Abstract
Electrochemical reduction of carbon dioxide with renewable energy is a sustainable way of producing carbon-neutral fuels. However, developing active, selective and stable electrocatalysts is challenging and entails material structure design and tailoring across a range of length scales. Here we report a cobalt-phthalocyanine-based high-performance carbon dioxide reduction electrocatalyst material developed with a combined nanoscale and molecular approach. On the nanoscale, cobalt phthalocyanine (CoPc) molecules are uniformly anchored on carbon nanotubes to afford substantially increased current density, improved selectivity for carbon monoxide, and enhanced durability. On the molecular level, the catalytic performance is further enhanced by introducing cyano groups to the CoPc molecule. The resulting hybrid catalyst exhibits >95% Faradaic efficiency for carbon monoxide production in a wide potential range and extraordinary catalytic activity with a current density of 15.0 mA cm−2 and a turnover frequency of 4.1 s−1 at the overpotential of 0.52 V in a near-neutral aqueous solution.
Similar content being viewed by others
Introduction
Converting carbon dioxide (CO2) to useful products is an attractive paradigm to mitigate the environmental problems associated with atmospheric CO2 concentration increase and to simultaneously benefit energy storage and chemical production1,2,3,4. Electrocatalytic CO2 reduction is of particular interest as it can work under ambient conditions in aqueous media and is compatible with utilization of renewable energy sources such as wind and solar energy5. However, the efficiency and practicality of CO2 electroreduction is currently hindered by the lack of cost-effective electrocatalysts with high catalytic activity, selectivity and durability6.
A range of materials including metals, oxides, chalcogenides, nitrogen-doped carbons and molecular complexes have been explored for catalysing CO2 electroreduction7,8,9,10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27. Among them, metal porphyrins and metal phthalocyanines constitute an attractive class of materials with distinct advantages in easy accessibility, chemical stability and structural tunability at molecular level28,29,30,31,32. Recently, a covalent organic framework (COF) based on cobalt-porphyrin has been reported for efficiently reducing CO2 to CO in aqueous electrolyte. The catalyst exhibits a Faradaic efficiency (FE) of 90% together with an optimized initial turnover frequency (TOF) as high as 3 s−1 at an overpotential of 0.55 V (ref. 14). In another case, iron-porphyrin derivative molecules immobilized on a carbon nanotube (CNT) electrode exhibited a TOF of 144 h−1 and an FE of 93% in converting CO2 to CO at an overpotential of 0.48 V (ref. 16). Cobalt phthalocyanine (CoPc) molecules absorbed on graphite electrode are also capable to reduce CO2 to CO, but the activity and selectivity are modest13. By modification with poly-4-vinylpridine (P4VP), the catalytic performance could be enhanced33,34. A current density of 2.0 mA cm−2 and a TOF of 4.8 s−1 with an FE of 89% for CO have been demonstrated for a CoPc-P4VP system at an overpotential of 0.64 V (ref. 34). Despite these progresses, better electrocatalyst materials are still deserved to be developed.
Here, we report a combined nanoscale and molecular approach to construct CoPc-based hybrid materials as efficient electrocatalysts for CO2 reduction to CO. On the nanoscale, CoPc molecules are uniformly anchored on CNTs. At an overpotential (E°CO2/CO=−0.11 V versus the reversible hydrogen electrode (versus RHE))15 of 0.52 V in 0.1 M KHCO3 aqueous solution, the CoPc/CNT hybrid catalyst shows a high and stable current density of over 10 mA cm−2 with a FE of over 90% for CO2 reduction to CO, corresponding to a TOF of 2.7 s−1. We find that the hybridization with CNTs significantly improves not only the catalytic activity but also the product selectivity and catalytic stability as well. The catalyst material is further upgraded with molecular level structure optimization. By introducing cyano groups to the CoPc molecular structure, we realize a superior CoPc-CN/CNT hybrid catalyst which reduces CO2 to CO with a TOF of 4.1 s−1 and a FE of 96% at an overpotential of 0.52 V, representing to the best of our knowledge the most active and selective molecular-based electrocatalyst for CO2 reduction to CO so far.
Results
Synthesis and characterization of CoPc/CNT
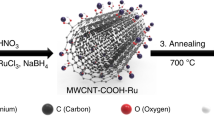
The CoPc/CNT hybrid was prepared by interacting CoPc and multi-walled CNTs in N,N-dimethyl formamide (DMF) with the assistance of sonication and magnetic stirring (see Methods for experimental details). DMF is a good solvent for dispersing CoPc and CNTs, allowing for effective anchoring of CoPc molecules on CNTs via strong π–π interactions35. Transmission electron microscopy (TEM) reveals that the morphology of the CoPc/CNT (Fig. 1a,b) resembles that of the original CNTs (Supplementary Fig. 1a,b) as nanotubular structures with an average diameter of ∼20 nm. No aggregated CoPc particles were observed. The scanning TEM image and the corresponding energy dispersive X-ray spectroscopy maps show that the distributions of C and N elements overlap and match the nanotube structures (Fig. 1c), which confirms that the CoPc molecules are uniformly dispersed on the sidewalls of the CNTs. The Co map overlaps partially with the C or N map, possibly due to the low atomic content of Co in the hybrid material. It should be pointed out that no Co signals could be detected in the original CNT sample (Supplementary Fig. 1c).
(a,b) TEM images of the CoPc/CNT(6%) hybrid. Inset in b shows a schematic representation of the CoPc/CNT hybrid. (c) STEM image of the CoPc/CNT(6%) material and the corresponding EDS maps of C, N and Co in the blue dash area. (d) Raman spectra of pure CoPc, the CoPc/CNT(6%) hybrid and pure CNTs. Scale bars, 100 nm (a); 20 nm (b); and 200 nm (c). EDS, energy dispersive X-ray spectroscopy; STEM, scanning transmission electron microscopy.
Inductively coupled plasma mass spectrometry (ICP-MS) was employed to determine the Co amount and to derive the CoPc content in the hybrid material. The Co amount was found to be 0.63 wt%, corresponding to 6.0 wt% of CoPc in the hybrid (denoted as CoPc/CNT(6%) hereafter). Raman spectroscopy was further used to characterize the CoPc/CNT hybrid (Fig. 1d). Signature vibrational peaks of CNT and CoPc can be discerned in the spectrum. It is noted that some of the CoPc vibrational features are not observed for the hybrid material, suggesting strong CoPc-CNT electronic interactions that prohibit some of the vibrational modes of the CoPc molecules on CNT. The CoPc content in the hybrid was adjusted in the range from 26 to 0.50 wt% (Supplementary Table 1). The TEM and Raman spectroscopy results of the corresponding materials are shown in Supplementary Figs 2–4. With a CoPc content of 26 wt%, wrinkled layers are clearly observed on the sidewalls of the CNTs (Supplementary Fig. 2) and the Raman spectrum shows most of the CoPc vibrational features (Supplementary Fig. 4), suggesting that CoPc aggregates have formed with such a high loading. With a CoPc loading of 2.5 wt% or lower, the CNT sidewalls appear smooth (Supplementary Fig. 3), indicating that CoPc is possibly dispersed on CNTs at molecular level.
Electrocatalytic performance of CoPc/CNT
The catalyst materials were loaded on carbon fibre paper (CFP) substrates (catalyst loading is 0.4 mg cm−2 unless otherwise mentioned). Cyclic voltammetry was first performed in a phosphate buffer solution (0.2 M, pH 7.2) saturated with Ar or CO2 (Supplementary Fig. 5). The CoPc/CNT(6%) hybrid under Ar exhibited considerable cathodic current density at potentials <−0.35 V versus RHE, which was ascribed to hydrogen evolution reaction because hydrogen was detected as the only product with a high FE. When the solution was saturated with CO2, significant current increase was observed and CO2 reduction products were detected (Supplementary Fig. 5a). In contrast, the CFP without catalyst showed much smaller current density (Supplementary Fig. 5b). These results suggest that the CoPc/CNT hybrid has significant catalytic activity for reducing CO2. Control experiments further reveal that the CoPc/CNT hybrid has much higher catalytic activity than CoPc or CNTs alone (Supplementary Fig. 5a,b). CoPc/CNT hybrids with different CoPc contents were also studied (Supplementary Fig. 6). It is found that the reduction current increases with the CoPc percentage but starts to saturate when the CoPc percentage goes over 2.5 wt%. Therefore, we focus on the CoPc/CNT(2.5%) hybrid (the cobalt content is 0.26 wt%) in the following studies.
Electrochemical CO2 reduction in a 0.1 M KHCO3 aqueous solution saturated with CO2 (pH 6.8) was performed under controlled electrode potentials. Figure 2a shows the chronoamperograms of CoPc/CNT(2.5%) at different potentials. Little current decay (<5%) after 1 h was observed at each potential. The CoPc molecular structure remains intact over the electrolysis (Supplementary Fig. 7). A high current density of >10 mA cm−2 was achieved at −0.63 V versus RHE. Gas chromatography (GC) and nuclear magnetic resonance spectroscopy were used to analyse the gas and liquid products respectively. H2 and CO were the major gas products and no liquid products could be detected (Fig. 2b). The product distribution was found to be dependent on the applied potential. At a low potential of −0.46 V versus RHE, the FE for CO production (FE(CO)) was determined to be 59±3.4%. The FE(CO) increased with larger overpotential applied, and reached over 92% at −0.59 and −0.63 V versus RHE. In contrast, CoPc directly loaded on CFP showed significantly lower current density and faster decay (Supplementary Fig. 8). The FE(CO) was only around 68% at −0.59 and −0.63 V versus RHE (Fig. 2b). For pure CNTs, the reduction current density at −0.63 V versus RHE was smaller than 0.10 mA cm−2 (Supplementary Fig. 8a), and only H2 could be detected as the reduction product at this potential. Figure 2c shows the partial current densities of the reduction products over the CoPc/CNT(2.5%) and CoPc catalysts at various potentials. The CO production rate over the CoPc/CNT is much higher than that over the CoPc directly loaded on CFP. These results indicate that CoPc/CNT exhibits not only higher catalytic activity, but also enhanced stability and product selectivity.
(a) Representative chronoamperograms of CO2 electroreduction catalysed by the CoPc/CNT(2.5%) hybrid for 1 h at various potentials in 0.1 M KHCO3 aqueous solution. (b) Faradaic efficiencies of CO2 reduction products in the gas phase for CoPc/CNT(2.5%) (red) and CoPc (blue) at various potentials. (c) Partial current densities of CO2 reduction products in the gas phase for CoPc/CNT(2.5%) (red) and CoPc (blue) at different potentials. The average values and error bars in (b,c) are based on six measurements during three reaction runs (two product analysis measurements were performed in each run). The error bars represent s.d. of six measurements. (d) Long-term stability of the CoPc/CNT(2.5%) hybrid catalyst for CO2 reduction operated at −0.63 V versus RHE for 10 h. The data are all iR corrected.
A long-term operation was conducted at −0.63 V versus RHE for the CoPc/CNT catalyst. The initial current density of ∼10 mA cm−2 was maintained for 10 h and the FE(CO) was over 90% during the entire period (Fig. 2d), corresponding to a remarkable turnover number of 97,000 for CO2 conversion to CO. The quantity of CO molecules generated is ∼3,000 times more than the total number of C atoms contained in all the CoPc molecules of the CoPc/CNT catalyst. Combined with the observation that no CO or other CO2 reduction products are detected when either CNTs or bare CFP is used as catalyst, the result unambiguously confirms that the produced CO originates from CO2.
CoPc hybridized with other forms of nano-carbons including reduced graphene oxide (RGO) and carbon black (CB) was also studied (Supplementary Table 1). Compared with CoPc/CNT(2.5%), CoPc/RGO(2.2%) and CoPc/CB(3.3%) showed less than 1/3 of the current density at −0.59 V versus RHE with ∼10% lower FE(CO) and inferior catalytic stability (Fig. 3). The results clearly reflect the advantage of CNTs in enhancing the catalytic performance. The CNT has a higher graphitic degree than either RGO or CB and is thus likely to afford better π–π interactions with CoPc and higher electron conduction36. We also measured a Pc/CNT hybrid and observed much smaller reduction current density (Fig. 3b) with a much lower FE(CO) of only 19% (Fig. 3c), indicating that the Co centres in the CoPc/CNT are the catalytically active sites. The low but non-zero conversion of CO2 to CO on Pc/CNT is attributed to the catalytic activity of Pc itself. Recent experimental and theoretical studies have found that nitrogen dopants such as pyridinic, pyrrolic and graphitic nitrogen atoms in carbon materials can catalyse CO2 electroreduction to CO (refs 12, 37). Thus, it is reasonable that the nitrogen-containing Pc supported on CNTs could reduce CO2 to CO with certain activity.
(a) Cyclic voltammograms at 5 mV s−1, (b) chronoamperograms at −0.59 V versus RHE, (c) Faradaic efficiencies of CO2 reduction products, and (d) partial current densities of CO2 reduction products for Pc/CNT, CoPc/RGO and CoPc/CB in comparison with CoPc/CNT in 0.1 M KHCO3 solution. The average values and error bars in (c,d) are based on six measurements during three reaction runs (two product analysis measurements were performed in each run). The error bars represent s.d. of six measurements. The data are all iR corrected.
Cyano-substituted CoPc hybrid
We further explored the potential of tuning the CoPc molecular structure for optimizing catalytic performance. Inspired by previous reports that electron-withdrawing substituents on metal phthalocyanine structures can increase the electrocatalytic performance for CO2 reduction to CO (refs 38, 39, 40), we synthesized cobalt-2,3,7,8,12,13,17,18-octacyano-phthalocyanine (CoPc-CN) and prepared a CoPc-CN/CNT hybrid containing 3.5 wt% of CoPc-CN (the cobalt content is 0.27 wt%, similar to that of CoPc/CNT(2.5%)) (Supplementary Fig. 9). In 0.1 M KHCO3, the CoPc-CN/CNT hybrid exhibits even larger reduction current density than the previous CoPc/CNT hybrid (Supplementary Fig. 10 and Fig. 4a). More impressively, higher selectivity for CO production at low overpotentials can be achieved with the CoPc-CN/CNT catalyst. The FE(CO) is already over 90% at −0.46 V versus RHE (Fig. 4b), compared with only 59% for the CoPc/CNT at the same potential. The FE(CO) maintains over 95% from −0.53 V to −0.63 V versus RHE (Fig. 4b). We also tested the CoPc-CN/CNT hybrid catalyst in 0.5 M KHCO3 aqueous solution. At −0.46 V versus RHE, a high current density of 5.6 mA cm−2 with a FE(CO) of 88% could be obtained (Supplementary Fig. 11).
(a) Chronoamperograms and (b) Faradaic efficiencies of reduction products at different potentials for CoPc-CN/CNT (solid line) in comparison with CoPc/CNT (dotted line). Inset in (b) shows the molecular structure of CoPc-CN, which is anchored on CNT. The average values and error bars in b are based on six measurements during three reaction runs (two product analysis measurements were performed in each run). The error bars represent s.d. of six measurements. The data are all iR corrected.
Discussion
The CoPc-CN/CNT hybrid material demonstrates outstanding catalytic performance for CO2 electroreduction to CO. At −0.63 V versus RHE in 0.1 M KHCO3, the catalyst delivers a reduction current density as high as 15.0 mA cm−2, with 98% of the electrons devoted to CO production. Assuming all the loaded CoPc-CN molecules are catalytically active (the electrochemically active coverage of the molecules could not be readily determined from the broad CV peaks), the TOF value for CO production is calculated to be 4.1 s−1, representing the lower limit of the actual TOF. The calculated TOF is slightly higher than that of other CO-selective electrocatalysts based on molecular catalytic sites (Table 1). Furthermore, our hybrid catalysts deliver much higher geometric current densities than other molecular-based catalysts under similar conditions (Table 1). At −0.46 V versus RHE in 0.5 M KHCO3, our CoPc-CN/CNT catalyst reaches 5.6 mA cm−2 with a FE(CO) of 88% (corresponding to a TOF of 1.4 s−1), which is already comparable to the most-active noble metal-based electrocatalysts for CO2 reduction to CO (Table 1). We note that the catalyst shows higher catalytic activity in 0.5 M KHCO3 than in 0.1 M KHCO3 (Supplementary Fig. 11), which is possibly due to improved mass transport of CO2 to the catalytic sites41.
A clear advantage of our CoPc/CNT and CoPc-CN/CNT hybrid materials is that they can deliver high geometrical catalytic current densities comparable to the best heterogeneous catalysts while maintaining good per-site activity comparable to the best molecular systems for CO2 electroreduction to CO42. The efficient molecule/CNT hybridization strategy allows us to realize one order of magnitude larger catalyst molecule loading (∼1.8 × 10−8 mol cm−2 for CoPc or CoPc-CN) without compromising per-molecule activity, leading to one order of magnitude increase in catalytic current density compared with the previously reported CoPc-P4VP loaded on edge-plane graphite with similar TOF34. For hybrid materials with higher CoPc contents, lower TOFs were expectedly observed due to aggregation of molecules (Supplementary Table 2).
The exceptional catalytic performance (activity, selectivity and durability) originates from the CNT hybridization on the nanoscale and the cyano substitution on the molecular level. The strong interactions between CoPc-CN (or CoPc) and CNTs allow for uniform distribution of the molecules on the highly conductive carbon support and thus enable a high degree of catalytic site exposure, beneficial for achieving high catalytic current densities. Rapid electron transfer from electrode to surface CoPc-CN (or CoPc) molecules anchored on CNTs facilitates fast repetitive cycling between Co(II) and Co(I) to support CO2 conversion to CO during the electrocatalytic process. Moreover, uniform coverage of CNTs by CoPc molecules in the CoPc/CNT catalyst material structure also minimizes exposure of carbon surface which may catalyse hydrogen evolution reaction but not CO2 reduction. All these contribute to the high selectivity of CO2 reduction over proton reduction of our hybrid catalysts43. Attachment to CNTs could also lower the possibility of molecule detachment from electrode and thus enhance catalytic durability.
It should be noted that our solution-phase hybridization strategy distinguishes from previous approaches where metal porphyrin or metal phthalocyanine molecules are drop-dried or dip-coated on electrodes pre-loaded with CNTs16,38. Such direct-drying methods may generate molecular aggregates, which harms catalytic site exposure and impedes efficient electron delivery from electrode to catalyst surface. To prove this concept, we used SEM to check the morphology of the CoPc loaded on CFP by drop-drying its ethanol dispersion, and observed obvious CoPc aggregates (Supplementary Fig. 12a). Replacing the ethanol with DMF is able to reduce the aggregation (Supplementary Fig. 12b), likely due to the improved CoPc solubility and higher boiling point of DMF, and thus increases the CO2 reduction current density (Supplementary Fig. 13). However, the catalytic performance is still substantially inferior to that of the CoPc/CNT hybrid. For the CoPc catalysts, electrons have to go through the less-conductive aggregate bulk to reach the surface molecules, which could hamper the reduction of Co(II) to Co(I). A smaller fraction of Co(I) sites on the CoPc surface and/or slower redox cycling between Co(II) and Co(I) can explain the observed lower product selectivity compared with the CoPc/CNT catalyst.
The cyano substituent on the phthalocyanine ligand is another essential contributor. The electron-withdrawing cyano groups can facilitate the formation of Co(I) which is considered as the active sites for reducing CO2 (ref. 44). This is supported by the more significant Co(II)/Co(I) redox transition observed at more positive potential for the CoPc-CN/CNT as compared with the CoPc/CNT (Supplementary Fig. 10b). Even though the cyano substituents may make the Co(I) sites less nucleophilic and thus bind CO2 less strongly, the positive shift of the Co(II)/Co(I) redox potential renders a higher fraction of Co(I) sites in the CoPc-CN/CNT catalyst than in the CoPc/CNT at low overpotentials. In the potential range (−0.46 to −0.63 V) we examined, the CoPc/CNT is only partially reduced to Co(I) (Supplementary Fig. 10b). This explains the higher current density and thus higher TOF (based on all the molecules loaded on the electrode) for the CoPc-CN/CNT hybrid catalyst. It can also be responsible for the observed higher CO selectivity for the CoPc-CN/CNT catalyst at low overpotentials. The electron-withdrawing substituents can also reduce the affinity of the cobalt centre to CO (ref. 39), which can accelerate product removal and catalytic turnover45. As a result, cyano substitution further enhances the catalytic performance on the basis of the CoPc/CNT hybrid material, which itself is already remarkably active and selective.
In conclusion, we have devised a combined nanoscale and molecular-level approach to construct easily accessible cobalt-phthalocyanine/CNT hybrid materials which catalyse electroreduction of CO2 to CO with remarkable activity, selectivity and durability in aqueous solution. The CoPc-CN/CNT shows unprecedented electrocatalytic performance, owing to the stacked effects of CNT hybridization and cyano-group substitution in the molecular structure. With the molecularly tunable phthalocyanine unit and the structurally engineerable nano-carbon support, these molecule/CNT hybrid materials represent an attractive class of electrocatalysts for converting CO2 emissions to sustainable fuels.
Methods
Chemicals
Chemicals were purchased from commercial sources and used without further purification unless otherwise noted. CoPc-CN was synthesized based on a reported method46. All aqueous solutions were prepared with Millipore water (18.2 MΩ cm). Organic solvents used were analytical grade. The CNTs were purchased from C-Nano (FT 9000). The purification of CNTs was done by calcining the CNTs at 500 °C in air for 5 h. After cooling down to room temperature, the CNTs were transferred into a 5 wt% HCl aqueous solution and sonicated for 30 min. The purified CNTs were collected by filtration and washed with ultrapure water for over 10 times. The quality of the CNTs was evaluated by Raman, SEM and TEM.
Preparation of the hybrid materials
30 mg of purified CNTs were dispersed in 30 ml of DMF with the assistance of sonication for 1 h. Then, a calculated amount of CoPc or CoPc-CN dissolved in DMF was added to the CNT suspension followed by 30 min of sonication to obtain a well-mixed suspension. The mixed suspension was further stirred at room temperature for 20 h. Subsequently, the mixture was centrifuged and the precipitate was washed with DMF for three times and ethanol twice. Finally, the precipitate was lyophilized to yield the final product. Other CoPc/nano-carbon hybrids were prepared by the same method. RGO was synthesized following a previously reported method.47,48
Material characterizations
TEM and energy dispersive X-ray spectroscopy were performed on a FEI Tecnai G2 F30 transmission electron microscope. Raman spectra were taken with Horiba LabRAM HR Evolution and Jobin Yvon LabRAM Aramis Raman spectrometers. ICP-MS was performed on an Agilent Technologies 7,700 series instrument.
Electrochemical measurements
All electrochemical measurements were conducted using a CHI 660E Potentiostat in three-electrode configuration. Catalyst ink was prepared by dispersing 2 mg of catalyst material in a mixture of 130 μl of 0.25 wt% Nafion solution and 870 μl of ethanol with the assistance of sonication. The working electrodes were prepared by drop-drying 100 μl of catalyst ink onto carbon fibre paper (AvCarb MGL190 from Fuel Cell Store) to cover an area of 0.5 cm2 (loading: 0.4 mg cm−2). The loading of other catalysts on CFP was 0.4 mg cm−2 unless otherwise mentioned. The cyclic voltammetry and chronoamperometry measurements were performed in a gas-tight two-compartment electrochemical cell with a piece of glass frit as the separator (Supplementary Fig. 14). A 1 cm2 piece of platinum gauze was used as the counter electrode. Unless otherwise stated, the electrolyte was 0.1 or 0.5 M KHCO3 solution saturated with CO2 (pH 6.8 or 7.2). All potentials were measured against an Ag/AgCl reference electrode and converted to RHE scale based on Nernst equation. In the electrochemical measurements, iR corrections were made to assess the activity and selectivity of the catalyst under actual electrode potentials, so that the catalytic performance of different catalyst materials could be compared on the same bias42. The uncorrected potentials are listed in Supplementary Table 3. During constant-potential electrolysis, high-purity CO2 gas (99.999%) was delivered into the cathodic compartment at a flow rate of 5 s.c.c.m. to convey the gas products into the gas-sampling loop of a gas chromatograph (GC, SRI Instruments) for analysing the gas products. The reported TOFs and Faradaic efficiencies are average values based on three reaction runs with each containing two GC measurements (a GC measurement was initiated every 30 min). The reported cyclic voltammograms and chronoamperograms are representative data for these runs. The GC was equipped with a packed Molecular Sieve 5 A capillary column and a packed HaySep D column. Helium (99.999%) was used as the carrier gas. A helium ionization detector (HID) was used to quantify H2 and CO concentrations. The partial current density of CO production was calculated from the GC peak area as follows:


where α and β are conversion factors for CO and H2, respectively, determined from the calibration of the GC with standard samples, p=1.013 bar and T=293.15 K.
Data availability
The data that support the findings of this study are available within the paper and its Supplementary Information file or are available from the corresponding authors upon request.
Additional information
How to cite this article: Zhang, X. et al. Highly selective and active CO2 reduction electrocatalysts based on cobalt phthalocyanine/carbon nanotube hybrid structures. Nat. Commun. 8, 14675 doi: 10.1038/ncomms14675 (2017).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Aresta, M. & Dibenedetto, A. Utilisation of CO2 as a chemical feedstock: opportunities and challenges. Dalton Trans. 36, 2975–2992 (2007).
Anagnostou, E. et al. Changing atmospheric CO2 concentration was the primary driver of early Cenozoic climate. Nature 533, 380–384 (2016).
Sakakura, T., Choi, J.-C. & Yasuda, H. Transformation of carbon dioxide. Chem. Rev. 107, 2365–2387 (2007).
Williamson, P. Scrutinize CO2 removal methods. Nature 530, 153–155 (2016).
Kuhl, K. P. et al. Electrocatalytic conversion of carbon dioxide to methane and methanol on transition metal surfaces. J. Am. Chem. Soc. 136, 14107–14113 (2014).
Jones, J. P., Prakash, G. & Olah, G. A. Electrochemical CO2 reduction: recent advances and current trends. Isr. J. Chem. 54, 1451–1466 (2014).
Qiao, J., Liu, Y., Hong, F. & Zhang, J. A review of catalysts for the electroreduction of carbon dioxide to produce low-carbon fuels. Chem. Soc. Rev. 43, 631–675 (2014).
Zhu, C. & Dong, S. Recent progress in graphene-based nanomaterials as advanced electrocatalysts towards oxygen reduction reaction. Nanoscale 5, 1753–1767 (2013).
Kim, C. et al. Achieving selective and efficient electrocatalytic activity for CO2 reduction using immobilized silver nanoparticles. J. Am. Chem. Soc. 137, 13844–13850 (2015).
Sarfraz, S., Garcia-Esparza, A. T., Jedidi, A., Cavallo, L. & Takanabe, K. Cu–Sn bimetallic catalyst for selective aqueous electroreduction of CO2 to CO. ACS Catal. 6, 2842–2851 (2016).
Chen, Y., Li, C. W. & Kanan, M. W. Aqueous CO2 reduction at very low overpotential on oxide-derived Au nanoparticles. J. Am. Chem. Soc. 134, 19969–19972 (2012).
Wu, J. et al. Achieving highly efficient, selective, and stable CO2 reduction on nitrogen-doped carbon nanotubes. ACS Nano 9, 5364–5371 (2015).
Lieber, C. M. & Lewis, N. S. Catalytic reduction of carbon dioxide at carbon electrodes modified with cobalt phthalocyanine. J. Am. Chem. Soc. 106, 5033–5034 (1984).
Lin, S. et al. Covalent organic frameworks comprising cobalt porphyrins for catalytic CO2 reduction in water. Science 349, 1208–1213 (2015).
Asadi, M. et al. Nanostructured transition metal dichalcogenide electrocatalysts for CO2 reduction in ionic liquid. Science 353, 467–470 (2016).
Maurin, A. & Robert, M. Noncovalent immobilization of a molecular iron-based electrocatalyst on carbon electrodes for selective, efficient CO2-to-CO conversion in water. J. Am. Chem. Soc. 138, 2492–2495 (2016).
Li, C. W. & Kanan, M. W. CO2 reduction at low overpotential on Cu electrodes resulting from the reduction of thick Cu2O films. J. Am. Chem. Soc. 134, 7231–7234 (2012).
Rosen, J. et al. Electrodeposited Zn dendrites with enhanced CO selectivity for electrocatalytic CO2 reduction. ACS Catal. 5, 4586–4591 (2015).
Gao, D. et al. Size-dependent electrocatalytic reduction of CO2 over Pd nanoparticles. J. Am. Chem. Soc. 137, 4288–4291 (2015).
Asadi, M. et al. Robust carbon dioxide reduction on molybdenum disulphide edges. Nat. Commun. 5, 4470 (2014).
Varela, A. S. et al. Metal-doped nitrogenated carbon as an efficient catalyst for direct CO2 electroreduction to CO and hydrocarbons. Angew Chem. Int. Ed. Engl. 54, 10758–10762 (2015).
Kumar, B. et al. Renewable and metal-free carbon nanofibre catalysts for carbon dioxide reduction. Nat. Commun. 4, 2819 (2013).
Studt, F. et al. Discovery of a Ni-Ga catalyst for carbon dioxide reduction to methanol. Nat. Chem. 6, 320–324 (2014).
Torelli, D. A. et al. Nickel-gallium-catalyzed electrochemical reduction of CO2 to highly reduced products at low overpotentials. ACS Catal. 6, 2100–2104 (2016).
Kas, R. et al. Three-dimensional porous hollow fibre copper electrodes for efficient and high-rate electrochemical carbon dioxide reduction. Nat. Commun. 7, 10748 (2016).
Shen, J. et al. Electrocatalytic reduction of carbon dioxide to carbon monoxide and methane at an immobilized cobalt protoporphyrin. Nat. Commun. 6, 8177 (2015).
Kim, D., Resasco, J., Yu, Y., Asiri, A. M. & Yang, P. Synergistic geometric and electronic effects for electrochemical reduction of carbon dioxide using gold–copper bimetallic nanoparticles. Nat. Commun. 5, 4948 (2014).
Takahashi, K., Hiratsuka, K., Sasaki, H. & Toshima, S. Electrocatalytic behavior of metal porphyrins in the reduction of carbon dioxide. Chem. Lett. 8, 305–308 (1979).
Meshitsuka, S., Ichikawa, M. & Tamaru, K. Electrocatalysis by metal phthalocyanines in the reduction of carbon dioxide. J. Chem. Soc. Chem. Commun. 5, 158–159 (1974).
Kornienko, N. et al. Metal–organic frameworks for electrocatalytic reduction of carbon dioxide. J. Am. Chem. Soc. 137, 14129–14135 (2015).
Costentin, C., Drouet, S., Robert, M. & Savéant, J.-M. A local proton source enhances CO2 electroreduction to CO by a molecular Fe catalyst. Science 338, 90–94 (2012).
Weng, Z. et al. Electrochemical CO2 reduction to hydrocarbons on a heterogeneous molecular Cu catalyst in aqueous solution. J. Am. Chem. Soc. 138, 8076–8079 (2016).
Yoshida, T. et al. Selective electrocatalysis for CO2 reduction in the aqueous-phase using cobalt phthalocyanine/poly-4-vinylpyridine modified electrodes. J. Electroanal. Chem. 385, 209–225 (1995).
Kramer, W. W. & McCrory, C. C. L. Polymer coordination promotes selective CO2 reduction by cobalt phthalocyanine. Chem. Sci. 7, 2506–2515 (2016).
Tasis, D., Tagmatarchis, N., Bianco, A. & Prato, M. Chemistry of carbon nanotubes. Chem. Rev. 106, 1105–1136 (2006).
Liang, Y., Li, Y., Wang, H. & Dai, H. Strongly coupled inorganic/nanocarbon hybrid materials for advanced electrocatalysis. J. Am. Chem. Soc. 135, 2013–2036 (2013).
Sharma, P. P. et al. Nitrogen-doped carbon nanotube arrays for high-efficiency electrochemical reduction of CO2: on the understanding of defects, defect Density, and selectivity. Angew Chem. Int. Ed. Engl. 54, 13701–13705 (2015).
Manbeck, G. F. & Fujita, E. A review of iron and cobalt porphyrins, phthalocyanines and related complexes for electrochemical and photochemical reduction of carbon dioxide. J. Porphyrins Phthalocyanines 19, 45–64 (2015).
Morlanés, N., Takanabe, K. & Rodionov, V. Simultaneous reduction of CO2 and splitting of H2O by a single immobilized cobalt phthalocyanine electrocatalyst. ACS Catal 6, 3092–3095 (2016).
Abe, T. et al. Electrochemical CO2 reduction catalysed by cobalt octacyanophthalocyanine and its mechanism. J. Porphyrins Phthalocyanines 1, 315–321 (1997).
Min, X. & Kanan, M. W. Pd-catalyzed electrohydrogenation of carbon dioxide to formate: High mass activity at low overpotential and identification of the deactivation pathway. J. Am. Chem. Soc. 137, 4701–4708 (2015).
Stevens, M. B. et al. Measurement techniques for the study of thin film heterogeneous water oxidation electrocatalysts. Chem. Mater. 29, 120–140 (2016).
Hod, I., Farha, O. K. & Hupp, J. T. Electrocatalysis powered by porphyrin packing. Nat. Mater. 14, 1192–1193 (2015).
Leung, K., Nielsen, I. M. B., Sai, N., Medforth, C. & Shelnutt, J. A. Cobalt−porphyrin catalyzed electrochemical reduction of carbon dioxide in water. 2. Mechanism from first principles. J. Phys. Chem. A 114, 10174–10184 (2010).
Froehlich, J. D. & Kubiak, C. P. The homogeneous reduction of CO2 by [Ni(cyclam)]+: increased catalytic rates with the addition of a CO scavenger. J. Am. Chem. Soc. 137, 3565–3573 (2015).
Kim, S. J., Matsumoto, M. & Shigehara, K. Synthesis and electrical properties of one-dimensional octacyanometallophthalocyanine (M≡Fe, Co) polymers. J. Porphyrins Phthalocyanines 4, 136–144 (2000).
Liang, Y. Y. et al. Co3O4 nanocrystals on graphene as a synergistic catalyst for oxygen reduction reaction. Nat. Mater. 10, 780–786 (2011).
Jiang, Y. Y. et al. Enhanced catalytic performance of Pt-free iron phthalocyanine by graphene support for efficient oxygen reduction reaction. ACS Catal 3, 1263–1271 (2013).
Tatin, A. et al. Efficient electrolyzer for CO2 splitting in neutral water using earth-abundant materials. Proc. Natl Acad. Sci. USA 113, 5526–5529 (2016).
Zhu, W. et al. Active and selective conversion of CO2 to CO on ultrathin Au nanowires. J. Am. Chem. Soc. 136, 16132–16135 (2014).
Lu, Q. et al. A selective and efficient electrocatalyst for carbon dioxide reduction. Nat. Commun. 5, 3242 (2014).
Acknowledgements
Z.W. and H.W. acknowledge funding support from Yale University and the Global Innovation Initiative from Institute of International Education. Y.L. acknowledges financial supports from ‘The Recruitment Program of Global Youth Experts of China’, Shenzhen fundamental research funding (JCYJ20160608140827794), Shenzhen Key Lab funding (ZDSYS201505291525382) and Peacock Plan (KQTD20140630160825828).
Author information
Authors and Affiliations
Contributions
Y.L. and H.W. conceived the project and designed the experiments. Xing Z., Z.W., L.L., Xiao Z., Y.Li., H.X., X.Li., X.Y., Z.Z. carried out the synthesis, material characterizations and electrocatalytic measurements. Y.L., H.W., Xing Z. and L.L. analysed the data and wrote the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures and Supplementary Tables (PDF 1525 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, X., Wu, Z., Zhang, X. et al. Highly selective and active CO2 reduction electrocatalysts based on cobalt phthalocyanine/carbon nanotube hybrid structures. Nat Commun 8, 14675 (2017). https://doi.org/10.1038/ncomms14675
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms14675
This article is cited by
-
Spatial engineering of single-atom Fe adjacent to Cu-assisted nanozymes for biomimetic O2 activation
Nature Communications (2024)
-
Potential-driven structural distortion in cobalt phthalocyanine for electrocatalytic CO2/CO reduction towards methanol
Nature Communications (2024)
-
The solvation environment of molecularly dispersed cobalt phthalocyanine determines methanol selectivity during electrocatalytic CO2 reduction
Nature Catalysis (2024)
-
CO2-to-methanol electroconversion on a molecular cobalt catalyst facilitated by acidic cations
Nature Catalysis (2024)
-
Electrochemical CO2 reduction passes the acid test
Nature Nanotechnology (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.