change with respect to the cold bath. In Methods, we show that the total work extracted in both steps is W=(TH−TC)(SH−SC). Furthermore, by applying the first law of thermodynamics to the first step, it follows that QH=TH(SH−SC). Therefore, combining these two results yields

which is precisely the Carnot efficiency. By running this process backwards, we can also construct the corresponding heat pump.
As in standard thermodynamics, the second law prevents us from constructing any heat engine more efficient than the one above. If such an engine was possible, we could subsequently run our Carnot engine as a heat pump such that the net heat flow into the cold bath was zero. In this case, work would be extracted and the hot bath would decrease in energy by a finite amount. The entropy of the hot bath must have decreased as a result, and the entropy of the cold bath can also only have decreased (as its average energy is unchanged and it was originally in a thermal state). As before, the entropy increase in the weight can be made as small as you like by choosing an appropriate initial state. This creates a contradiction with the total entropy conservation expressed in equation (2).
Discussion
In summary, in this paper, we presented a framework for extending thermodynamics to individual quantum systems. Within this framework, we proved that the second law of thermodynamics holds and gave an explicit protocol to extract the maximum amount of work from an arbitrary individual quantum system in conjunction with a thermal bath. This work is equal to the change in free energy of the system. Our results apply to any quantum system in an arbitrary initial state, in particular including non-equilibrium situations. The optimal protocol is essentially reversible, similar to classical Carnot cycles, and indeed, we can use it to construct a quantum Carnot engine.
A key element of our framework is to associate classical energetic quantities (internal energy, heat and work) with average quantum quantities. However, we want to emphasize that, although we use average quantities, we do not require an ensemble of multiple quantum systems to be processed collectively; our protocols act on an individual quantum system.
A significant difference between our framework and other approaches14,15,16,17 is that the allowed transformations need to only satisfy average energy conservation rather than the stronger requirement of strict energy conservation (that is, unitaries that commute with the total Hamiltonian). The fundamental reason for allowing such transformations is that average energy conservation corresponds precisely to the first law of thermodynamics in our framework (as we defined all energetic quantities in terms of averages). Demanding strict energy conservation is more than what the first law (in our framework) requires.
Allowing protocols that only conserve average energy has major consequences. In particular, when the initial state of the system contains coherences between energy levels, protocols satisfying strict energy conservation cannot extract work equal to the full change in free energy (see Methods). The work deficit equals the difference in free energy between the true state and its energy-decohered version. That is, such protocols simply cannot make any use of the free energy associated with coherences between energy levels. In our protocol, this free energy is extracted in stage 1, which is the only part of the protocol that does not satisfy strict energy conservation.
It is interesting to note a further subtle difference between average energy-conserving unitaries and strict energy-conserving ones. To be optimal, both must be ‘state dependent’, that is, they have to be designed with a particular initial state of the system in mind. However, if we use a unitary designed for a particular system state on a different initial state, in the case of strict energy conservation, the external machinery that implements the unitary continues to remain ‘neutral’, that is, it doesn’t change the energy of the system–bath–weight complex, while in the average energy-conserving case the external machinery may exchange energy with the complex.
We also note that unlike in classical thermodynamics, there will also be fluctuations, for example, in the final position of the weight. Analysing these fluctuations is an interesting issue for future study.
A subtle aspect that we want to mention is that as our protocols involve a sequence of changing unitaries, we have implicitly assumed the existence of an external clock by which to control the protocol. This raises some interesting points—the first is whether the clock is a resource that costs work to establish and maintain (in which case we may have over estimated the amount of work we can extract). Second is to extend the framework to explicitly incorporate the clock, with protocols being implemented via a global time-independent Hamiltonian. Finally, in the light of the difference between strictly energy-conserving unitaries and average energy-conserving unitaries, it is important to investigate whether or not there is any essential difference in the use they make of the energy coherence in the clocks. These are very interesting areas for future work.
Recently, there has been considerable progress in studying and understanding the foundations of statistical mechanics, see for example refs 39, 40, 41, 42, 43, 44. It would be extremely important to connect the present results on quantum thermodynamics to that line of research.
To conclude, the resource theory framework seems to be a natural and powerful way to approach thermodynamics. It has already delivered significant results and we believe that further investigation along these lines will lead to a much deeper understanding of the foundations of thermodynamics.
Methods
Independence of the work on the initial state of the weight
Each step of our protocol can be represented by a unitary transformation of the form

where  , the states |i› and
, the states |i› and  form different orthonormal bases for the system and the relevant portion of the bath, and Γa is the translation operator on the weight, given by
form different orthonormal bases for the system and the relevant portion of the bath, and Γa is the translation operator on the weight, given by  , where
, where  is the usual momentum operator satisfying
is the usual momentum operator satisfying  .
.
It is easy to see that all such unitaries commute with translations on the weight. We now show that the work extracted by a unitary of this form does not depend on the initial state of the weight (even if it is initially correlated with the state of the system). Let us denote the initial state of the system, bath and weight by the density operator ρ. The work extracted is given by

where  is the Hamiltonian of the weight (defined for convenience as an operator on the entire system). Now note that
is the Hamiltonian of the weight (defined for convenience as an operator on the entire system). Now note that

Inserting this expression in equation (11) and simplifying, we obtain

where ρSB=trW(ρ) is the reduced density matrix of the system and bath. Hence, the amount of work extracted is independent of the initial state of the weight as desired.
Proof of second law of thermodynamics
In this section, we provide further details for our proof of the second law.
We first argue that any reduction in the average energy of an initially thermal state (with positive temperature) must also yield a reduction in its entropy. We only need to use the fact that a thermal state is the maximal entropy state with a given average energy, which is well known and can easily be proven using the method of Lagrange multipliers to maximize the entropy subject to the constraints that the average energy is constant and the state is normalized. If a system starts in a thermal state and is transformed to a final state with fixed average energy, the entropy change ΔSB will be maximized when the final state is also thermal. In the case where the average energy decreases, and the initial state has positive temperature, the final state will be a thermal state of lower temperature, and thus lower entropy.
We now show that the entropy change of the weight in any particular protocol may be made as small as desired by choosing its initial state to be a very narrow wavepacket in momentum space (corresponding to a very broad wavepacket in real space).
As any allowed unitary transformation must be invariant under translations of the weight, it can always be written as

where the first element of the tensor product corresponds to a unitary operation on the combined system and bath (as a function of the weight momentum) and the second corresponds to a projector onto the un-normalized momentum eigenstate |p› of the weight. We choose the basis states |j› to be the eigenbasis of the initial system and bath state, so  .
.
For V to be well defined, there must exist a momentum p0 at which ɛij(p) is a continuous function of p for all i, j. We define new functions ηij(p)≡ɛij(p)−ɛij(p0) corresponding to the small variations in ɛij(p) about p0.
For any ɛ>0, we can construct an initial pure state of the weight |φ›, which has support on a sufficiently narrow interval in momentum space (centred on p0), such that |ηij(p)|≤ɛ for all i, j whenever φ(p)≡‹p|φ›≠0.
The final state of the weight is given by

where  is the un-normalized state (with norm≤ɛ):
is the un-normalized state (with norm≤ɛ):

The distance between the initial and final states of the weight in terms of the trace norm, defined as  , is
, is

where d is the combined dimension of the system and bath (note that this only includes the finite number of systems from the bath used in the protocol).
As the final state of the weight lives in a finite dimensional subspace, its entropy can be shown to be continuous due to Fannes’ inequality45.

where  is the trace distance between the initial and final states of the weight. We can make D as small as we like by choosing sufficiently small ɛ and therefore make the entropy change as small as we like.
is the trace distance between the initial and final states of the weight. We can make D as small as we like by choosing sufficiently small ɛ and therefore make the entropy change as small as we like.
Work extraction details
In this section, we provide further details regarding stage 2 of our protocol.
We begin by showing that the final state of the system is  after applying the protocol, where
after applying the protocol, where

is such that q1=p1+δp, q0=p0−δp and qk=pk in all other cases (that is, for all k>1). At the beginning of stage 2 of the protocol, the combined state of the system, bath and weight can be written as

where  . After applying the unitary V that is given by equation (7), and can also be expressed in terms of the translation operator Γa as
. After applying the unitary V that is given by equation (7), and can also be expressed in terms of the translation operator Γa as
V=⊗⊗+|E1›‹E0|⊗|0›‹1|⊗Γɛ+|E0›‹E1|⊗|1›‹0|⊗Γ−ɛ−|E0›‹E0|⊗|1›‹1|⊗−|E1›‹E1|⊗|0›‹0|⊗,
we find

In later steps of the protocol, the state can be written in a similar form to equation (20) (up to re-labellings), with each state  being a mixture of translated versions of ρW.
being a mixture of translated versions of ρW.
As the weight is only shifted up or down by ɛ when the system and bath are in  or
or  respectively, the work extracted is given by
respectively, the work extracted is given by

It is straightforward to see that

The change in the entropy of the system is given by

where in the last step we have used the fact that log(1+x)=x−(x2) for |x|<1. Hence, to first order in δp

This shows that we extract work equal to the reduction in free energy of the system, up to a deficit of (δp2).
Next, we consider increasing the occupation probability of an energy state, which initially has probability zero. This situation would arise if we were trying to extract work from an initial pure state, as after stage 1 of our protocol the state would be |E0›‹E0|.
Let us consider the case in which we increase p1 from 0 to r in N steps, while p0 is decreased from s to s−r. After k steps, the occupation probabilities for the system will be given by


where  . From equations (22) and (23), it follows respectively that the change in the internal energy of the system during the kth step will be
. From equations (22) and (23), it follows respectively that the change in the internal energy of the system during the kth step will be

and the work extracted will be

where  is the energy gap of the bath qubit used in the kth step. We recall also that since we are considering thermal states, this energy gap satisfies the relation
is the energy gap of the bath qubit used in the kth step. We recall also that since we are considering thermal states, this energy gap satisfies the relation

It remains to calculate the entropy change of the system in the kth step,

When k=0, this is equal to


When k>0, it is given by equation 31

By expanding the logarithm as a power series in  , we find
, we find

As this is an alternating sequence with terms of decreasing magnitude, it follows that

and hence

To obtain the total discrepancy between the work extracted and the free energy loss, we must sum over all steps kε{0,1,2,...,N−1}, obtaining

where in the second line we have used the fact that

It follows from equation (37) that as N→∞ the work extracted by the protocol approaches the free energy loss of the system, as desired.
Carnot engine details
Consider two thermal baths, a hot bath with temperature TH and a cold bath with temperature TC<TH. As before, we also have a quantum system (used as a working system that links the two baths) and a weight. Imagine that the system is initially in the thermal state relative to the cold bath (with internal energy UC and entropy SC). Our Carnot cycle is as follows: first bring the system in contact with the hot bath and use the protocol given in Methods to transform it into the thermal state at temperature TH (with internal energy UH and entropy SH). In the asymptotic limit, this allows us to extract work

Second, move the system back in contact with the cold bath and use the same protocol to transform it back into the thermal state at temperature TC, extracting work

The total work extracted in both steps is

Now, by applying the first law of thermodynamics (ΔU=Q−W) to the first step above, we find

where QH is the heat flow out of the hot bath. Substituting this in equation (39), we obtain

Finally, combining this with equation (41) we find

Example of average energy-conserving unitary
Throughout the paper, we treated the unitaries in an abstract way. It is instructive however to give a concrete example of how an average energy-conserving unitary could be implemented. In particular, we consider here stage 1 of our protocol, as this is the only part that satisfies average energy conservation but not strict energy conservation.
Suppose our system is a spin 1/2 particle in a magnetic field of magnitude B polarized along the z direction, the two energy eigenstates are |↑z› and |↓z›, that is, spin polarized ‘up’ or ‘down’ along the z axis, corresponding to the energy eigenvalues E1=−E2=ħω with  where γ is the gyromagnetic factor. Hence HS=ħωσz, where σi for iε{x,y,z} denotes the usual Pauli operators.
where γ is the gyromagnetic factor. Hence HS=ħωσz, where σi for iε{x,y,z} denotes the usual Pauli operators.
Let the state of the system be some arbitrary given density matrix ρS. On diagonalization, ρS can be written as

with  and
and  being the eigenstates of ρS, and p a real number satisfying 0≤p≤1. The states
being the eigenstates of ρS, and p a real number satisfying 0≤p≤1. The states  and
and  are orthogonal to each other (being eigenstates of ρS) and will, in general, be superpositions of energy eigenstates—hence they contain coherences between energy levels. Without any loss of generality, we can take
are orthogonal to each other (being eigenstates of ρS) and will, in general, be superpositions of energy eigenstates—hence they contain coherences between energy levels. Without any loss of generality, we can take  to be the state of the spin polarized in an arbitrary direction in the x–z plane, that is,
to be the state of the spin polarized in an arbitrary direction in the x–z plane, that is,

where θ is the angle it forms with the z axis. The orthogonal state  is therefore
is therefore

The average energy of  is
is  and that of
and that of  is
is  .
.
In this particular case, the unitary for stage 1 of our protocol is given by

where  .
.
One straightforward way to implement this unitary would be to first apply a field that performs the rotation on the spin, and then to perform a conditional shift on the weight given the state of the spin. More concretely, we could first apply  followed by
followed by  . Here, however, although the product V=U2U1 is an interaction that preserves average energy, neither U1 nor U2 does individually. Although all we need is for the overall unitary V to conserve the average energy, one may like a more refined protocol that conserves average energy at all times.
. Here, however, although the product V=U2U1 is an interaction that preserves average energy, neither U1 nor U2 does individually. Although all we need is for the overall unitary V to conserve the average energy, one may like a more refined protocol that conserves average energy at all times.
One can do so by moving to a continuous time picture, thus specifying an interaction Hamiltonian Hint(t) that implements V after time τ, such that if the interaction were switched off at an intermediate time t′, the unitary implemented would still be average energy conserving. Note that this requires us to preserve the expected value of the free Hamiltonian HS+HW at all times, rather than the full Hamiltonian HS+HW+Hint(t). The latter could also be conserved if desired by adding a time-dependent constant to the Hamiltonian. We will take τ to be sufficiently short that we can neglect the free evolution of the weight during the interaction—for larger τ the weight will also pick up some additional phases due to its free evolution, but we will nevertheless extract the same amount of work and perform the same transformation on ρS.
Such an Hint(t) can be constructed by considering the simple example given above, by continuously rotating the spin and conditionally shifting the weight. More precisely, consider the interaction Hamiltonian

where

Note that this interaction Hamiltonian is translationally invariant on the weight, as we would desire in our formalism. If the last term of equation (50) were excluded, it is straightforward to see that the effect of HS+Hint(t) would be to rotate the system spin into the energy eigenbasis of HS in time τ, with the initial eigenstates of ρS tranforming at time t into


These states are instantaneous eigenstates of σ(t), hence the last term in the interaction Hamiltonian does not affect the evolution of ρS.
However, on the weight, this additional term now produces the desired conditional shift, conditioned on the instantaneous eigenstates of σ(t). The rate at which we need to move the weight is given by the rate of change in the average energy of the system,

This thus constitutes a model that will implement the desired evolution while conserving the average energy throughout the interaction time τ. Note that the same evolution would work for any state with the same eigenbasis as ρS.
Limitations of protocols satisfying strict energy conservation
Here we consider protocols satisfying strict energy conservation (that is, unitaries that commute with the total Hamiltonian). We show that such protocols cannot extract work equal to the full change in free energy for systems in initial states having coherences between energy levels, following a similar approach to ref. 14.
First, note that as we require the average work extracted by a protocol to be independent of the initial state of the weight, we are free to choose that state however we like—here we choose it to be a very narrow wavepacket centred on zero. Now consider a decomposition of the total state space into subspaces, each of which has total energy (of the system, bath and weight) close to one of the energy eigenvalues of the system and bath, in particular the ith subspace corresponds to the total energy E lying in the range  , where Ei is the ith energy eigenvalue of the system and bath. Furthermore, we choose the width of the weight’s initial wavepacket to be narrower than the smallest subspace. Note that any work-extraction protocol can be followed by a transformation that decoheres the state with respect to these total energy subspaces, without affecting the average work extracted. However, this decohering operation commutes with the unitaries used in the protocol, so we can move it to the beginning of the protocol without changing the work extracted. At the beginning of the protocol, this operation has the sole effect of decohering the system in its energy eigenbasis (changing ρ to
, where Ei is the ith energy eigenvalue of the system and bath. Furthermore, we choose the width of the weight’s initial wavepacket to be narrower than the smallest subspace. Note that any work-extraction protocol can be followed by a transformation that decoheres the state with respect to these total energy subspaces, without affecting the average work extracted. However, this decohering operation commutes with the unitaries used in the protocol, so we can move it to the beginning of the protocol without changing the work extracted. At the beginning of the protocol, this operation has the sole effect of decohering the system in its energy eigenbasis (changing ρ to  , where
, where  is the projector onto the ith energy subspace). Hence, the protocol extracts the same amount of work as it would have if it had operated on ω, and therefore there is a work deficit equal to F(ρ)−F(ω).
is the projector onto the ith energy subspace). Hence, the protocol extracts the same amount of work as it would have if it had operated on ω, and therefore there is a work deficit equal to F(ρ)−F(ω).
Additional information
How to cite this article: Skrzypczyk, P. et al. Work extraction and thermodynamics for individual quantum systems. Nat. Commun. 5:4185 doi: 10.1038/ncomms5185 (2014).
References
Bekenstein, J. D. Black holes and entropy. Phys. Rev. D 7, 2333–2346 (1973).
Bardeen, J. M., Carter, B. & Hawking, S. W. The four laws of black hole mechanics. Comm. Math. Phys. 31, 83–170 (1973).
Geva, E. & Kosloff, R. The quantum heat engine and heat pump: An irreversible thermodynamic analysis of the three-level amplifier. J. Chem. Phys. 104, 7681–7699 (1996).
Youssef, M., Mahler, G. & Obada, A.-S. F. Quantum optical thermodynamic machines: lasing as relaxation. Phys. Rev. E 80, 061129 (2009).
Tonner, F. & Mahler, G. Autonomous quantum thermodynamic machines. Phys. Rev. E 72, 066118 (2005).
Linden, N., Popescu, S. & Skrzypczyk, P. How small can thermal machines be? The smallest possible refrigerator. Phys. Rev. Lett. 105, 130401 (2010).
Levy, A. & Kosloff, R. Quantum absorption refrigerator. Phys. Rev. Lett. 108, 070604 (2012).
Horodecki, M. & Oppenheim, J. (Quantumness in the context of) Resource theories. Int. J. Mod. Phys. B 27, 1345019 (2013).
Bennett, C. H., Bernstein, H., Popescu, S. & Schumacher, B. Concentrating partial entanglement by local operations. Phys. Rev. A 53, 2046–2052 (1996).
Horodecki, R., Horodecki, P., Horodecki, M. & Horodecki, K. Quantum entanglement. Rev. Mod. Phys. 81, 865–942 (2009).
Bartlett, S. D., Rudolph, T. & Spekkens, R. W. Reference frames, superselection rules, and quantum information. Rev. Mod. Phys. 79, 555–609 (2007).
Horodecki, M., Horodecki, P. & Oppenheim, J. Reversible transformations from pure to mixed states and the unique measure of information. Phys. Rev. A 67, 062104 (2003).
Marvian, I. & Spekkens, R. W. The theory of manipulations of pure state asymmetry: I. basic tools, equivalence classes and single copy transformations. New J. Phys. 15, 033001 (2013).
Horodecki, M. & Oppenheim, J. Fundamental limitations for quantum and nanoscale thermodynamics. Nat. Commun. 4, 2059 (2013).
Brandao, F. G. S. L., Horodecki, M., Oppenheim, J., Renes, J. M. & Spekkens, R. W. Resource theory of quantum states out of thermal equilibrium. Phys. Rev. Lett. 111, 250404 (2013).
Brandao, F. G. S. L., Horodecki, M., Ng, N. H. Y., Oppenheim, J. & Wehner, S. The second laws of quantum thermodynamics. Preprint at http://arXiv.org/abs/1305.5278 (2013).
Janzing, D., Wocjan, P., Zeier, R., Geiss, R. & Beth, Th. Thermodynamic cost of reliability and low temperatures: tightening Landauer’s principle and the second law. Int. J. Theor. Phys. 39, 2717–2753 (2000).
Procaccia, I. & Levine, R. D. Potential work: a statisticalmechanical approach for systems in disequilibrium. J. Chem. Phys. 65, 3357–3364 (1976).
Alicki, R., Horodecki, M., Horodecki, P. & Horodecki, R. Thermodynamics of quantum information systems—Hamiltonian description. Open Syst. Inf. Dyn. 11, 205–217 (2004).
Gemma, G., Michel, M. & Mahler, G. Quantum Thermodynamics Springer (2004).
Gyftopoulos, E. P. & Beretta, G. P. Thermodynamics: Foundations and Applications Dover (2005).
Hasegawa, H.-H., Ishikawa, J. & Driebe, D. J. Generalization of the second law for a nonequilibrium initial state. Phys. Lett. A 374, 1001–1004 (2010).
Takara, K., Hasegawa, H.-H. & Driebe, D. J. Generalization of the second law for a transition between nonequilibrium states. Phys. Lett. A 375, 88–92 (2010).
Esposito, M. & Van den Broeck, C. Second law and Landauer principle far from equilibrium. Europhys. Lett. 95, 40004 (2011).
Åberg, J. Truly work-like work extraction via a single-shot analysis. Nat. Commun. 4, 1925 (2013).
Verley, G. & Lacoste, D. Fluctuation theorems and inequalities generalizing the second law of thermodynamics out of equilibrium. Phys. Rev. E 86, 051127 (2012).
Allahverdyan, A. E., Balian, R. & Nieuwenhuizen, T. M. Maximal work extraction from finite quantum systems. Europhys. Lett. 67, 565–571 (2004).
Allahverdyan, A. E., Serral Graci, R. & Nieuwenhuizen, T. Work extraction in the spin-boson model. Phys. Rev. E 71, 046106 (2005).
Allahverdyan, A. E., Johal, R. S. & Mahler, G. Work extremum principle: structure and function of quantum heat engines. Phys. Rev. E 77, 041118 (2008).
Allahverdyan, A. E. & Hovhannisyan, K. V. Work extraction from microcanonical bath. Europhys. Lett. 95, 60004 (2011).
Dahlsten, O. C. O., Renner, R., Rieper, E. & Vedral, V. Inadequacy of von Neumann entropy for characterizing extractable work. New J. Phys. 13, 053015 (2011).
Rio, L. d., Åberg, J., Renner, R., Dahlsten, O. C. O. & Vedral, V. The thermodynamic meaning of negative entropy. Nature 474, 61–63 (2011).
Egloff, D., Dahlsten, O. C. O., Renner, R. & Vedral, V. Laws of thermodynamics beyond the von Neumann regime. Preprint at http://arXiv.org/abs/1207.0434 (2012).
Faist, P., Dupuis, F., Oppenheim, J. & Renner, R. A quantitative Landauer’s principle. Preprint at http://arXiv.org/abs/1211.1037 (2012).
Brunner, N., Linden, N., Popescu, S. & Skrzypczyk, P. Virtual qubits, virtual temperatures, and the foundations of thermodynamics. Phys. Rev. E 85, 05111 (2012).
Åberg, J. Catalytic coherence. Preprint at http://arXiv.org/abs/1304.1060 (2013).
Lieb, E. H. & Yngvason, J. The physics and mathematics of the second law of thermodynamics. Phys. Rep. 310, 1–96 (1999).
Ladyman, J., Presnell, S. & Short, A. J. The use of the information-theoretic entropy in thermodynamics. Stud. Hist. Phil. Sci. B 39, 315–324 (2008).
Popescu, S., Short, A. J. & Winter, A. Entanglement and the foundations of statistical mechanics. Nat. Phys. 2, 754–758 (2006).
Reimann, P. Foundation of statistical mechanics under experimentally realistic conditions. Phys. Rev. Lett. 101, 190403 (2008).
Linden, N., Popescu, S., Short, A. J. & Winter, A. Quantum mechanical evolution towards thermal equilibrium. Phys. Rev. E 79, 061103 (2009).
Reimann, P. Canonical thermalization. New J. Phys. 12, 055027 (2010).
Short, A. J. Equilibration of quantum systems and subsystems. New J. Phys. 13, 053009 (2011).
Riera, A., Gogolin, C. & Eisert, J. Thermalization in nature and on a quantum computer. Phys. Rev. Lett. 108, 080402 (2012).
Nielsen, M. A. & Chuang, I. L. Quantum Computation and Quantum Information Cambridge Univ. Press (2000).
Acknowledgements
SP acknowledges support from the European project (Integrated Project ‘Q-ESSENCE’), the EPSRC DIQIP, the ERC (Advanced Grant ‘NLST’) and the Templeton Foundation. AJS acknowledges support from the Royal Society. Part of this work was supported by the COST Action MP1209 ‘Thermodynamics in the quantum regime’. PS acknowledges support by the Marie Curie COFUND action through the ICFOnest program. PS is grateful to Lidia del Rio, Johan Åberg, Joe Renes, Philippe Faist, Renato Renner, Oscar Dahlsten and Luis Masannes for interesting discussions.
Author information
Authors and Affiliations
Contributions
All authors contributed extensively to all aspects of this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Skrzypczyk, P., Short, A. & Popescu, S. Work extraction and thermodynamics for individual quantum systems. Nat Commun 5, 4185 (2014). https://doi.org/10.1038/ncomms5185
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms5185
This article is cited by
-
Work, Heat and Internal Energy in Open Quantum Systems: A Comparison of Four Approaches from the Autonomous System Framework
Journal of Statistical Physics (2024)
-
Remote creation of quantum coherence via indefinite causal order
Quantum Information Processing (2023)
-
Resource Theory of Heat and Work with Non-commuting Charges
Annales Henri Poincaré (2023)
-
The nonequilibrium cost of accurate information processing
Nature Communications (2022)
-
The tight Second Law inequality for coherent quantum systems and finite-size heat baths
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.