Abstract
Cell identity is specified in the early mammalian embryo by the generation of precursors for two cell lineages: the pluripotent inner cell mass and differentiating trophectoderm. Here we identify Angiomotin as a key regulator of this process. We show that the loss of Angiomotin, together with Angiomotin-like 2, leads to differentiation of inner cell mass cells and compromised peri-implantation development. We show that Angiomotin regulates localization of Yap, and Yap-binding motifs are required for full activity of Angiomotin. Importantly, we also show that Angiomotin function can compensate for the absence of Lats1/2 kinases, indicating the ability of Angiomotin to bypass the classical Hippo pathway for Yap regulation. In polarized outside cells, Angiomotin localizes apically, pointing to the importance of cell polarity in regulating Yap to promote differentiation. We propose that both Hippo pathway-dependent and Hippo pathway-independent mechanisms regulate Yap localization to set apart pluripotent and differentiated lineages in the pre-implantation mouse embryo.
Similar content being viewed by others
Introduction
As development progresses, cell identity must be correctly specified. In early mammalian development, three cell types have to be set apart by the blastocyst stage: epiblast, which will give rise to the embryo proper, trophectoderm (TE) and primitive endoderm (PE), which will provide the extra-embryonic structures such as the placenta and yolk sac. Specifying these three cell lineages is mediated through three waves of asymmetric cell divisions that start at the eight-cell stage and generate nonpolar inside and polar outside cells1,2. Inside cells constitute the so-called inner cell mass (ICM) and will develop further as either the pluripotent epiblast or the PE, largely depending on whether an inside cell is generated by the first, second or third wave of asymmetric division3,4. Outside cells will progressively differentiate into TE. Thus, inside and outside cells are progenitors for pluripotent ICM and TE lineages, respectively.
Three lines of evidence indicate that regulation of expression of the transcription factor Cdx2 is particularly crucial to initiate diversification of ICM and TE lineages5,6,7,8. First, Cdx2 is the first transcription factor identified thus far whose expression becomes restricted to the outside cells as soon as they form6, whereas pluripotency factors are initially expressed in both inside and outside cells. Second, Cdx2 downregulation leads to the upregulation of pluripotency genes in outside cells and, consequently, inhibition of TE formation and developmental arrest5,6. Finally, experimental upregulation of Cdx2 in inside cells prevents pluripotency of the ICM8.
The differential expression of Cdx2 between ICM and TE progenitor cells is brought about along two major routes. We recently found that one of these routes involves asymmetric, apical localization of Cdx2 mRNA that results in outside cells inheriting more Cdx2 mRNA than inside cells upon asymmetric cell division6,8. Accordingly, preventing the asymmetric localization and inheritance of Cdx2 mRNA leads to the accumulation of Cdx2 protein in inside cells and, consequently, inhibition of pluripotency genes such as Nanog8. In a second route, transcriptional control is achieved through localization of a transcriptional co-activator Yap, which permits Tead4-mediated transcription of Cdx2 (ref. 99). In the polarized outside cells, Yap is nuclear, whereas in inside non-polarized cells Yap is cytoplasmic. This localization of Yap in mouse embryos is surprising because in other model systems Yap is retained in the cytoplasm of polarized cells, through the activity of apically associated proteins such as Crumbs, Merlin, Expanded and Angiomotin (Amot)10,11,12,13. This raises the question of whether, and if so how, cell polarity might be involved in regulation of Yap localization in the mouse embryo. To resolve this conundrum, it has been proposed that the differential localization of Yap in the mouse is achieved through differential Hippo signalling due to differences in the cell–cell contacts between the inside and outside cells9. However, whether there might be an alternative mechanism involved in controlling Yap localization to regulate expression of differentiation genes such as Cdx2 and lineage segregation in mouse development has remained unknown.
Here we wished to elucidate further the mechanisms underlying the regulation of Cdx2 expression and the potential role of cell polarity in affecting localization of Yap to control this process. To this end, we chose to follow the role of Amot, a protein known to bind to both tight-junction proteins and also to Yap/Taz11,12,13,14,15,16,17. Amot is known to have an important role in a variety of developmental process18,19, but any role for Amot in the pre-implantation embryo has been unknown. Our results indicate that cell polarity contributes to directing the localization of Amot and of Yap in early mouse development. Importantly, Yap localization and therefore Cdx2 expression is regulated not only through a Hippo pathway-dependent mechanism but also through a Hippo pathway-independent mechanism.
Results
Differential expression of Amot in ICM and TE precursors
To determine the potential function of Amot in regulating cell-lineage specification in mouse embryos, we first examined its expression throughout pre-implantation development. Interrogation of our previously generated genome-wide microarray data set20 indicated that Amot expression rises from the four-cell stage to peak at the early blastocyst stage. This prompted us to analyse the localization pattern of Amot protein, using an antibody that detects the C-terminal part of both p130 and p80 isoforms (Fig. 1). We were able to detect Amot protein from the late eight-cell stage onwards when it becomes localized to the membrane, consistent with Amot being a membrane-associated protein21. However, when progenitors of the ICM and TE lineages become set apart, at the 8- to 16-cell transition, Amot becomes localized throughout the membrane in the inside cells, whereas in outside cells it is restricted to the apical domain (Fig. 1). As development progresses, Amot becomes highly expressed in the ICM but barely detectable in the TE (Fig. 1). These results indicate that Amot becomes expressed and localized differentially between the progenitors of the pluripotent ICM and the differentiating TE lineages.
Immunofluorescent confocal images of Amot. Amot is first detected at the compacted eight-cell stage, where it is localized to the membrane (N=16). At the 8- to 16-cell transition, Amot is localized apically in the outside cells and throughout the membrane of the inside cells (N=12). By the blastocyst stage, Amot is much more abundant in the ICM compared with the TE (N=18). Weak Amot staining can be seen on the apical domain of TE cells. Yellow areas and dotted lines mark inside cells. Scale bar, 10 μm.
Amot is required to prevent differentiation of the ICM
To determine the role of Amot, we used RNA interference (RNAi) to specifically downregulate its expression from the zygote stage22. Amot small interfering RNAs (siRNAs) were injected into the zygote, together with Ruby mRNA as a marker of successful injection, and developmental progression was compared with embryos injected with control siRNA (Fig. 2a). Three different siRNAs for Amot were injected and the siRNA resulting in the highest knockdown efficiency (79.1±4% by qRT–PCR) (Fig. 2c) was used in subsequent experiments. We also confirmed Amot depletion at the protein level (Fig. 2a). Amot depletion did not prevent blastocyst formation (Fig. 2a) and did not affect total cell number (Fig. 2b). However, we found that Cdx2, critical for TE differentiation, was detectable not only in the TE but also within the ICM in Amot-RNAi embryos (Fig. 2a). Overall, 61.8±5% of total ICM cells in Amot-RNAi blastocysts were clearly expressing Cdx2, as compared with 5.0±2% in control embryos (Fig. 2b). As we observed that the absence of Amot leads to Cdx2 expression in the ICM, we examined the localization of the co-activator of Cdx2 expression, Yap9. We found Yap to be localized to the nuclei of ICM cells upon Amot-RNAi (Fig. 2a). Amot depletion also led to the expression of other TE-marker genes such as Eomes and Ck-8 (Troma-1) in the ICM (Fig. 2a). The use of an siRNA control, together with rescue experiments (described later), indicate that the Amot-RNAi phenotype is specific and not an artefact of either microinjection or off-target effects.
(a) Immunofluorescent confocal images of TE/ICM marker genes of control and Amot-RNAi blastocysts. Control (N=20)/Amot siRNA (N=30) were injected into zygotes and cultured to blastocysts; Amot protein was undetectable in Amot-RNAi blastocysts. In the absence of Amot, TE markers Cdx2 (N=30), Eomes (N=16) and Ck-8 (antibody: Troma-1) (N=16) become upregulated in the ICM and Yap localized to the nuclei of ICM cells (N=12) in contrast to control blastocysts, where Yap is cytoplasmic (N=12). Pluripotency markers Oct4 and Nanog (N=11) are still visible. Yellow areas and dotted lines mark inside cells. Scale bar, 10 μm. (b) Total cell number of Amot-RNAi versus control blastocysts. The total number of cells between Amot-RNAi (46.3±2cells, N=20 embryos) and control blastocysts (43.2±2cells, N=30 embryos) is not significantly different (P=0.51, Student’s t-test). The percentage of Cdx2-positive cells in the ICM of Amot-RNAi blastocysts (61.8±5%, N=434 cells, 30 embryos) is greater than in control blastocysts (5.0±2%, N=311 cells, 20 embryos). Error bars represent s.e.m.. Student’s t-test was used to test significance. (c) qRT–PCR of whole embryos, comparing expression levels of several TE/ICM markers of Amot-RNAi (N=60) versus control (N=60) blastocysts. Error bars represent s.e.m. Student’s t-test was used to test significance. (d) Immunofluorescence of embryos clonally depleted of Amot, showing that Amot governs cell fate in a cell-autonomous manner. One blastomere of two-cell embryos was injected with control/Amot siRNA and Ruby mRNA, and cultured to blastocysts. Ruby-positive cells mark the injected clone. In the ICM of the mosaic Amot-RNAi embryos, only the Ruby-positive cells expressed Cdx2 (N=21 cells) (arrows). Yellow areas mark the ICM. Dotted lines mark Ruby-positive ICM cells. Scale bar, 10 μm. (e) Immunofluorescence of embryos that are clonally overexpressing Amot, showing that Amot is not sufficient to drive cells to ICM. One blastomere of two-cell embryos was injected with Ruby mRNA only or Amot and Ruby mRNA, and cultured to blastocysts (N=10). Ruby-positive cells mark the overexpressing clone. Yellow areas mark the ICM. Dotted lines mark Ruby-positive cells. Scale bar, 10 μm.
We next wished to determine the consequences of Amot-RNAi on pluripotency factors. qRT–PCR of Amot-RNAi blastocysts revealed that the transcript levels of Oct4, Nanog and Sox2 were significantly reduced to 38.5±11%, 29.8±2% and 17.4±5%, respectively (P<0.05, Student’s t-test) (Fig. 2c), although Oct4 and Nanog proteins were still detectable (Fig. 2a). In contrast, and in agreement with our earlier analyses, the transcript levels of Cdx2 and Eomes increased to 214.9±18% and 281.0±19% of control levels, respectively, whereas Gata3, another TE-marker gene, was not significantly elevated (Fig. 2c). Together, these results suggest that, in the absence of Amot, although cells can be allocated to the ICM, they express TE-specific genes and the pluripotency network (Oct4, Nanog and Sox2) becomes suppressed.
Amot acts in a cell-autonomous manner
As Amot localizes to membranes, we wondered whether it might be able to signal and affect the fate of neighbouring cells. To investigate this, we injected Amot siRNA into one blastomere at the late two-cell embryo, co-injecting Ruby mRNA as a lineage marker, and analysed embryo development at the blastocyst stage. We found that the Amot-depleted clones contributed to both inside (53.3±1%) and outside (46.7±1%) cells, suggesting that the cell allocation process per se was unaffected. In all embryos, only the clones of Amot-depleted cells were expressing Cdx2 in the ICM (Fig. 2d), suggesting that loss of Amot in one cell does not affect a neighbouring cell, and therefore Amot regulates cell fate in a cell-autonomous manner.
Amot overexpression is insufficient to drive cells to ICM
After establishing that Amot is necessary for correct ICM specification, we sought to determine whether Amot is sufficient to induce pluripotent ICM fate when overexpressed. To examine this, we injected p130-Amot mRNA into one blastomere at the late two-cell embryo, co-injecting Ruby mRNA as a marker (Fig. 2e). We confirmed Amot overexpression by immunofluorescence (Supplementary Fig. S1). Analysis of cell allocation revealed that the Amot overexpressing clone contributed to both ICM (46.8±2%) and TE (53.2±2%) lineages, suggesting that Amot is insufficient to drive cells into the ICM. Interestingly, by the blastocyst stage, Amot remained highly expressed only in the ICM and markedly downregulated in the TE (Supplementary Fig. S2), pointing to a potential regulatory mechanism in the TE that degrades Amot to prevent incorrect fate specification.
Depletion of Amot with Amotl2 enhances ICM differentiation
Our results indicate that the lack of Amot leads to the expression of differentiation genes within the ICM and downregulation of pluripotency. It has been reported that Amot−/− embryos can progress beyond implantation18,19, prompting us to test whether Amot-related proteins might be able to compensate for Amot function. Amot belongs to the motin family that contains two other members: Amot-like 1 (Amotl1) and Amot-like 2 (Amotl2) (Fig. 3a)23, both of which have overlapping functions with Amot11,12,13,24. To eliminate all three motins, we injected Amot, Amotl1, Amotl2 siRNAs into zygotes and examined developmental progression. We confirmed knockdown by qRT–PCR: Amot transcripts were depleted to 21.6±3% and Amotl2 to 19.6±3%, whereas Amotl1 transcripts were undetectable (Fig. 3c). We found that co-depletion of Amot and Amotl2 permitted blastocyst formation (9/10, 90%; control siRNA: 9/10, 90%) and did not affect the total cell number or number of ICM cells per embryo (Fig. 3b). Strikingly, however, the proportion of Cdx2-expressing cells within the ICM was much higher in the combined motin-RNAi embryos than upon depletion of Amot alone. Nearly all (98.0±1%) ICM cells of the Amot- and Amotl2-RNAi embryos were Cdx2 positive compared with the 61.8±5% of Amot only-RNAi and 5.0±2% of control embryos (Fig. 3b,d). This suggests that Amot and Amotl2 both function to suppress Cdx2 expression in the ICM.
(a) The motin family proteins with key regions outlined. (b) Total cell number and ICM cell number are not significantly different between control (N=21), Amot-RNAi (N=31) and Amot; Amotl1; Amotl2-RNAi (N=12) blastocysts. However, in the triple motin-RNAi embryos, 98.0±1% of ICM cells of the triple RNAi embryos were Cdx2 positive at the blastocyst stage (N=210 cells, 12 embryos) compared with the 61.8±5% of Amot-RNAi (N=434 cells, 30 embryos) and 5.0±2% of control embryos (N=311 cells, 21 embryos). Error bars represent s.e.m. Student’s t-test was used to test significance. (c) qRT–PCR of whole embryos, comparing expression levels of Amot and Amotl2 in control embryos (N=21) and triple knockdown embryos (N=21). Amot and Amotl2 were downregulated to 21.6±3% and 19.6±3%, respectively, relative to control levels. Amotl1 expression could not be detected. Error bars represent s.e.m. Student’s t-test was used to test significance. (d) Z-section series of immunofluorescent confocal images of control, Amot-RNAi (N=30) and Amot; Amotl1; Amotl2-RNAi (N=12) blastocysts, with the highest proportion of Cdx2-expressing cells in the triple knockdown. Dotted lines outline the ICM. Scale bar,: 10 μm.
Amot depletion compromises developmental potential
To determine whether the ICM differentiation caused by the loss of Amot would affect development in vivo, Amot-RNAi and Amot, Amotl1, Amotl2-RNAi embryos were transferred into pseudo-pregnant surrogates to assess their developmental potential (Supplementary Table S1). siRNAs were injected into both blastomeres at the two-cell stage, rather than the zygote, to prolong the RNAi effect (Fig. 4a). GFP transgenic embryos injected with control siRNA were used as transfer controls to normalize for transfer efficiency in different recipients. After injection, the embryos were cultured to the blastocyst stage and sampled to confirm their respective phenotypes (Fig. 4b). To assess for successful development beyond implantation, the embryos were recovered at E7.5 (Fig. 4c). After normalizing for the transfer efficiency of the different recipients and background of the embryos, we found that 100% of the control-RNAi embryos progressed successfully to E7.5, compared with 32.7% of Amot-RNAi embryos (P<0.05, χ2-test, compared with control) and 27.8% of the combined motin-RNAi embryos (P<0.05, χ2-test, compared with control). There was no significant difference between the developmental success of Amot-RNAi and combined motin-RNAi embryos (P=0.67, χ2-test), although loss of all three motins might lead to a more severe outcome (Supplementary Table S2). Overall, our result suggests that loss of Amot compromises development, potentially as a result of a more differentiated ICM, signifying an important role for Amot in early mouse development.
(a) Experimental outline: siRNA was injected to both blastomeres of two-cell-stage embryo, cultured to E3.5 and transferred to 2.5 dpc pseudo-pregnant recipients. The embryos were recovered 5 days later at E7.5. Embryos were injected with either control siRNA, Amot siRNA or a combination of Amot, Amotl1 and Amotl2 siRNA. For each of the three groups (control, Amot-RNAi and Amot; Amotl1; Amotl2-RNAi), 20 WT embryos and 20 GFP embryos were transferred to two surrogates. (b) Embryos from each group were sampled to confirm their phenotype following RNAi. Amot-RNAi embryos (N=9) and Amot; Amotl1; Amotl2-RNAi (N=6) expressed CDX2 in the inside cells, whereas in GFP (N=10) and WT (N=10) embryos injected with control siRNA CDX2 expression was restricted to the outside cells. (c) The outcome of the embryo transfers. To assess for developmental success, the embryos were recovered at E7.5. For the control-RNAi group, out of the 40 embryos transferred, 38 embryos were recovered, of those 20 (100%) were GFP+ and 18 (90%) were GFP−. For Amot-RNAi, out of 40 embryos transferred, 22 embryos were recovered, of those 17 (85%) were GFP+ and 5 (25%) were GFP−. For the Amot; Amotl1; Amotl2-RNAi, out of 40 embryos transferred, 20 embryos were recovered, of those 16 (80%) were GFP+ and 4 (20%) were GFP−. After normalizing for the transfer efficiency of the different recipients and background of the embryos, 100% of the control-RNAi embryos progressed successfully to E7.5, compared with 32.7% of Amot-RNAi embryos (P<0.05, χ2-test, compared with control) and 27.8% of Amot;Amotl1;Amotl2-RNAi embryos (P<0.05, χ2-test, compared with control). There was no significant difference between the developmental success of Amot-RNAi and Amot; Amotl1; Amotl2-RNAi embryos (P=0.67, χ2-test). Scale bar, 100 μm.
p130-Amot is the isoform involved in cell fate regulation
The Amot protein is expressed as two isoforms, p80 and p130 (refs 21, 25). The p130 isoform has a unique N-terminal extension and the rest of the protein is identical to the p80 isoform (Fig. 5a). To determine which isoform is required for the first cell lineage specification, we depleted Amot with a dsRNA targeting the 3′-UTR in the zygote and then attempted to rescue Amot-RNAi effect with either the p80- or p130-Amot isoforms at the eight-cell stage. Successful knockdown by the dsRNA was confirmed by qRT–PCR (Supplementary Fig. S3). For the rescue, we co-injected either p80- or p130-Amot mRNA, with Ruby mRNA as a marker, into 2–3 blastomeres at the eight-cell stage. To quantify the extent of the rescue, we determined the percentage of the Ruby- and Cdx2-positive cells in the ICM (Fig. 5b). In control blastocysts, 12.6±5% of the Ruby-marked ICM cells were Cdx2-positive (Fig. 5c) (most likely owing to inherited Cdx2 protein from an outside mother cell). This value increased significantly (P<0.05, Student’s t-test) to 88.7±4% of the Amot-RNAi/Ruby-expressing embryos (Fig. 5c). When we injected p130-Amot mRNA to the Amot-RNAi embryos, the proportion of Cdx2-expressing cells in the ICM dropped significantly to 17.4±7%. This value was significantly different (P<0.05, Student’s t-test) from Amot-RNAi/Ruby-expressing embryos and not significantly different from control embryos (P=0.84, Student’s t-test), indicating that p130-Amot is able to fully rescue the effects of Amot depletion. We found that providing p80-Amot into Amot-RNAi embryos still resulted in 71.2±7% of ICM cells expressing Cdx2, which was significantly different from the control embryos (P<0.05, Student’s t-test) (Fig. 5b,c). Therefore, the Amot-RNAi effect can be rescued only by the p130 isoform, indicating that p130-Amot function is required to suppress the differentiation pathway in the ICM.
(a) Amot constructs with key regions outlined. The p80- and p130-Amot isoforms are identical, except the p130 isoform has an N-terminal extension, which contains three Yap-binding motifs. (b) Eight sets of experiments (i)–(viii) are shown, which follow the same experimental outline: siRNA/dsRNA was injected into the zygote and mRNA was injected into two or three blastomeres at the eight-cell stage. Cdx2-positive inside cells from all eight sets of experiments were counted manually. The phenotype was quantified by calculating the percentage of Cdx2-positive cells from the Ruby-positive inside cells at the blastocyst stage (or the morula stage in the case of Lats1/2 knockdown). Panels show confocal images of representative embryos from each set of experiments. Dotted lines mark Ruby-positive inside cells. Scale bar, 10 μm. (c) Combined quantifications from the experiments outlined in (b). Number of biological replicates: (i) N=41 cells, 15 embryos; (ii) N=96 cells 34 embryos; (iii) N=53 cells, 35 embryos; (iv) N=44 cells, 11 embryos; (v) N=20 cells, 40 embryos; (i) N=22 cells, 9 embryos; (vi) 139 cells, 24 embryos; (vii) N=145 cells, 43 embryos; (viii) N=58 cells, 18 embryos. Error bars represent s.e.m. This quantification shows that the p130, but not the p80, isoform is able to rescue the Amot phenotype and the Yap-binding motifs are required for the full activity of p130-Amot. Lats1/2 depletion leads to an increased Cdx2-positive inside cell number, but overexpression of p130-Amot is able to prevent Cdx2 expression in the absence of Lats1/2, demonstrating a Hippo pathway-independent mechanism. Confirming this conclusion overexpressing the Amot mutant for Yap-binding motifs in a Lats1/2-RNAi background does not affect the Lats1/2 phenotype.
Yap-binding motifs are involved in Amot function
The N-terminal extension of the p130-Amot contains two PPxY motifs. Mutating the tyrosine residues to alanine in the PPEY motifs abolishes the Yap-binding ability of Amot and its ability to tether Yap in the cytoplasm11,12,13. A third motif conserved across all motins, LPTY, has been suggested to mediate interaction between Yap and Amotl1 (ref. 26). These motifs therefore represent a Yap-dependent but Hippo pathway-independent activity of Amot. We sought to determine whether Amot might function in the mouse embryo independently of the Hippo pathway. We hypothesized that if overexpression of the triple motif mutant (Y110/243/288 A) could rescue the Amot-depletion phenotype, Yap-binding motifs are consequently not important. If the opposite is true, it would follow that Amot functions through a Hippo pathway-independent, Yap-binding mechanism. To test this, we depleted Amot, from the zygote as before, and subsequently injected mRNA for Y110/243/288A Amot with Ruby mRNA as a lineage marker, into 2–3 blastomeres at the eight-cell stage. We found that, at the blastocyst stage, 42.0±7% of the clonal Ruby-positive ICM cells were expressing Cdx2 (Fig. 5c), which was significantly lower than that in Amot-RNAi embryos (88.7±4%, P<0.05). This suggests the Yap-binding motifs are required for the full activity of Amot, and therefore Amot function is, in part, Yap binding dependent. In agreement with this, Yap staining in Amot overexpressing embryos suggests possible co-localization between Yap and Amot (Supplementary Fig. S4), as previously shown in MCF10A cells11. In some embryos (Fig. 5c, panel v), however, Cdx2 was not detectable in the ICM upon the Y110/243/288A mutant overexpression, suggesting that the mutant Amot was partially able to suppress Cdx2 expression. Indeed, the proportion of Ruby- and Cdx2-positive ICM cells following injection of mRNA for Y110/243/288A Amot was 42.0±7%, an intermediate value between 17.4±7% of the control and 88.7±4% of the Amot-RNAi embryos (Fig. 5c). These results indicate that the Yap-binding sites are required for the full activity of Amot, raising the possibility that pluripotent lineage specification can be regulated in a Yap-binding-dependent but a Hippo pathway-independent manner.
Amot compensates for Lats1/2 function
The above results opened up a possibility that Amot can regulate Yap localization, and consequently Cdx2 expression, independently of the Hippo pathway. If this is the case, Amot overexpression should suppress Cdx2 expression in the absence of Hippo pathway kinases, Lats1/2, which phosphorylate Yap to prevent its nuclear localization9. To directly test this, we eliminated Lats1 and Lats2 transcripts by injecting zygotes with Lats1/2 siRNAs and, at the eight-cell stage, injected 2–3 blastomeres with either Ruby mRNA only or p130-Amot mRNA with Ruby mRNA. We verified successful depletion of Lats1/2 by qRT–PCR (Supplementary Fig. S5). On assessing at the morula stage, we found that Lats1/2-RNAi/Ruby-expressing embryos had 35.7±6% of Ruby-positive inside cells expressing Cdx2, which was significantly more than the 5.9±4% in control-RNAi/Ruby-expressing embryos of the same stage (P<0.05, Student’s t-test), in agreement with previous report9. Importantly, overexpression of Amot following Lats1/2 depletion significantly decreased the proportion of Cdx2- and Ruby-positive inside cells to 18.2±4% (Fig. 5c), compared with Lats1/2-RNAi/Ruby-expressing embryos (P<0.05, Student’s t-test). This value was not significantly different from the control-RNAi/Ruby-expressing embryos (P=0.06, Student’s t-test). These results suggest that by increasing the amount of Amot its Hippo pathway-independent activity can compensate for the loss of the Hippo pathway kinase Lats1/2.
To confirm this conclusion, we next overexpressed Y110/243/288A Amot that cannot bind Yap in the Lats1/2-RNAi background. To this end, we depleted Lats1/2 as above, but now co-injected Y110/243/288A Amot, with Ruby mRNA as a lineage tracer, to 2–3 blastomeres at the eight-cell stage. In these embryos, 41.4±8% of the Ruby-positive inside cells were Cdx2 positive, a value not significantly different from the 35.7±6% of the Lats1/2-RNAi/Ruby-expressing embryos (P=0.58, Student’s t-test) (Fig. 5c). Thus, the Y110/243/288A mutant is not able to compensate for the loss of Lats1/2, further pointing to a direct Amot/Yap interaction that bypasses the classical Hippo pathway. On a side note, this also indicates that the Lats1/2 phenotype is Yap dependent, as only Amot with intact Yap-binding sites can compensate for Lats1/2 function. Taken together, these results further indicate that Amot can function in a Yap-dependent but Hippo pathway-independent manner to specify cell fate in the mouse embryo.
Discussion
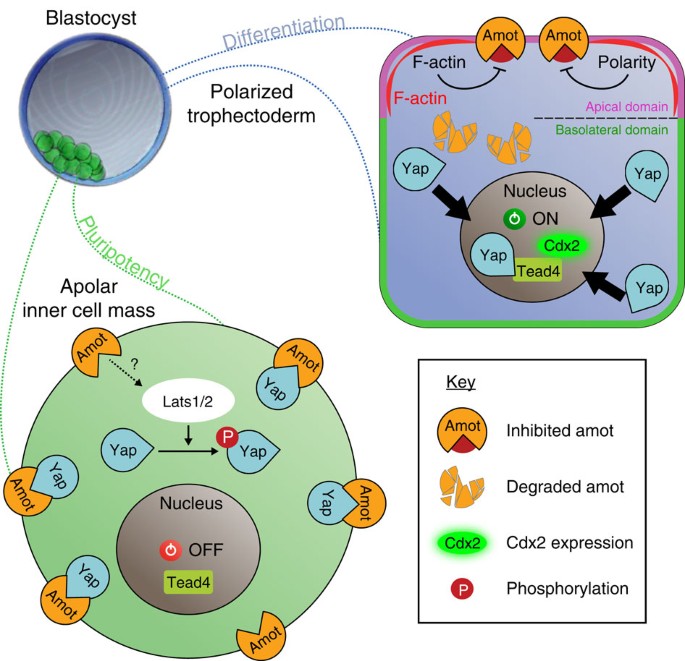
Analyses of spatial and temporal expression patterns together with the downregulation and overexpression studies we present here identify Amot as a new important regulator of the first cell fate specification in the mouse embryo. We find that Amot becomes enriched in the ICM versus TE progenitor cells as development progresses; Amot is critical to prevent expression of TE-marker genes in the pluripotent lineage; ICM differentiation is significantly enhanced by co-depletion of Amot with Amotl2; Amot regulates localization of Yap; full activity of Amot is dependent on the Yap-binding motifs, suggesting a Hippo pathway-independent function; Amot function can compensate for the absence of Lats1/2, supporting its ability to function independently of the Hippo pathway; and mutating the Yap-binding motifs affects, but does not completely abolish, Amot activity, suggesting that Amot might also act through an alternative mechanism, such as through activating Lats1/2 via the Hippo pathway or independent of Yap and, finally, that Amot depletion, together with depletion of Amotl2, results in compromised development at peri-implantation stages. Overall, our results identify a role of Amot in the first-lineage specification to prevent an inappropriate cell fate program in the pluripotent ICM and suggest that Amot has two distinct roles in this process, sequestering Yap, which is Hippo pathway independent, and also activating the Hippo pathway kinase Lats1/2 (Fig. 6).
The results we present here show that Amot becomes enriched in the ICM as cell lineages become progressively established, and that Amot functions to prevent differentiation of the pluripotent lineage. Our results suggest that Amot acts through two routes to prevent ICM differentiation, of which one is dependent and another is independent of the Hippo pathway. The Hippo pathway-independent (Lats1/2–independent) mechanism is likely to involve Amot’s ability to tether Yap to the cytoplasm/membrane, as we find that Yap-binding motifs of Amot are essential for its full activity. But, it can also involve Amot interacting with actin/cell polarity molecules. The Hippo pathway-dependent mechanism is likely to involve Amot’s role in activating Lats1/2 (as shown in24) to allow phosphorylation of Yap, which prevents its transition to the nucleus9. In agreement, we find that mutation of the Yap-binding sites does not completely abolish the activity of Amot. Combined, this results in Yap being sequestered from the nucleus and therefore the expression of TE-genes, such as Cdx2, is switched off and the ICM pluripotency maintained. In contrast, in TE cells, most of Amot is degraded and the remaining Amot is sequestered to the apical domain. We hypothesize that in this location Amot is shielded from binding Yap through its interactions with F-actin and/or polarity factors. In agreement with this hypothesis, Amot is reported to associate with actin32 and tight-junction proteins via its PDZ-binding domain in tissue culture cells14,15,16,17. This suggests that cell polarity is involved in allowing Yap to translocate to the nucleus to act, together with Tead4, to stimulate Cdx2 expression and TE differentiation.
Our results indicate that loss of Amot results in the upregulation of genes that promote differentiation and, consequently, downregulation of pluripotency genes such as Oct4, Nanog and Sox2 in the ICM. Despite this, we observe that some pluripotency-associated transcription factors are still present, possibly as a result of pre-existing protein. This finding is likely to explain why Amot−/− embryos can survive through implantation and arrest only at early post-implantation stages18,19. This could also be due to developmental plasticity, as the mouse embryo has been shown to require establishment of only four pluripotent cells by implantation to support its development to birth4. Finally, genetic redundancy could also be a contributing factor, as we show here that simultaneous depletion of Amot and Amotl2 lead to a much stronger phenotype than depletion of Amot on its own.
As development progresses, Amot becomes upregulated in ICM but not TE progenitors. A deep sequencing analysis of global gene expression patterns between ICM and TE progenitors after the first wave of asymmetric division, at the 16-cell stage, reveals similar levels of Amot mRNA (Krzysztof Wicher, Sarah Graham and Magdalena Zernicka-Goetz, unpublished observations). Although this does not rule out differential transcription later, our results suggest that Amot enrichment in the ICM might result from differential stability of the protein between ICM and TE cells. In support of this, we show that when Amot is overexpressed, it is downregulated in TE but not in ICM cells (Supplementary Fig. S2). Moreover, one of the E3 ubiquitin ligases able to promote Amot degradation27, NEDD4L, is expressed two-fold higher in TE than in ICM progenitors (Krzysztof Wicher, Sarah Graham and Magdalena Zernicka-Goetz, unpublished observations), thus giving the potential of reducing Amot levels in TE. In addition, cytoplasmic Yap has been found to protect Amotl1 from NEDD4-mediated degradation26, raising an interesting possibility that Yap could act in a positive feedback loop to prevent Amot degradation in the ICM.
Two models are often considered for the first cell fate decision in the mouse embryo. The polarity model1 stresses the importance of cell polarity in directing cell fate, and the positional model28,29 stresses the importance of cell position. It has been demonstrated that cell polarity and cell position re-enforce each other30, and these two models can be brought together conceptually31. Identifying Amot as a cell fate regulator is a molecular step towards this unification, because it lends support to both: Amot becomes enriched in inside but not outside cells (supporting a positional ‘inside-outside’ model) and its localization is polarized in outside cells (supporting a ‘polarity’ model). Apically localized Amot in polarized outside cells may interact with polarity factors, and, in support of this idea, it has been already shown to associate with tight-junction proteins via its PDZ-binding domain14,15,16,17. Therefore, we would like to suggest that, in the mouse embryo, as in other model systems, cell polarity is involved in Yap regulation as it can, for example, inhibit Amot’s ability to bind Yap to restrain its translocation to the nucleus (Fig. 6).
The ability of Amot to function through Yap is strongly suggested by the three putative Yap-binding motifs of p130-Amot that are absent in p80-Amot and which we show here are essential for the full activity of Amot. Indeed, Yap seemingly co-localizes with p130-Amot when p130-Amot is overexpressed (Supplementary Fig. S4). Another way to test Amot’s Yap dependence would be through an Amot; Yap double knockdown. However, interpreting such an experiment would be difficult because of the redundancy of Yap with its homologue Taz9 (Supplementary Fig. S6).
We find that Amot is partly functional following mutation of its Yap-binding sites, which suggests that two possible mechanisms of Amot function are tenable. One would reflect the direct interaction between Amot and Yap, independent of the Hippo pathway. The second would reflect the residual function of this Yap-binding defective Amot mutant that would still be able to activate Lats1/2, leading to Yap phosphorylation and cytoplasmic retention (as demonstrated in HEK293 cells24). Our results cannot also rule out the possibility that the residual function of this Amot mutant reflects function that is entirely independent of Yap.
The key regulatory domain of Amot appears to be its N-terminal part, as we show that the p80 isoform does not have a role in cell fate regulation. This N-terminal domain could be responsible for activation of Lats1/2-dependent phosphorylation of Yap, as supported by in vitro data24. We also consider the actin-binding properties of the N-terminal domain of p130-Amot32 could relate to its ability to suppress Cdx2 expression in the pluripotent lineage. In agreement with this hypothesis, it has been reported that an increase in filamentous actin results in increased nuclear Yap and activation of Yap target genes33,34,35,36,37,38. Finally, we show that overexpressing p130-Amot prevents the expression of Cdx2 in inside cells lacking Lats1/2. Hence, in the absence of the Hippo pathway, high levels of Amot are sufficient to sequester most of the Yap molecules from the nucleus. Taken together, our results suggest that both Hippo pathway-dependent and -independent processes are involved in regulating Yap localization to generate pluripotent cells in the mouse embryo.
Methods
Embryo collection and culture
Animals were maintained in the Animal Facility of Gurdon Institute at 12:12 light cycle and provided with food and water ad libitum. All experiments were conducted in compliance with Home Office regulations. For embryo collection, 4- to 6-week-old F1 generation mice from the C57Bl6 and CBA strains were superovulated with 10 IU of pregnant mare serum gonadotropin (PMSG; Intervet) and 10 IU human chorionic gonadotropin (hCG; Intervet) 48 h later and mated with F1 (C57Bl6 × CBA) male mice. Embryos were dissected out of the oviducts into M2 medium. For zygote recovery, cumulus cells were removed by hyaluronidase treatment (1 mg ml−1 in M2). Embryos were cultured in drops of KSOM media (Millipore) under paraffin oil in 5% CO2 at 37 °C.
Immunohistochemistry and analysis
Embryos were fixed in 4% PFA for 20 min and washed twice for 5 min in PBS-T (PBS with 0.1% Tween). They were then permeabilized with 0.5% Triton X-100 in PBS for 20 min and washed three times for 5 min in PBS-T. The embryos were incubated with primary antibodies diluted in 3% BSA (Sigma) for 1 h. Cdx2 antibody (Biogenex) was used at a dilution of 1:200; Oct4 antibody (Santa Cruz) at 1:200; Nanog antibody (2BScientific) at 1:200; Yap antibody (Santa Cruz) at 1:400; Eomes antibody (Abcam) at 1:250; Troma-1 at 1:100; and Amot antibody (Santa Cruz) at 1:2,000. The embryos were then washed three times for 5 min in PBS-T. Secondary antibodies (Invitrogen, AlexaFluor) and DAPI (Invitrogen) were applied for 1 h at a dilution of 1:400 in 3% BSA (Sigma). After washing three times for 5 min in PBS-T, the embryos were washed overnight in M2 medium. Embryos were imaged on glass-bottom dishes rather than on slides in order to avoid flattening the embryo. Confocal images were captured with a Leica SP5 confocal microscope with a Z-resolution of 4 μm. All images are layer-normalized using IMARIS to account for signal intensity differences from different sections. To count total cell numbers, DAPI was used as a nuclear marker to represent each cell. Acquisition of Cdx2 stainings for cell counting was performed using identical settings on a Leica SP5. To count Cdx2-positive cells, the confocal z-stacks were first exported to IMARIS and layer-normalized using the built-in function. The normalized sections were then assessed for the presence of Cdx2 signal. Any nuclei with signal detectable by eye were scored as ‘Cdx2 positive’. Cells were then manually counted in ImageJ using the MTrackJ plug-in. Student’s t-test, unless otherwise stated, was used to test statistical significance.
RNAi reagents and construct preparation
p130- and p80-Amot were cloned from mouse kidney cDNA and sequence verified. mRNA was synthesized using the mMESSAGE mMACHINE Kit (Life Technologies). dsRNA was designed and evaluated using E-RNAi, a web application for the design of RNAi reagents39. The DNA template of the dsRNA was amplified from mouse kidney cDNA, and dsRNA was synthesized from DNA using the T7 MEGAscript kit (Life Technologies). The dsRNA was annealed by heating to 70 °C for 5 min and allowed to cool at room temperature for 2 h. It was treated with RNAse T1/A and Proteinase K to remove ssRNA and proteins. Site-directed mutagenesis of the PPxY motifs was performed via overlap extension PCR. Four PCR fragments were amplified from Amot DNA using mutagenic primers, with two fragments overlapping over each PPxY motif. All four fragments were pooled, annealed and PCR amplified into one complete piece of DNA, which was sequence verified. The following is the sequence of Amot siRNA used: 5′-CAGGAGAAGCCTACTCAGCTA-3′. Primers used for cloning are as follows: p80-Amot 5′-TTTTTGGATCCACCATGCCTCGGGCTCAG-3′ 5′-TTCGTTTGCGGCCGCTTAGATGAGATATTCC-3′; p130-Amot 5′- TTTTTTTTTTGGATCCACCATGAGAAGTTCTGACGATCAGCC-3′ 5′-TTTTTTTTTTGCGGCCGCTTAGATGAGATATTCCACC-3′. The following primers were used to generate the p130-Amot Y110/243/288A mutant: 5′-TTTTTTTTTTGGATCCACCATGAGAAGTTCTGACGATCAGCC-3′; 5′-GACCTTGGCCTCTTCGGCGGTTGGGAGTTCTTC-3′; 5′-GAAGAACTCCCAACCGCCGAAGAGGCCAAGGTC-3′; 5′-TTTTTTTTTTATGCATCCTTCTAATCTCGCCCTCCAGCTTGTTCC-3′; 5′-CCCTTGAAAGGGGCTTCTGGCGGGGGACCTC-3′; 5′-GAGGTCCCCCGCCAGAAGCCCCTTTCAAGGG-3′; 5′-CTGCCCCGGCCTCAGGGGGGTGCTGATA-3′; 5′-TATCAGCACCCCCCTGAGGCCGGGGCAG-3′. The following primers were used to generate dsRNA targeting the 3′-UTR of Amot: 5′-TAATACGACTCACTATAGGGTGTGTTTGGGGAGAAAAGGA-3′; 5′-TAATACGACTCACTATAGGGAAGTCCAGGAAAAGGCCTGA-3′.
Embryo microinjection
Microinjection was performed on a Leica DM IRB with attached Leica micromanipulators. Injections were administered via air pressure using an Eppendorf Femtojet microinjector, and negative capacitance was used to facilitate membrane penetration. All injections were carried out on embryos suspended in M2 medium under paraffin oil on a depression slide. Successful microinjections were confirmed by the presence of fluorescent markers co-injected with the constructs. Ruby mRNA (0.4 mg ml−1) was co-injected for all experiments except in the rescue experiments where 0.4 mg ml−1 Gap-RFP was co-injected to mark the membrane without interfering with the cytoplasmic Ruby introduced at the eight-cell stage.
qRT–PCR
RNA was extracted from embryos using the Arcturus PicoPure RNA Isolation Kit. qRT–PCR was carried out using SYBR Green in a StepOne Plus Real-time PCR machine (Applied Biosystems). Gapdh was used as an endogenous control. The following primers were used: Gapdh 5′-AGAGACGGCCGCATCTTC-3′ 5′-CCCAATACGGCCAAATCCGT-3′; Amot 5′-GATGTGCAACCCAGATAAGCC-3′ 5′-TCTCTGCATCAGGCTCTTGC-3′; Cdx2 5′-AAACCTGTGCGAGTGGATG-3′ 5′-TCTGTGTACACCACCCGGTA-3′; Eomes 5′-CGGCCTACCAAAACACGGAT-3′ 5′-AAGCCGTGTACATGGAATCGTA-3′; Gata3 5′-CCGAAACCGGAAGATGTCTA-3′ 5′-AGATGTGGCTCAGGGATGAC-3′; Oct4 5′-TTGGGCTAGAGAAGGATGTGGTT-3′ 5′-GGAAAAGGGACTGAGTAGAGTGTGG-3′; Nanog 5′-GGTTGAAGACTAGCAATGGTTCTGA-3′ 5′-TGCAATGGATGCTGGGATACTC-3′; Sox2 5′-TGCTGCCTCTTTAAGACTAGGG-3′ 5′-CGCCGCGATTGTTGTGATT-3′; Lats1 5′-AACAAGTTCTTCCCGGAGCC-3′ 5′-ACCATGTCCTCATCAAAGCCA-3′; Lats2 5′-GAATGCGGGATGTGACCAG-3′ 5′-TACTCCAAGGCAGCTTCGAT-3′.
Additional information
How to cite this article: Leung, C.Y. and Zernicka-Goetz M. Angiomotin prevents pluripotent lineage differentiation in mouse embryos via Hippo pathway-dependent and independent mechanisms. Nat. Commun. 4:2251 doi: 10.1038/ncomms3251 (2013).
References
Johnson, M. H. & Ziomek, C. A. The foundation of two distinct cell lineages within the mouse morula. Cell 24, 71–80 (1981).
Bruce, A. W. & Zernicka-Goetz, M. Developmental control of the early mammalian embryo: competition among heterogeneous cells that biases cell fate. Curr. Opin. Genet. Dev 20, 485–491 (2010).
Morris, S. A., Teo, R. T. Y., Li, H., Robson, P., Glover, D. M. & Zernicka-Goetz, M. Origin and formation of the first two distinct cell types of the inner cell mass in the mouse embryo. Proc. Natil Acad. Sci. USA 107, 6364 (2010).
Morris, S. A., Guo, Y. & Zernicka-Goetz, M. Developmental plasticity is bound by pluripotency and the Fgf and Wnt signaling pathways. Cell Rep. 2, 756–765 (2012).
Strumpf, D. et al. Cdx2 is required for correct cell fate specification and differentiation of trophectoderm in the mouse blastocyst. Development 132, 2093–2102 (2005).
Jedrusik, A. et al. Role of Cdx2 and cell polarity in cell allocation and specification of trophectoderm and inner cell mass in the mouse embryo. Genes Dev. 22, 2692–2706 (2008).
Jedrusik, A. et al. Maternally and zygotically provided Cdx2 have novel and critical roles for early development of the mouse embryo. Dev. Biol. 344, 66–78 (2010).
Skamagki, M., Wicher, K. B., Jedrusik, A., Ganguly, S. & Zernicka-Goetz, M. Asymmetric localization of Cdx2 mRNA during the first cell-fate decision in early mouse development. Cell. Rep. 3, 442–457 (2013).
Nishioka, N. et al. The Hippo signaling pathway components Lats and Yap pattern Tead4 activity to distinguish mouse trophectoderm from inner cell mass. Dev. Cell 16, 398–410 (2009).
Genevet, A. & Tapon, N. The Hippo pathway and apico-basal cell polarity. Biochem. J 436, 213–224 (2011).
Zhao, B. et al. Angiomotin is a novel Hippo pathway component that inhibits Yap oncoprotein. Genes Dev. 25, 51–63 (2011).
Wang, W., Huang, J. & Chen, J. Angiomotin-like proteins associate with and negatively regulate Yap1. J. Biol. Chem. 286, 4364–4370 (2011).
Chan, S. W., Lim, C. J., Chong, Y. F., Pobbati, A. V., Huang, C. & Hong, W. Hippo pathway-independent restriction of TAZ and Yap by angiomotin. J. Biol. Chem. 286, 7018–7026 (2011).
Sugihara-Mizuno, Y. et al. Molecular characterization of angiomotin/JEAP family proteins: interaction with MUPP1/Patj and their endogenous properties. Genes Cells 12, 473–486 (2007).
Wells, C. D. et al. A Rich1/Amot complex regulates the Cdc42 GTPase and apical-polarity proteins in epithelial cells. Cell 125, 535–548 (2006).
Ernkvist, M. et al. The Amot/Patj/Syx signaling complex spatially controls RhoA GTPase activity in migrating endothelial cells. Blood 113, 244–253 (2009).
Yi, C. et al. A tight junction-associated Merlin-angiomotin complex mediates Merlin’s regulation of mitogenic signaling and tumor suppressive functions. Cancer Cell 19, 527–540 (2011).
Shimono, A. & Behringer, R. R. Angiomotin regulates visceral endoderm movements during mouse embryogenesis. Curr. Biol 13, 613–617 (2003).
Aase, K. et al. Angiomotin regulates endothelial cell migration during embryonic angiogenesis. Genes Dev. 21, 2055–2068 (2007).
Wang, Q. T. et al. A genome-wide study of gene activity reveals developmental signaling pathways in the preimplantation mouse embryo. Dev. Cell 6, 133–144 (2004).
Bratt, A. et al. Angiomotin regulates endothelial cell-cell junctions and cell motility. J. Biol. Chem. 280, 34859–34869 (2005).
Wianny, F. & Zernicka-Goetz, M. Specific interference with gene function by double-stranded RNA in early mouse development. Nat. Cell Biol 2, 70–75 (2000).
Bratt, A. et al. Angiomotin belongs to a novel protein family with conserved coiled-coil and PDZ binding domains. Gene 298, 69–77 (2002).
Paramasivam, M., Sarkeshik, A., Yates, J. R. 3rd, Fernandes, M. J. G. & McCollum, D. Angiomotin family proteins are novel activators of the Lats2 kinase tumor suppressor. Mol. Biol. Cell 22, 3725–3733 (2011).
Ji, W. R. et al. Characterization of kringle domains of angiostatin as antagonists of endothelial cell migration, an important process in angiogenesis. FASEB J 12, 1731–1738 (1998).
Skouloudaki, K. & Walz, G. Yap1 recruits c-Abl to protect angiomotin-like 1 from Nedd4-mediated degradation. PLoS One 7, e35735 (2012).
Wang, C. et al. The Nedd4-like ubiquitin E3 ligases target angiomotin/p130 to ubiquitin-dependent degradation. Biochem. J. 444, 279–289 (2012).
Hillman, N., Sherman, M. I. & Graham, C. The effect of spatial arrangement on cell determination during mouse development. J. Embryol. Exp. Morphol. 28, 263–278 (1972).
Tarkowski, A. K. & Wróblewska, J. Development of blastomeres of mouse eggs isolated at the 4- and 8-cell stage. J. Embryol. Exp. Morphol. 18, 155–180 (1967).
Plusa, B. et al. Downregulation of Par3 and aPKC function directs cells towards the ICM in the preimplantation mouse embryo. J. Cell. Sci. 118, 505 (2005).
Zernicka-Goetz, M., Morris, S. A. & Bruce, A. W. Making a firm decision: multifaceted regulation of cell fate in the early mouse embryo. Nat. Rev. Genet 10, 467–477 (2009).
Ernkvist, M. et al. p130-angiomotin associates to actin and controls endothelial cell shape. FEBS J. 273, 2000–2011 (2006).
Dupont, S. et al. Role of Yap/TAZ in mechanotransduction. Nature 474, 179–183 (2011).
Fernández, B. G., Gaspar, P., Brás-Pereira, C., Jezowska, B., Rebelo, S. R. & Janody, F. Actin-capping protein and the hippo pathway regulate F-actin and tissue growth in Drosophila. Development 138, 2337–2346 (2011).
Sansores-Garcia, L. et al. Modulating F-actin organization induces organ growth by affecting the Hippo pathway. EMBO J. 30, 2325–2335 (2011).
Visser-Grieve, S. et al. Lats1 tumor suppressor is a novel actin-binding protein and negative regulator of actin polymerization. Cell Res. 21, 1513–1516 (2011).
Wada, K.-I., Itoga, K., Okano, T., Yonemura, S. & Sasaki, H. Hippo pathway regulation by cell morphology and stress fibers. Development 138, 3907–3914 (2011).
Zhao, B., Li, L., Wang, L., Wang, C.-Y., Yu, J. & Guan, K.-L. Cell detachment activates the Hippo pathway via cytoskeleton reorganization to induce anoikis. Genes Dev. 26, 54–68 (2012).
Horn, T. & Boutros, M. E-RNAi: a web application for the multi-species design of RNAi reagents--2010 update. Nucleic Acids. Res. 38, W332–W339 (2010).
Acknowledgements
We thank W. Mansfield for assistance on the embryo transfer experiments and our colleagues for advice and to the Wellcome Trust for funding.
Author information
Authors and Affiliations
Contributions
All experiments were performed in M.Z.G.’s laboratory by C.Y.L. The data were analysed and interpreted by C.Y.L. and M.Z.G. The manuscript was written by C.Y.L. and M.Z.G.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1-S6 and Supplementary Tables S1-S2 (PDF 657 kb)
Rights and permissions
This article is licensed under a Creative Commons Attribution 3.0 Unported Licence. To view a copy of this licence visit http://creativecommons.org/licenses/by/3.0/.
About this article
Cite this article
Leung, C., Zernicka-Goetz, M. Angiomotin prevents pluripotent lineage differentiation in mouse embryos via Hippo pathway-dependent and -independent mechanisms. Nat Commun 4, 2251 (2013). https://doi.org/10.1038/ncomms3251
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms3251
This article is cited by
-
Tead4 and Tfap2c generate bipotency and a bistable switch in totipotent embryos to promote robust lineage diversification
Nature Structural & Molecular Biology (2024)
-
The nuclear lamina couples mechanical forces to cell fate in the preimplantation embryo via actin organization
Nature Communications (2023)
-
Combination of epigenetic erasing and mechanical cues to generate human epiBlastoids from adult dermal fibroblasts
Journal of Assisted Reproduction and Genetics (2023)
-
A monoastral mitotic spindle determines lineage fate and position in the mouse embryo
Nature Cell Biology (2022)
-
ASPP2 maintains the integrity of mechanically stressed pseudostratified epithelia during morphogenesis
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.