Abstract
Carbon dioxide capture from point sources like coal-fired power plants is considered to be a solution for stabilizing the CO2 level in the atmosphere to avoid global warming. Methane is an important energy source that is often highly diluted by nitrogen in natural gas. For the separation of CO2 and CH4 from N2 in flue gas and natural gas, respectively, sorbents with high and reversible gas uptake, high gas selectivity, good chemical and thermal stability, and low cost are desired. Here we report the synthesis and CO2, CH4, and N2 adsorption properties of hierarchically porous electron-rich covalent organonitridic frameworks (PECONFs). These were prepared by simple condensation reactions between inexpensive, commercially available nitridic and electron-rich aromatic building units. The PECONF materials exhibit high and reversible CO2 and CH4 uptake and exceptional selectivities of these gases over N2. The materials do not oxidize in air up to temperature of 400 °C.
Similar content being viewed by others
Introduction
Flue gas emitted from coal-fired power plants constitutes 15–16% CO2, 6–7% H2O, 3–4% O2, and about 70% N2 (ref. 1). Carbon dioxide capture from coal-fired power plant emitters is currently considered as a possible technology to stabilize the CO2 level in the atmosphere to avoid climate changes2. A variety of sorbent materials are currently under investigation for carbon dioxide capture. The key features of suitable sorbents for carbon dioxide capture are high selective uptake of CO2, facile regeneration of the sorbent at low-energy penalty, low cost of the sorbent material, chemical stability towards H2O, O2, SOx, and NOx, as well as good thermal stability. Porous materials have been discussed as promising sorbents for carbon dioxide capture. Several classes of porous materials are currently under investigation, predominantly metal–organic frameworks (MOFs)3,4,5,6,7,8,9,10,11, activated carbons12,13,14, molecular organic solids15, and amine-functionalized silicas16,17,18,19,20. More recently, also reports on CO2 sorption in porous covalent organic frameworks and polymers have been published21,22,23,24. Generally, sorbents which physisorb CO2 (for example, MOFs and carbons) tend toward lower selectivity and gas uptake at low CO2 pressure (≤1 atm) but exhibit facile reversibility, while strongly chemisorbing materials such as amine-functionalized silicas tend towards higher selectivities and capacities but typically show less facile reversibility. Recently, amine-functionalized MOFs have been reported that exhibit increased CO2 sorption at low pressure as well as significantly improved selectivity4,5. Still, MOFs tend toward chemical instability (oxidation, hydrolysis) due to the dative nature of the metal–ligand bonds. It is apparent that sorbents with enhanced chemical stabilities and heats of adsorption at the borderline between strong physisorption and weak chemisorption (ca. 25–50 kJ mol−1) are an attractive target to reach a good trade-off between selectivity and reversibility.
Here we produce novel porous electron-rich covalent organonitridic frameworks (PECONFs) as potential sorbents for carbon capture. In PECONFs, inorganic nitridic building units are interconnected via electron-rich aromatic units to form porous covalent frameworks. Carbon dioxide sorption is expected to occur because of the Lewis-acid Lewis-base interactions between the electron-rich aromatic constituents and the electron-poorer carbon dioxide molecules. In addition, weak Lewis acid-base interactions between the lone pairs at the nitridic building units and CO2 could contribute. Both types of interactions are likely stronger than simple dispersion forces (5–20 kJ mol−1) but weaker than strong chemisorptive interactions (>50 kJ mol−1), because no covalent bonds are formed between the sorbent and CO2. The covalent and inert nature of the bonds in PECONFs suggests high chemical stability.
Results
Synthesis
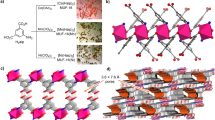
We have produced archetypes of porous electron-rich covalent organonitridic frameworks (PECONFs) from inexpensive 3,3′-diaminobenzidine (DAB) and hexachlorocyclotriphosphazene (Fig. 1) in simple polycondensation reactions using dimethylsulphoxide (DMSO) as the solvent. DAB acts as the electron-rich aromatic building block25,26,27 due to its four amino groups while hexachlorocyclotriphosphazene has the role of the nitridic building unit. These building blocks are significantly less expensive than those used for covalent organic frameworks and polymers typically used so far21,22,23,24. The materials were obtained as monoliths, which could be solvent-exchanged and dried without cracking or disintegration (Supplementary Fig. S1). The microstructures of the monoliths could be modulated by systematically decreasing the amount of the solvent (DMSO) in the reaction mixture (see Supporting Information).
Electron microscopy and X-ray diffraction
The scanning electron microscopy (SEM) (Fig. 2) and transmission electron microscopy (TEM) (Supplementary Fig. S2) images of the PECONFs show that the materials are composed of intergrown, nearly spherical particles. The diameter of the spheres is a function of the reactant concentration. PECONF-1, which was synthesized at the highest concentrations, showed the largest spheres with diameters around 600 nm. PECONF-2 and PECONF-3, which were synthesized at decreased reactant concentrations, exhibited decreased sphere diameters of around 400 and 200 nm, respectively. The moderate intergrowth of the spheres produced macropores between the spheres. PECONF-4, which was prepared at the lowest reactant concentrations, has a polygranular structure with grain sizes in the order of a few tens of nanometers. As a consequence, PECONF-4 does not exhibit macroporosity. All materials were non-crystalline, according to powder X-ray diffraction (Supplementary Fig. S3) and selective area electron diffraction (Supplementary Fig. S4).
MAS NMR spectroscopy
To investigate the structure of the materials in more detail, we have studied the 31P MAS NMR and 13C CP MAS NMR spectra of the PECONFs. Figure 3a shows the 13C spectra of PECONFs. Strong signals around 141 and 131 p.p.m. with a shoulder at 121 p.p.m. and a small weak signal at 106 p.p.m. were observed in the 13C CP MAS spectra. The chemical shifts are consistent with the presence of the DAB building block. This confirms that the electron-rich aromatic DAB unit was incorporated into the PECONF materials. An additional signal was observed around 39 p.p.m., which can be assigned to residual DMSO that was used as the solvent for the synthesis.
The 31P MAS NMR spectra (Fig. 3b) of the PECONFs showed signals in the range from 0 to −25 p.p.m. which are typical for P(V)N4 tetrahedra25,26,28. In PECONF-1, two strong, and one weak signal were observed at 0.4, −10.4, and −22.5 p.p.m., respectively. In PECONF-2, 3, and 4 three signals at almost identical chemical shifts were seen. All the signals were astonishingly sharp given that all the materials were non-crystalline indicating a comparatively high degree of order. The observation of three signals suggests that three different phosphorus environments are present in the frameworks. This may be explained by the fact the P atoms can be surrounded by bridging as well as terminal DAB units in the framework.
Fourier transform infrared spectroscopy
The PECONF materials were further studied by Fourier transform infrared spectroscopy (Supplementary Fig. S5). All the spectra show a broad band around 3,420 cm−1 and a sharp band around 1,617 cm−1. These bands can be attributed to the N–H stretching and bending vibrations, respectively. Furthermore, several broad bands are observed in the region from 1,500 to 500 cm−1. The bands at ~1,090 and ~950 cm−1 can be attributed to the νas(P–NH–P) vibrations. The bands at 1,218 and 1,420 cm−1 are due to the νas(P=N–P) vibrations. The band around 520 cm−1 can be assigned to the δ(P=N–P) vibrations. Similar bands are also observed in phosphorus nitride, nitride imides, and oxynitrides29,30,31,32,33. The sharpness of the bands in the fingerprint region suggests a relatively high degree of order in the materials which corroborates the results from 31P MAS NMR spectroscopy.
Thermal and chemical properties
We further studied the thermal behaviour and the oxidative stability of the PECONFs. Supplementary Figure S6 shows the thermogravimetric analysis (TGA) and differential thermogravimetry (DTG) thermograms of PECONFs. The materials are thermally stable up to a temperature of 400 °C in air. The thermograms demonstrate that mass loss occurs in four steps. The first two steps (mass loss ~20%) below 300 °C are attributed to the desorption of small amounts of water and occluded DMSO (the presence of DMSO was confirmed from the 13C MAS NMR). Only above 400 °C, continuous mass loss was observed that can be attributed to oxidation and framework decomposition (Supplementary Fig. S6). The mass loss at ~500 °C can be assigned to the oxidation of the organic units. Furthermore, a shoulder was observed at ~700 °C which is due to the decomposition of the P–N units. The TGA thermograms of the samples heated in N2 environment are shown in Supplementary Figure S7. All the thermograms show a three-step mass loss with a total mass loss of about 60%. The first two steps were observed below 300 °C and are due to desorption of small amount of water and occluded DMSO. Unlike the sample heated in air, no significant mass loss was observed between 400 to 600 °C. This confirmed that the mass loss at ~500 °C in air is due to the oxidation of the organic units28. The materials are thermally stable up to a temperature of 600 °C in N2 atmosphere. The decomposition of the P-N units did not occur below 800 °C in N2.
Gas adsorption
To probe for porosity in the PECONFs, we recorded N2 isotherms of little monolithic pieces (~2–3 mm diameter) at 77 K. Figure 4a shows the N2 sorption isotherms of PECONF-1, PECONF-2, and PECONF-3. All of these materials show a type-I isotherm, which is typical for microporous materials. The surface area of these samples increases systematically from PECONF-1 to PECONF-3. The Langmuir surface areas were estimated to be 583, 742 and 969 m2 g−1 for PECONF-1, PECONF-2 and PECONF-3, respectively (Table 1). These values are further comparable with the cumulative surface areas (559, 717, and 939 m2 g−1 for PECONF-1, PECONF-2 and PECONF-3, respectively) calculated by density functional theory (DFT) & Monte-Carlo analysis (Table 1). The calculated Brunauer–Emmett–Teller surface areas (499, 637, and 851 m2 g−1 for PECONF-1, PECONF-2 and PECONF-3, respectively) have somewhat lower values compared with the corresponding Langmuir surface areas. The pore volumes estimated at p/p0=0.95 are 0.29, 0.35 and 0.43 cm3 g−1 for PECONF-1, PECONF-2 and PECONF-3, respectively. Details about the physicochemical properties are given in Table 1. No hysteresis was observed between p/p0 of 0.40 and 0.85 in these materials confirming the absence of mesopores. The sharp N2 uptake above p/p0=0.95 confirms the macroporosity, which was already seen by TEM and SEM. The isotherms are not completely closed yet at p/p0=0.20. A similar non-closed isotherm for a non-crystalline covalent organic framework was recently observed by Hupp et al.21
(a) N2 and (b) CO2 sorption of PECONFs (monolithic form) measured at 77 and 273 K, respectively. N2 and CO2 uptake increases from PECONF-1 to PECONF-3. (c) N2 and CO2 sorption isotherms of PECONF-4 monoliths. Due to the absence of macropores, the gas diffusion into the micropores is associated with slow kinetics which leads to a very low N2 uptake. (d) Methane sorption isotherms of PECONF-1, PECONF-2, PECONF-3, and PECONF-4 (powder form) measured at 273 K. Isotherms of PECONF-2 and PECONF-4 are very similar and not distinguishable.
The microporous nature and the high surface area of PENCONF-1, 2, and 3 prompted us to study their CO2 sorption properties. Figure 4b shows the CO2 sorption isotherms of PECONF-1, PECONF-2, and PECONF-3 measured at 273 K. For all materials, the CO2 sorption was completely reversible and no significant hysteresis was observed. The CO2 uptake of PECONF-1, PECONF-2, and PECONF-3 reaches values of 1.86, 2.85, and 3.49 mmol g−1, respectively, at 1 atm. The cumulative surface areas calculated from the CO2 isotherm at 273 K using DFT & Monte-Carlo analysis show values of 438, 670, and 827 m2 g−1 for PECONF-1, PECONF-2, and PECONF-3, respectively (Supplementary Table S1). These values are somewhat lower compared with the surface areas calculated from the N2 sorption data of these materials using the Langmuir equation (Table 1). The results indicate that the CO2 gas uptake scales with the surface area of the material and is inversely proportional to the reactant concentration in the synthesis.
PECONF-4 adsorbed only very small amounts of N2 at 77 K (Fig. 4c) and appeared practically non-porous for N2. Surprisingly, however, it had a relatively large CO2 uptake of 1.81 mmol g−1 at 273 K (Fig. 4c). The surface area and the pore volume calculated from the CO2 sorption isotherm of this material using DFT showed values of 433 m2 g−1 and 0.14 cm3 g−1, respectively (Table 1). The CO2 uptake is remarkable given the small pore volume of only 0.14 mmol g−1. In fact, PECONF-4 has the highest gas uptake to pore volume ratio of all PECONF materials. The pore size distributions of PECONF-1 to PECONF-4 estimated from the CO2 sorption using DFT theory are shown in Supplementary Figure S8. PECONF-1 and PECONF-2 have a narrow monomodal pore size distribution centred on 0.55 nm. PECONF-3 begins to exhibit a bimodal pore-size distribution. PECONF-4 is trimodal with a small additional small maximum is observed at 0.62 nm. The small N2 uptake in PECONF-4 can be explained by the slow mass transport of N2 into the strictly microporous PECONF-4 monoliths at 77 K. In contrast, at 273 and 298 K, mass transport is large enough to allow for a significant CO2 gas uptake in PECONF-4. To confirm that mass transport has an important role in PECONF-4 monoliths, we pulverized PECONF-4 monoliths and remeasured the CO2 isotherms at 273 and 298 K and the N2 isotherms at 77 K. As expected, the PECONF-4 material showed at type I N2 isotherm (Supplementary Fig. S9). The CO2 capacity increased to a value of 2.95 mmol g−1 (Supplementary Fig. S10). This effect was absent for pulverized forms of PECONF-1, 2, and 3 demonstrating that their hierarchical microporous–macroporous structures provide effective mass transport into the monoliths (Supplementary Fig. S11). We have further studied the methane sorption behaviour of the PECONF materials in powder form. Figure 4d shows the CH4 sorption isotherms of the PECONF-1 to PECONF-4. All the isotherms are reversible with a high maximum uptake of 1.07 mmol g−1 observed for PECONF-2 and PECONF-4 at 1 bar and 273 K. The PECONF-3 adsorbs a somewhat smaller amount (1.00 mmol g−1) of methane. PECONF-1 has the lowest CH4 gas uptake of 0.83 mmol g−1. The methane uptake capacities of these samples are still high at 298 K with the maximum uptake of 0.67 mmol g−1 at a pressure of 1 bar observed for PECONF-4. This methane uptake (15 cc g−1) is higher compared with the methane sorption (0.8 wt % or 11.1 cc g−1) of microporous tris-o-phenylenedioxycyclotriphosphazene van der Waals crystals34 reported by Sozzani et al.35 but somewhat smaller than those of covalent organic cages reported by Tozawa et al.36 However, the latter can only be made by an expensive synthesis procedure. The methane sorption capacities of PECONF-1, PECONF-2, and PECONF-3 are 0.53, 0.63, and 0.58 mmol g−1, respectively at 298 K (Supplementary Fig. S12).
Heats of adsorption
To further understand the pore surface characteristics of the materials and the adsorption process, we have calculated the isosteric heats of adsorption (ΔH) for CO2 and CH4. ΔH was calculated using the Clausius–Clapeyron equation from the sorption data collected at 273, 298, and 323 K (refs 4, 37). The ΔH value can be obtained from the plot of ln p versus the reciprocal of the temperature. Figure 5a shows ΔH of CO2 for PECONF-1 to PECONF-4. At the adsorption onset, the ΔH values for PECONF-1, PECONF-2, PECONF-3, and PECONF-4 are 29, 31, 26, and 34 kJ mol−1, respectively. Remarkably, the isosteric heat of adsorption is nearly independent from the amount of CO2 adsorbed. The heat of adsorption drops only by 30% (PECONF-2) and 25% (PECONF 3 and 4), respectively. This suggests that if the pore volumes of the materials can be further increased a significantly higher CO2 gas uptake can probably be achieved even at low CO2 pressure.
We further calculated the isosteric heat of adsorption of methane for all the samples. Figure 5b shows ΔH of PECONF-1 to PECONF-4. At the adsorption onset, PECONF-1, PECONF-2, PECONF-3, and PECONF-4 show high values of 22, 27, 25, and 22 kJ mol−1, respectively. The heat of adsorption only weakly decreases with increased methane loading. It is noteworthy, that with the exception of PECONF-4 the methane gas uptake is increasing with increasing heat of adsorption, while the CO2 gas uptake increases with the surface area. Apparently, in the case of methane adsorption the heat of adsorption is more important than the surface area. The generally high methane uptake for all PECONFs may be explained by H-π interactions. Such interactions have been observed in Van der Waals crystals of tris-o-phenylenedioxycyclotriphosphazene molecules, that are structurally related to the building units of the PECONF materials35.
Gas selectivity
In addition to the high CO2 uptake and the reversibility, a high selectivity for CO2 over N2 is one of the necessary properties for a material to be used as a CO2 adsorbent. The selectivity for the PECONF monoliths was estimated using the ratios of the Henry law constants4. These constants can be calculated from the initial slopes of the isotherms. This method is one of the most common methods to calculate gas selectivities and has been applied extensively to determine gas selectivities of metal–organic frameworks, carbons, and covalent organic frameworks. The calculated CO2:N2 selectivity of PECONF-1 is the highest among the PECONF materials reaching values of 109:1 at 273 K and 51:1 at 298 K (Supplementary Table S1; Supplementary Figs. S13 and S14). The value is much higher compared with the next-higher value reported so far (81:1 at 273 K)4. Bae et al.38 reported a CO2/N2 selectivity of 95 at 298 K for a carborane-based metal–organic framework. However, this material appears to be more expensive than our materials. The CO2:N2 selectivities for PECONF-2 (Supplementary Figs S13 and S15) and PECONF-3 (Supplementary Figs S13 and S16) are still high (PECONF-2: 74:1 and 44:1 at 273 and 298 K, PECONF-3: 77:1 and 41:1 at 273 and 298 K). The higher selectivity of PECONF-1 compared with PECONF-2, and 3 can be attributed to its higher heat of adsorption. PECONF-4 has a selectivity of 83:1 and 51:1 at 273 and 298 K, respectively (Supplementary Figs S13 and S17).
We further calculated the selectivity of CO2:CH4 and CH4:N2 of these samples at 273 and 298 K (Supplementary Table S1; Supplementary Figs S13-S17). PECONF-4 has the highest CO2:CH4 selectivity of 12 at 273 K among all the materials. However, the CH4:N2 selectivity of 16 is maximum for PECONF-1 at 273 K. Sorbents with high selectivity for CH4 over N2 are of high practical interest, because methane is often highly diluted by N2 in natural gas wells. Currently, N2/CH4 separations are still being done cryogenically because of the absence of solid sorbents with high methane sorption capacities and suitable selectivities.
To verify the validity of the selectivities estimated from Henry's constant ratios, we have additionally calculated the selectivities by an independent method. We have used ideal adsorbed solution theory (IAST) which allows for the determination of the selectivities as a function of pressure. Because values calculated from Henry constant ratios represent selectivities close to zero pressure, IAST selectivities at zero pressure should match with selectivities calculated from Henry's law constants. Our calculations show that the IAST selectivities are in reasonable agreement with the values obtained from Henry's constant ratios (Supplementary Figs S18-S23). The CO2/N2 selectivity of PECONF-1 at 273 is even somewhat higher (135) compared with the value obtained from Henry's constants (109). It should be noted though that real selectivites can only be determined by measurements that mimic the proposed sorption conditions. For example, in post-combustion CO2 capture, the contact time with the sorbent would likely to be of the order of seconds; as such, any equilibrium sorption measurements have the potential to be misleading, and in this regard pore size and kinetic mass transport effects are also important.
Discussion
In conclusion, we have synthesized microporous electron-rich covalent organonitridic frameworks in facile condensation reactions from commercially available, relatively inexpensive building blocks. The structures are tunable despite their non-crystallinity and exhibit a remarkable degree of mid-range order. The hierarchical micro-macroporosity is a further advantageous feature of the materials due to the benefits of hierarchical porosity for the mass transport in porous materials. The tunability of the structures translates to their gas sorption properties. The materials adsorb high amounts of CO2 (up to 3.5 mmol g−1 at 1 atm and 273 K) which is exceeded by only a few other sorbents, namely the amine-functionalized silica MBA-2 (3.8 mmol)33, and the amine-functionalized MOFs bio-MOF-11 (6.0 mmol g−1), Zn2(C2O4)(C2N4H3)2·(H2O)0.5 (4.35 mmol g−1)4,5, and Mg/DOBDC (7.2 mmol g−1)39,40. PECONF-1 exhibits one of the highest selectivities (CO2 over N2) for a sorbent with a reversible CO2 adsorption–desorption isotherm. The high isosteric heat of adsorption and the high selectivity for CO2 over N2 can be explained by Lewis acid–base interactions between the electron-poor CO2 and the electron-rich sorbent. The heats of adsorption do not decline significantly even at high CO2 loadings. Similarly, high isosteric heats of adsorption have been observed only for metal-organic frameworks but not for covalent framework materials composed exclusively from non-metals41. The strong covalent P–N, N–C, and C–C bonds in the framework provide the materials with high chemical robustness compared with MOFs as seen by the high oxidative stability at temperatures as high as 400 °C. The combination of high thermal and oxidative stability, high gas uptake, low cost, and exceptional selectivity for CO2 over N2 make these materials interesting candidates for CO2 capture applications.
Furthermore, the PECONF materials show high sorption capacities and heats of adsorption for methane as well as high selectivities of CH4 over N2 which is of high interest for the purification of natural gas that is frequently diluted with N2.
The mechanically very stable monolithic structure as well as the hierarchical micro-macroporosity suggests further applications, for example in monolithic chromatography. The successful synthesis of the PECONF materials in monolithic shape and the strictly microporous nature of PECONF-4 suggests that this material can also be made as gas-selective membranes.
Methods
Materials
Hexachlorocyclotriphosphazene, Alfa Aesar, 98%. DAB, AK Scientific, 99%. Anhydrous dimethyl sulfoxide (DMSO), Alfa Aesar, 99.8%.
Synthesis of PECONFs
In a typical synthesis, about 1.5 mmol of DAB and 0.5 mmol of PNC were dissolved in 3, 7.5, 15 and 20 ml of DMSO for the synthesis of PECONF-1, PECONF-2, PECONF-3, and PECONF-4, respectively. The resulting solution was stirred for 30 min at RT. It was then heated at a temperature of 125 °C. Gelation starts as early as 5 min. After 30 min the sample cooled down to room temperature and solid monoliths were collected. All the experiments were carried out in N2 atmosphere using Schlenk's apparatus. The DMSO from the as-synthesized monoliths was removed by soxhlet extraction using diethyl ether. The obtained solid monoliths were sonicated in 30 ml of water, washed several times with water, and dried in vacuum at 100 °C. The sonicator used in the present research was Cavitator Ultrasonic ME 11 (Mettler Electronics) with a maximum power output of 200 W at 67 kHz.
Measurements
The microstructures of the specimens were studied by SEM and transmission electron microscopy (TEM). The SEM images of the specimens were taken on a Hitachi S-4300 SEM. The TEM images were taken on a JEOL JEM-2000 electron microscope operated at 200 kV. X-ray diffraction was carried out using a Rigaku Rotaflex diffractometer with a Cu Kα radiation source (λ=0.15405 nm). The thermogravimetric analysis experiments were carried out both in air and nitrogen using a TA TGA 2950 instrument with a heating rate of 10 °C per minute. 31P and 13C CP MAS NMR spectra were obtained at 75.468 MHz on a General Electric NMR Instrument model GN-300 equipped with a Doty Scientific 7 mm MAS probe. 13C NMR chemical shifts were referenced to the downfield line of adamantine at 38.55 p.p.m., and for 31P NMR, the chemical shift of 85% H3PO4 was set to zero using external reference standards. The formation of the microporous framework structures were studied by nitrogen and carbon dioxide sorption using an Autosorb-1 instrument (Quantachrome). Before analysis, all the samples were out-gassed overnight at 150 °C in vacuum. The Brunauer–Emmett–Teller surface area analysis was carried out using 5 data points in the pressure range between 0.05 and 0.35 atm. Before IAST calculations, all isotherms were fitted into a Langmuir–Freundlich isotherm. In all cases, a fit >99% was reached. IAST calculations were performed using the software package ChromWorks.
Additional information
How to cite this article: Mohanty, P. et al. Porous covalent electron-rich organonitridic frameworks as highly selective sorbents for methane and carbon dioxide. Nat. Commun. 2:401 doi: 10.1038/ncomms1405 (2011).
References
White, C. M., Strazisar, B. R., Granite, E. J., Hoffman, J. S. & Pennline, H. W. Separation and capture of CO2 from large stationary sources and sequestration in geological formations-coalbeds and deep saline aquifers. J. Air Waste Manage. Assoc. 53, 645–715 (2003).
Haszeldine, R. S. Carbon capture and storage: how green can black be? Science 352, 1647–1652 (2009).
Britt, D., Furukawa, H., Wang, B., Glover, T. G. & Yaghi, O. M. Highly efficient separation of carbon dioxide by a metal-organic framework replete with open metal sites. Proc. Natl Acad. Sci. USA 106, 20637–20640 (2009).
An, J., Geib, S. J. & Rosi, N. L. High and selective CO2 uptake in a cobalt adeninate metal-organic framework exhibiting pyrimidine- and amino-decorated pores. J. Am. Chem. Soc. 132, 38–39 (2010).
Vaidhyanathan, R., Iremonger, S. S., Dawson, K. W. & Shimizu, G. K. H. An-amine functionalized metal organic framework for preferantial CO2 adsorption at low pressures. Chem. Commun. 5230–5232 (2009).
Couck, S., Denayer, J. F. M., Baron, G. V., Remy, T., Gascon, J. & Kapteijn, F. An amine-functionalized MIL-53 metal-organic framework with large separation power for CO2 and CH4 . J. Am. Chem. Soc. 131, 6326–6327 (2009).
Wang, B., Cote, A. P., Furukawa, H., O'Keeffe, M. & Yaghi, O. M. Colossal cages in zeolitic imidazolate frameworks as selective carbon dioxide reservoirs. Nature 453, 207–212 (2008).
Banerjee, R. et al. High-throughput synthesis of zeolitic imidazolate frameworks and application to CO2 capture. Science 319, 939–943 (2008).
Banerjee, R., Furukawa, H., Britt, D., Knobler, C., O'Keeffe, M. & Yaghi, O. M. Control of pore size and functionality in isoreticular zeolitic imidazolate frameworks and their carbon dioxide selective capture properties. J. Am. Chem. Soc. 131, 3875–3877 (2009).
Hamon, L. et al. Co-adsorption and separation of CO2-CH4 mixtures in the highly flexible MIL-53(Cr) MOF. J. Am. Chem. Soc. 131, 17490–17499 (2009).
Cheon, Y. E., Park, J. & Suh, M. P. Selective gas adsorption in a magnesium-based metal-organic framework. Chem. Commun. 5436–5438 (2009).
Drage, T. C., Blackman, J. M., Pevida, C. & Snape, C. E. Evaluation of activated carbon adsorbents for CO2 capture in gassification. Energy Fuels 23, 2790–2796 (2009).
Ribeiro, R. P., Sauer, T. P., Lopes, F. V., Moreira, R. F., Grande, C. A. & Rodrigues, A. E. Adsorption of CO2, CH4, and N2 in activated carbon honeycomb monolith. J. Chem. Eng. Data 53, 2311–2317 (2008).
Przepiorski, J., Tryba, B. & Morawski, A. W. Adsorption of carbon dioxide in phenolic-resin based carbon spheres. Appl. Surf. Sci. 196, 296–300 (2002).
Tian, J., Thallapally, P. K., Dalgarno, S. J., McGrail, P. B. & Atwood, J. L. Amorphous molecular organic solids for gas adsorption. Angew. Chem. Int. Ed. 48, 5492–5495 (2009).
Guerrero, R. S., Da'na, E. & Sayari, A. New insights into the interactions of CO2 with amine functionalized silica. Ind. Eng. Chem. Res. 47, 9406–9412 (2008).
Kim, S. N., Son, W. J., Choi, J. S. & Ahn, W. S. CO2 adsorption using amine-functionalized mesoporous silica prepared via anionic surfactant-mediated synthesis. Micropor. Mesopor. Mater. 115, 497–503 (2008).
Zukal, A., Dominguez, I., Mayerova, J. & Cejka, J. Functionalization of delaminated zeolite ITQ-6 for the adsorption of carbon dioxide. Langmuir 25, 10314–10321 (2009).
Harlick, P. J. E. & Sayari, A. Applications of pore-expanded mesoporous silica. 5. Triamine grafted material with exceptional CO2 dynamic and equilibrium adsorption performance. Ind. Eng. Chem. Res. 46, 446–458 (2007).
Tang, Y. & Landskron, K. CO2-sorption properties of organosilicas with bridging amine functionalities inside the framework. J. Phys. Chem. C 114, 2494–2498 (2010).
Farha, O. K. et al. Synthesis, properties, and gas separation studies of a robust diimide-based microporous organic polymer. Chem. Mater. 21, 3033–3035 (2009).
Ben, T. et al. Targeted synthesis of a porous aromatic framework with high stability and exceptionally high surface area. Angew. Chem. Int. Ed. 48, 9457–9460 (2009).
McKeown, N. & Budd, P. Exploitation of intrinsic microporosity in polymer-based materials. Macromolecules 43, 5163–5176 (2010).
Jiang, J.-X. & Cooper, A. I. Microporous organic polymers: design, synthesis, and function. Top. Curr. Chem. 293, 1–33 (2010).
Allcock, H. R., Allen, R. W., Bissell, E. C., Smeltz, L. A. & Teeter, M. Molecular motion and molecular separations in cyclophophazene clathrates. J. Am. Chem. Soc. 98, 5120–5125 (1976).
Allcock, H. R. Cyclophophazene clathrates-exploring the adjustable tunnel. Acc. Chem. Res. 11, 81–87 (1978).
Allcock, H. R., Diefenbach, U. & Pucher, S. R. New mono- and trispirocyclotriphosphazenes from the reaction of (NPCl2)3 with aromatic ortho dinucleophiles. Inorg. Chem. 33, 3091–3095 (1994).
Mohanty, P. & Landskron, K. Simple systematic synthesis of size-tunable covalent organophosphonitridic framework nano- and microspheres. New J. Chem. 34, 215–220 (2010).
Mohanty, P. & Landskron, K. Synthesis of periodic mesoporous phosphorus-nitrogen frameworks by nanocasting from mesoporous silica using melt-infiltration. J. Mater. Chem. 19, 2400–2406 (2009).
Guo, Q., Yang, Q., Zhu, L., Yi, C. & Xie, Y. Large-scale synthesis of amorphous phosphorus nitride imide nanotubes with high luminescent properties. J. Mater. Res. 20, 325–330 (2005).
Gu, H., Gu, Y., Li, Z., Ying, Y. & Qian, Y. Low-temperature route to nanoscale P3N5 hollow spheres. J. Mater. Res. 18, 2359–2363 (2003).
Schnick, W., Lucke, J. & Krumeich, F. Phosphorus nitride P3N5: synthesis, spectroscopic, and electron microscopic investigations. Chem. Mater. 8, 281–286 (1996).
Meng, Z., Peng, Y. & Qian, Y. Microtubes and balls of amorphous phosphorus nitride imide (HPN2) prepared by a benzene-thermal method. Chem. Commun. 469–470 (2001).
Atwood, J. L., Barbour, L. J., Jerga, A. & Schottel, B. L. Guest transport in a nonporous organic solid via dynamic van der Waals cooperativity. Science 298, 1000–1002 (2002).
Sozzani, P., Bracco, S., Comotti, A., Ferretti, L. & Simoutti, R. Methane and carbon dioxide storage in a porous van der Waals crystal. Angew. Chem. Int. Ed. 44, 1816–1820 (2005).
Tozawa, T. et al. Porous organic cages. Nat. Mater. 8, 973–978 (2009).
Dinca, M. & Long, J. R. Strong H2 binding and selective gas adsorption within the microporous coordination solid Mg3(O2C-C10H6-CO2)3 . J. Am. Chem. Soc. 127, 9376–9377 (2005).
Bae, Y.-S., Spokoyny, A. M., Farha, O. K., Snurr, R. Q., Hupp, J. T. & Mirkin, C. A. Separation of gas mixtures using Co(II) carborane-based porous coordination polymers. Chem. Commun. 46, 3478–3480 (2010).
Ma, X., Wang, X. & Song, C. 'Molecular basket' sorbents for separation of CO2 and H2S from various gas streams. J. Am. Chem. Soc. 131, 5777–5783 (2009).
Caskey, S., Wong-Foy, A. & Matzger, A. Dramatic tuning of carbon dioxide uptake via metal substitution in a coordination polymer with cylindrical pores. J. Am. Chem. Soc. 131, 10870–10871 (2008).
Dawson, R., Adams, D. & Cooper, A. I. Chemical Tuning in CO2 sorption in robust nanoporous organic polymers. Chem. Sci. 2, 1173–1177 (2011).
Acknowledgements
The present work was supported by Lehigh University start-up funds and faculty grants. Dr Norm Zheng is gratefully acknowledged for MAS NMR measurements. We thank Mr Wade H. Bailey who supported our thermal analysis experiments.
Author information
Authors and Affiliations
Contributions
K.L. and P.M. designed experiments. P.M. and L.K. performed the experiments. K.L. and P.M. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1-S23 and Supplementary Tables S1-S5. (PDF 888 kb)
Rights and permissions
About this article
Cite this article
Mohanty, P., Kull, L. & Landskron, K. Porous covalent electron-rich organonitridic frameworks as highly selective sorbents for methane and carbon dioxide. Nat Commun 2, 401 (2011). https://doi.org/10.1038/ncomms1405
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms1405
This article is cited by
-
Molecular engineering toward sustainable development of multiple-doped hierarchical porous carbons for superior zinc ion storage
Science China Materials (2023)
-
Ferrocenyl building block constructing porous organic polymer for gas capture and methyl violet adsorption
Journal of Central South University (2020)
-
Porous covalent organonitridic frameworks for solid-phase extraction of sulfonamide antibiotics
Microchimica Acta (2019)
-
Template-free synthesis of porous carbon from triazine based polymers and their use in iodine adsorption and CO2 capture
Scientific Reports (2018)
-
A Facile Approach to Generate Cross-Linked Poly(cyclotriphosphazene-co-oxyresveratrol) Nanoparticle with Intrinsically Fluorescence
Journal of Inorganic and Organometallic Polymers and Materials (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.