Abstract
Catalytic oxidative C–H bond functionalization reactions that proceed without requiring stoichiometric amounts of external oxidants or pre-functionalized oxidizing reagents could maximize the atom- and step-economy in chemical syntheses. However, such a transformation remains elusive. Here, we report that a photo-driven catalytic process enables decarboxylative C–H trifluoromethylation of (hetero)arenes with trifluoroacetic acid as a trifluoromethyl source in good yields in the presence of an external oxidant in far lower than stoichiometric amounts (for example, 0.2 equivalents of Na2S2O8) using Rh-modified TiO2 nanoparticles as a photocatalyst, in which H2 release is an important driving force for the reaction. Our findings not only provide an approach to accessing valuable decarboxylative C–H trifluoromethylations via activation of abundant but inert trifluoroacetic acid towards oxidative decarboxylation and trifluoromethyl radical formation, but also demonstrate that a photo-driven catalytic process is a promising way to achieve external oxidant-free C–H functionalization reactions.
Similar content being viewed by others
Introduction
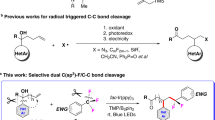
C–H bond functionalization, due to its simplified synthetic routes and readily available starting materials, provides a potentially atom- and step-economic synthetic approach complementary to the conventional functionality-based synthetic methods1,2,3,4,5. The potential power of the C–H functionalization strategy in organic synthesis has been clearly demonstrated by its application to natural product synthesis6, medicine synthesis7,8, late-stage functionalization of complex molecules9,10 and material preparations11. Full realization of the intrinsic advantage of the atom- and step-economy of C–H functionalization reactions, however, remains a significant challenge because such reactions generally require either stoichiometric amounts of external oxidants (Fig. 1a) such as metal salt oxidants or oxidizing reactants that are often prepared via multistep procedures (Fig. 1b). Natural photosynthesis, a process that enables photo-driven water splitting to molecular dioxygen and atomic hydrogen via coupling of the water oxidation half-reaction with the proton reduction half-reaction12, offers a blueprint for the design of oxidative C–H functionalization reactions that proceed without reliance on any external oxidant or oxidizing reactant.
(a) Minisci reaction: the example of the C–H functionalization with stoichiometric external oxidant. (b) C–H trifluoromethylation with Umemoto reagent: the example of C–H functionalization with pre-synthesized oxidazing reagent. (c) Photo-driven C–H trifluoromethylation without the reliance on external oxidant (this work).
On the other hand, the importance of trifluoromethyl (CF3) group-containing compounds in pharmaceuticals, agrochemicals and materials has spurred tremendous efforts devoted to the development of methods for the incorporation of trifluoromethyl group into molecular frameworks and the discoveries of trifluoromethylating reagents13. As a result of the inventions of a diverse range of trifluoromethylating reagents including nucleophilic ones (CF3−) and electrophilic ones (CF3+), a variety of metal-catalysed direct C–H trifluoromethylations have been achieved14,15,16,17,18. Benefiting from the identification of various precursors that are capable of generating trifluoromethyl radicals through regular oxidation19 or photoredox catalysis20 under mild conditions, the radical-based reactions have evolved into an efficient approach to construct C-CF3 bonds. In spite of these remarkable advances, the commonly used trifluoromethylating reagents are very expensive due to their time-consuming and multistep syntheses, which prompt chemists to develop readily available, cost-effective surrogates of existing trifluoromethylating reagents.
Herein, we report a photo-driven decarboxylative C–H trifluoromethylation of nonactivated (hetero)arenes with readily available, low-cost trifluoroacetic acid as a trifluoromethylating reagent. Using Rh-modified TiO2 NPs as a catalyst, our reaction can occur in the absence of external oxidant to afford a trifluoromethylation product in a modest yield with H2 release as a driving force for the reaction. Our findings demonstrate that the underlying redox chemistry of natural photosynthesis can operate in C–H functionalization reactions, which may provide an opportunity to maximize the step- and atom-economy of such reactions.
Results
Reaction design
Minisci reactions enable the use of alkyl carboxylic acids for C–H alkylation of electron-deficient pyridines via oxidative decarboxylation of alkyl carboxylic acids21. Recently, Minisci reaction of aryl carboxylic acids has been realized by us22. Minisci reactions have implied that trifluoroacetic acid (TFA) may act as a trifluoromethylating reagent via oxidative decarboxylation to generate trifluoromethyl radical23,24,25. In terms of its stability, ready availability and low price, trifluoroacetic acid is an intrinsically advantageous trifluoromethylating reagent. The extraordinary inertness of trifluoroacetic acid (electro-oxidation of TFA anion to generate trifluoromethyl radical occurs at potentials higher than +2.24 V versus the saturated calomel electrode (SCE)26), however, largely retards its application to the trifloromethylation reactions. For example, the decarboxylative C–H trifluoromethylation mediated by a stoichiometric amount silver salt required 2 equivalents of K2S2O8 as oxidant and tolerated only a few electron-deficient substituted benzenes25.
A recent report on using trifluoroacetic anhydride, a derivative of trifluoroacetic acid, for the C–H trifluoromethylation provides an alternative solution for the inexpensive trifluoromethyl source27, which, however, requires the use of stoichiometric pyridine N-oxide as a sacrificial oxidant.
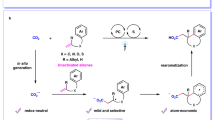
TiO2-based NPs are extensively used as photocatalysts for the photo-driven water splitting processes28. Significant redox potential (+3.1 V versus SCE)29 of the photo-generated hole in the valence band of TiO2, in principle, would make it capable of oxidizing trifluoroacetic acid to generate trifluoromethyl radicals via decarboxylation and therefore trigger the trifluoromethylation reaction. Accordingly, we envisioned that TiO2-based NPs could catalyse the photo-driven decarboxylative C–H trifluoromethylation reaction with trifluoroacetic acid as a trifluoromethyl source (Fig. 1c). More importantly, the similarity to the photocatalytic water splitting process also implies the possibility that this decarboxylative C–H trifluoromethylation reaction proceeds without the need for the stoichiometric amount of external oxidants. Only one example of TiO2-promoted photo-driven decarboxylative C–H trifluoromethylation reaction of benzenes in trifluoroacetic acid that required stoichiometric amount of silver trifluoroacetate as an oxidant and gave poor yields indicates that there is significant room to improve the photocatalyst23.
Establishment of photo-driven trifluoromethylation
We started our investigations by testing the trifluoromethylation of benzene with trifluoroacetic acid as a trifluoromethyl source. Numerous trials were performed to screen the reaction parameters such as different solvents, TiO2-based photocatalysts, light sources and external oxidants (details are provided in Supplementary Table 1). Selected results are presented in Table 1. Most strikingly, in this process C–H trifluoromethylation can happen with no sacrificial oxidant (entries 1 and 2), with very weak oxidant (entry 3), as well as with the dosage of external oxidant far less than the stoichiometric amount (entry 4).
When the reaction was conducted at room temperature under near-ultraviolet light irradiation from 365 nm Hg lamp (250 W), with 20 mol% commercial available anatase TiO2 NPs with a diameter of 60 nm as the photocatalyst, 6% yield was observed in the absence of external oxidant (entry 1). This reaction was accompanied by H2 gas formation, indicating that coupling of the photo-generated electrons with the protons released from decarboxylative C–H trifluoromethylation reaction occurred. The catalytic efficiency of the anatase TiO2 NPs could be enhanced by loading rhodium metal NPs of 0.1 wt % (relative to the amount of TiO2) onto their surface, which gave rise to 15% yield of trifluoromethylated benzene (entry 2). Interestingly, by employing 2 equivalences of Na2SO4 as additives, the reaction occurred smoothly to give the desired product in 51% yield (entry 3). Introduction of substoichiometric external oxidants, such as 0.2 equivalents of Na2S2O8 could increase the reaction yield to 70% (entry 4). The attempt to use visible light in place of ultraviolet light failed to give the satisfied result because of the wide band gap of TiO2 (entry 5). We also found that introducing one equivalence of CF3COONa into the reaction system almost shut down this reaction (entry 6), which presumably arose from the difference in substrate-TiO2 surface interaction between trifluoroacetic acid and CF3COONa. Moreover, control experiments revealed that both anatase TiO2 photocatalyst and ultraviolet light are necessary for the trifluoromethylation reaction to occur (entries 7 and 8).
Substrate scope and gram-scale reations
With the established reaction conditions for the photo-driven decarboxylative C–H trifluoromethylation, we evaluated substrate scope of substituted benzenes in the presence of substoichiometric amount of oxidant (0.1–0.4 equivalences of Na2S2O8), using trifluoroacetic acid as a trifluoromethylating reagent (for more information, see Supplementary Methods and Supplementary Fig. 53). As shown in Fig. 2, this developed C–H trifluoromethylation reaction was compatible with a broad range of functional groups, such as halogen (2b-2e), keto (2f, 2g), ester (2h, 2i) and nitrile (2j) groups. Interestingly, ultraviolet light-sensitive C-I bond remained intact during the course of C–H trifluoromethylation (2e). And benzoic acid, which is prone to oxidative decarboxylation under oxidative conditions, could undergo trifluoromethylation reaction, albeit in a diminished yield (2l). Disubstituted benzenes (2m, 2n and 2o) could also be trifluoromethylated in synthetically useful yields. Moreover, a variety of nitrogen-containing heteroarenes, such as pyridines (2p, 2q), pyrimidine (2r), pyrazolinopyrimidine (2w) and biologically active xanthines (2s-2v) were amenable to this trifluoromethylation reaction, affording a satisfying outcome. Importantly, the reactions of these nitrogen-containing heteroarenes exhibited a high level of position selectivity, and could be scaled up for the gram-scale synthesis as exemplified by the reactions of caffeine and theophylline (2s, 2t), highlighting the practicality of this approach. In the established reaction, the use of 15 ml trifluoroacetic acid as a solvent may result from the need for sufficient light-absorbing surface to obtain satisfying yields presumably due to relative low concentration of catalytic active centres of heterogeneous TiO2-based nanocatalyst. Despite this, excess TFA in this reaction is reusable by means of easy distillation for its purification and collection.
The established reaction conditions for arene trifluoromethylation were applicable to the reaction of iodine with trifluoroacetic acid to afford trifluoromethyl iodide (CF3I), a commonly used trifluoromethylating reagent13, in a 70% yield (Fig. 3a and Supplementary Fig. 1), which provides an operationally simple approach to the efficient synthesis of CF3I.
(a) Synthesis of CF3I via decarboxylative coupling of I2 with TFA. (b) Capture of CF3 radical by TEMPO. (c) Determination of CF3H and C2F6 side products in trifluoromethylation of caffeine. (d) Determination of H2 and CO2 from the photo-driven decarboxylation of TFA. (e) The proposed reaction mechanism.
Mechanistic insights and nanocatalyst characterization
Although the position selectivity of this decarboxylative trifluoromethylation reaction suggested that the reaction would proceed via trifluoromethyl radical formation, further experiments were still required to gain insights into reaction mechanism. As shown in Fig. 3b, on adding 2,2,6,6-tetramethyl-1-piperidinyloxy (TEMPO), a well-known radical scavenger, to trifluoroacetic acid solution under standard photoredox conditions, the trifluoromethyl radicals generated from decarboxylation of trifluoroacetic acid was trapped with TEMPO to form TEMPO-CF3 adduct that was confirmed by 19F NMR spectroscopy (Supplementary Fig. 2).
In addition, both trifluoromethane (CF3H) and hexafluoroethane (C2F6) were also observed as side-products during the trifluoromethylation reaction of caffeine (Fig. 3c and Supplementary Figs 3 and 4), providing further evidence to support that trifluoromethyl radical is the intermediate of the decarboxylative trifluoromethylation reaction. Furthermore, formations of both H2 and CO2 were observed when trifluoroacetic acid was subjected to the standard reaction conditions in the absence of any arene reactant (Fig. 3d and Supplementary Figs 5 and 6), which is in accord with the aforementioned observation in the decarboxylative trifluoromethylation of benzene. The fact that the oxidative decarboxylation of trifluoroacetic acid is concurrent with H2 formation strongly suggested that the redox chemistry of the photo-driven water splitting to H2 and O2 operates in the decarboxylative C–H trifluoromethylation reaction with trifluoacetic acid as a trifluoromethyl source.
On the basis of mechanistic investigation, a possible reaction pathway was proposed for the TiO2-catalysed photo-driven decarboxylative trifluoromethylation with trifluoroacetic acid as trifluoromethyl source (Fig. 3e). Initially, the oxidation of trifluoroacetic acid by the photo-generated hole in the valence band of TiO2 leads to rapid decarboxylation to form trifluoromethyl radical with carbon dioxide and proton released. The resultant trifluoromethyl radical is then intercepted by benzene to produce trifluoromethyl benzene radial. Finally, this trifluoromethyl benzene radical undergoes oxidation through a single-electron transfer process and subsequent deprotonation to afford a trifluoromethylated benzene. Meanwhile, the released protons accept the photo-generated electron to release hydrogen gas, a process similar to the photo-driven water splitting to H2 evolution30. Alternatively, the external oxidant introduced into the reaction system can also accept the photo-generated electron to drive the reaction. Consequent, combination of contributions of proton and a substoichiometric amount of external oxidant provided good yields for the photo-driven decarboxylative trifluoromethylation reaction.
The photocatalyst used in this reaction, that is, the Rh-modified TiO2 NPs, were structurally characterized. The inductively coupled plasma mass spectrometry revealed that this Rh/TiO2 photocatalyst contained Rh content of 0.1 wt%. The powder X-ray diffraction pattern of this nanocatalyst could be exclusively indexed to anatase-phase TiO2 (Supplementary Fig. 7). The transmission electron microscopy (TEM) image (Fig. 4a) and high-resolution TEM image (Fig. 4b) showed that the metallic Rh NPs with an average size of 2.04 nm (Supplementary Figs 8–10) was located on the surface of anatase NPs of about 60 nm. Besides, X-ray photoelectron spectroscopy indicated the signals of Rh 3d5/2 and 3d3/2 with binding energies of 306.42 and 311.27 eV, typical of Rh (0), further verifying that Rh existed in the form of metallic Rh (Supplementary Figs 11–15)31.
Discussion
On the basis of the characterization of the Rh-modified anatase TiO2 NPs, we attempt to rationalize the photocatalysis efficiency enhancement associated with the attachment of metallic Rh NPs onto the surface of anatase TiO2 NPs. Presumably, in the Rh-modified anatase NPs, the difference in Fermi levels between metal Rh and semiconductor TiO2 may give rise to the migration of photo-generated electron from TiO2 with higher Fermi level (the work function of TiO2 is 4.8 eV) to Rh with lower Fermi level (the work function of Rh is 5.6 eV) until equilibrium alignment of two Femi levels32. As a result, the Schottky barrier is created at the interface between Rh and TiO2 NPs33. Such a barrier may act as an effective trap for electron capture, and prevent from recombination between photo-generated electron/hole pairs, which eventually leads to the improvement of catalytic efficiency (shown in Supplementary Fig. 16).
In this work, we have developed a photo-driven C–H trifluoromethylation reaction that enables the use of readily available and cheap trifluoroacetic acid as a trifluoromethyl source for high-valuable trifluoromethylation reactions. Importantly, such a photo-driven reaction could proceed with the dosage of external oxidant far less than the stoichiometric amount or even without any external oxidant, in which H2 release provided an essential driven force for reaction to occur. This reaction demonstrates that the principle underlying the photo-driven water splitting to H2 and O2 is applicable to C–H functionalization reaction, which will inspire the development of photocatalysts to improve efficiencies of both utility of light and coupling of the proton from C–H activation process with the photo-generated electron and ultimately achieve general approach to external oxidant-free C–H functionalization reactions34.
Methods
Preparation of 0.1 wt% Rh-modified anatase TiO2 nanopaticles
In a 30 ml quartz round bottom flask, a mixture of 60 nm anatase TiO2 (Aladdin reagent, 3.0 g) and RhCl3·xH2O (0.023 g, Rh content of 39%) was deoxygenated and filled with N2, followed by addition of deoxygenated methanol (30 ml). The suspension was stirred with an irradiation of 250 W high-pressure Hg lamp (365 nm ultraviolet) for 48 h. After illumination, nanocatalysts were separated from the solution by centrifugation (8,000 r.p.m., 2 min, 298 K). The separated nanocatalysts were washed for three times by ethanol and three times by water, dried in vacuum at 298 K for 8 h. The Rh content of as-prepared nanocatalyst was 0.1 wt% by inductively coupled plasma analysis.
General procedure for photo-driven C–H trifluoromethylation
The photo-driven C–H trifluoromethylation reaction of caffeine is used as an example. Photocatalyst (0.016 g, 0.2 mmol), caffeine (0.0971, g, 0.5 mmol) and Na2S2O8 (0.0238, g, 0.1 mmol) were introduced into a Schlenk tube. Then, the tube was fitted with a rubber septum. After evacuation and N2 backfill for three times, distilled trifluoroacetic acid (15 ml) was added to the Schlenk tube through the rubber septum using syringes, and the rubber septum was replaced by a Teflon cap under N2 flow. The reaction was performed under illumination of 250 W high-pressure Hg lamp (365 nm ultraviolet) at room temperature for 48 h. After reaction, the trifluoroacetic acid was removed under reduced pressure and the residue was purified by flash chromatography on silica gel (eluent: EtOAc/hexanes) to provide the corresponding product. The yield were also determined by 19F NMR spectrum using 1-methoxy-4-(trifluoromethoxy)benzene (76 μl, 0.5 mmol, δ–58.4 p.p.m.) as an internal reference. For NMR spectra of the compounds in this article, see Supplementary Figs 17–52.
Data availability
The data supporting this study are available from the corresponding author upon reasonable request.
Additional information
How to cite this article: Lin, J. et al. Photo-driven redox-neutral decarboxylative carbon-hydrogen trifluoromethylation of (hetero)arenes with trifluoroacetic acid. Nat. Commun. 8, 14353 doi: 10.1038/ncomms14353 (2017).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Hartwig, J. F. Evolution of C–H bond functionalization from methane to methodology. J. Am. Chem. Soc. 138, 2–24 (2016).
Chen, X., Engle, K. M., Wang, D.-H. & Yu, J.-Q. Palladium(II)-catalyzed C–H activation/C-C cross-coupling reactions: versatility and practicality. Angew Chem. Int. Ed. Engl. 48, 5094–5115 (2009).
Dick, A. R. & Sanford, M. S. Transition metal catalyzed oxidative functionalization of carbon–hydrogen bonds. Tetrahedron 62, 2439–2463 (2006).
Verma, S., Baig, R. B. N., Hanb, C., Nadagouda, M. N. & Varma, R. S. Magnetic graphitic carbon nitride: application in C–H activation of amines. Chem. Commun. 51, 15554–15557 (2015).
Verma, S., Baig, R. B. N., Hanb, C., Nadagouda, M. N. & Varma, R. S. Oxidative esterification via photocatalytic C–H activation. Green Chem. 18, 251–254 (2016).
Chen, K. & Baran, P. S. Total synthesis of eudesmane terpenes by site-selective C–H oxidations. Nature 459, 824–828 (2009).
Tsai, A. S., Bergman, R. G. & Ellman, J. A. Asymmetric synthesis of (-)-incarvillateine employing an intramolecular alkylation via Rh-catalyzed olefinic C–H bond activation. J. Am. Chem. Soc. 130, 6316–6317 (2008).
Kang, T., Kim, Y., Lee, D., Wang, Z. & Chang, S. Iridium-catalyzed intermolecular amidation of sp3 C–H bonds: late-stage functionalization of an unactivated methyl group. J. Am. Chem. Soc. 136, 4141–4144 (2014).
Simmons, E. M. & Hartwig, J. F. Catalytic functionalization of unactivated primary C–H bonds directed by an alcohol. Nature 483, 70–73 (2012).
Chen, M. S. & White, M. C. A predictably selective aliphatic C–H oxidation reaction for complex molecule synthesis. Science 318, 783–787 (2007).
Zhang, Q., Kawasumi, K., Segawa, Y., Itami, K. & Scott, L. T. Palladium-catalyzed C–H activation taken to the limit. flattening an aromatic bowl by total arylation. J. Am. Chem. Soc. 134, 15664–15667 (2012).
Barber, J. Photosynthetic energy conversion: natural and artificial. Chem. Soc. Rev. 38, 185–196 (2009).
Furuya, T., Kamlet, A. S. & Ritter, T. Catalysis for fluorination and trifluoromethylation. Nature 473, 470–477 (2011).
Ball, N. D., Kampf, J. W. & Sanford, M. S. Aryl-CF(3) bond-forming reductive elimination from palladium(IV). J. Am. Chem. Soc. 132, 2878–2879 (2010).
Wang, X., Truesdale, L. & Yu, J. Q. Pd(II)-catalyzed ortho-trifluoromethylation of arenes using TFA as a promoter. J. Am. Chem. Soc. 132, 3648–3649 (2010).
Chu, L. & Qing, F. L. Copper-catalyzed direct C–H oxidative trifluoromethylation of heteroarenes. J. Am. Chem. Soc. 134, 1298–1304 (2012).
Cho, E. J. et al. The palladium-catalyzed trifluoromethylation of aryl chlorides. Science 328, 1679–1681 (2010).
Jarrige, L. et al. Photoredox-catalyzed three-component tandem process: an assembly of complex trifluoromethylated phthalans and isoindolines. Org. Lett. 18, 2906–2909 (2016).
Fujiwara, Y. et al. Practical and innate carbon-hydrogen functionalization of heterocycles. Nature 492, 95–99 (2012).
Nagib, D. A. & MacMillan, D. W. Trifluoromethylation of arenes and heteroarenes by means of photoredox catalysis. Nature 480, 224–228 (2011).
Duncton, M. A. J. Minisci reactions: versatile CH-functionalizations for medicinal chemists. Med. Chem. Commun. 2, 1135–1161 (2011).
Kan, J., Huang, S., Lin, J., Zhang, M. & Su, W. Silver-catalyzed arylation of (hetero)arenes by oxidative decarboxylation of aromatic carboxylic acids. Angew Chem. Int. Ed. Engl. 54, 2199–2203 (2015).
Lai, C. & Mallouk, T. E. A new approach to the photochemical trifluoromethylation of aromatic compounds. J. Chem. Soc. Chem. Commun. 17, 1359–1361 (1993).
Tanabe, Y., Matsuo, N. & Ohno, N. Direct perfluoroalkylation including trifluoromethylation of aromatics with perfluoro carboxylic acids mediated by xenon difluoride. J. Org. Chem. 53, 4582–4585 (1988).
Shi, G., Shao, C., Pan, S., Yu, J. & Zhang, Y. Silver-catalyzed C–H trifluoromethylation of arenes using trifluoroacetic acid as the trifluoromethylating reagent. Org. Lett. 17, 38–41 (2015).
Andreev, V. N. et al. Anodic trifluoromethylation of 10-undecylenic acid. Russ. J. Electrochem. 49, 996–1000 (2013).
Beatty, J. W., Douglas, J. J., Cole, K. P. & Stephenson, C. R. J. A scalable and operationally simple radical trifluoromethylation. Nat. Commun. 6, 7919 (2015).
Fujishima, A. & Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 238, 37–38 (1972).
Fox, M. A. Organic heterogeneous photocatalysis: chemical conversions sensitized by irradiated semiconductors. Acc. Chem. Res. 16, 314–321 (1983).
Zou, Z., Ye, J., Sayama, K. & Arakawa, H. Direct splitting of water under visible light irradiation with an oxide semiconductor photocatalyst. Nature 414, 625–627 (2001).
Moulder, J. F., Stickle, W. F., Sobol, P. E. & Bomben, K. D. Handbook of X-ray Photoelectron spectroscopy: A Reference Book of Standard Spectra for Identification and Interpretation of XPS Data 116–117Perkin-Elmer Corporation Physical Electronics Division (1992).
Raskó, J. & Solymosi, F. Infrared spectroscopic study of the photoinduced activation of CO2 on TiO2 and Rh/TiO2 catalysts. J. Phys. Chem. 98, 7147–7152 (1994).
Renzas, J. R. & Somorjai, G. A. Rh Thin-film nanocatalysts as chemical sensors — the hot electron effect. J. Phys. Chem. C 114, 17660–17664 (2010).
Meng, Q.-Y. A Cascade cross-coupling hydrogen evolution reaction by visible light catalysis. J. Am. Chem. Soc. 135, 19052–19055 (2013).
Acknowledgements
Financial supports from the NSFC (21431008, 21332001, 21403240, 21301173, 21401197 and u1505242), the CAS/SAFEA International Partnership Program for Creative Research Teams and the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB20000000) are greatly appreciated. We thank Prof Xinchen Wang at Fuzhou University for H2 determination assisstance, and Prof Lei Liu and Prof Wei He at Tsinghua University for participating in discussion of this project.
Author information
Authors and Affiliations
Contributions
J.L., Z.L., J.K. and S.H. performed and analysed experiments. W.S. and Y.L. designed the experiments and also prepared this manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures, Supplementary Table, Supplementary Methods and Supplementary References (PDF 2750 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Lin, J., Li, Z., Kan, J. et al. Photo-driven redox-neutral decarboxylative carbon-hydrogen trifluoromethylation of (hetero)arenes with trifluoroacetic acid. Nat Commun 8, 14353 (2017). https://doi.org/10.1038/ncomms14353
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms14353
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.