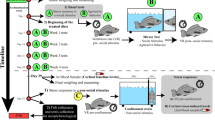
Abstract
Migration is an important life-history event in a wide range of taxa, yet many migrations are influenced by anthropogenic change. Although migration dynamics are extensively studied, the potential effects of environmental contaminants on migratory physiology are poorly understood. In this study we show that an anxiolytic drug in water can promote downward migratory behaviour of Atlantic salmon (Salmo salar) in both laboratory setting and in a natural river tributary. Exposing salmon smolt to a dilute concentration of a GABAA receptor agonist (oxazepam) increased migration intensity compared with untreated smolt. These results implicate that salmon migration may be affected by human-induced changes in water chemical properties, such as acidification and pharmaceutical residues in wastewater effluent, via alterations in the GABAA receptor function.
Similar content being viewed by others
Introduction
Animal migration, be it over land, in the oceans, or through the air, is an astonishing phenomenon that expose animals to extraordinary challenges as they travel into and across unknown and often risky environments. Although the ecological factors underlying migration have been extensively studied in many animals, the neurobiological mechanisms governing migration are still poorly understood. Atlantic salmon (Salmo salar) is an iconographic species with immense biological, cultural and socio-economic value1,2. Its life cycle includes a vast and critical migration event, starting with the juveniles (smolts) leaving their natal river, heading thousands of kilometres out into the ocean to feed and grow, returning a few years later to the same river to spawn as large adults3. Understanding what regulates salmon migration dynamics is imperative for effective management and conservation of salmon populations, especially in the face of declining populations4,5. One of the most critical phases of the migration cycle is when the smolts leave their nursery habitat in the river and migrate downstream to the ocean6. Although decades of research have identified many of the underlying environmental and ontogenetic factors influencing the onset and intensity of salmon smolt migration4,7,8, a deeper understanding of general mechanisms controlling migration is still lacking4,9. So far, no studies have looked beyond hard-wired environmental cause–effect relationships in their effort to explain migration behaviour in salmon.
Downstream migration is characterized by high risk and uncertainty for the smolt, as they enter an unknown and potentially dangerous environment, often full of predators6,10. In other migrating fish, risk-prone individuals have been found more willing to migrate than risk-averse individuals11, but little is known about the mechanistic process underlying such differentiation. Recently, attention to cognitive and psychological dimensions in how animals respond to risk and danger has provided intriguing new insights in behavioural ecology and management12,13. Sustained stress grounded in anticipation of fear and danger may permeate an individual’s motivation, choices and behaviour, and has been found to influence a wide range of ecological characteristics, including species distribution and population demographics13, complementing more established ecological drivers such as resource availability and environmental conditions. Such anxiety-related emotion-based stress is thought to be adaptive, as it may minimize unnecessary risk taking and maintain alertness even though no immediate stressor, such as a predator, might be present12.
In vertebrates, anxiety can be reduced by activation of the ligand-gated GABAA receptor in the central nervous system, either by the endogenous inhibitory neurotransmitter gamma-aminobutyric acid (GABA) or by other agonistic molecules, reducing overall neurotransmission by decreasing the neurons action potential14. The GABAA receptor system is highly conserved within vertebrates15, and several psychiatric drugs intended to reduce anxiety in humans also have anxiolytic-like effects on other animals, such as rodents and fish16,17. In fish, where the relevance of cognitive and emotional concepts are intensely debated18,19, drugs targeting the GABAA receptor function constitute powerful neurobiological tools to understand and interpret cognitive aspects of behavioural responses to risk and danger. Further, altered behaviour in fish due to modulation of the GABAA receptor function following exposure to anthropogenic water pollution, such as pharmaceutical residues and/or acidification, has been highlighted as a major concern in aquatic ecotoxicology and conservation17,20.
In this study, we investigated whether exposure to environmental relevant concentrations of oxazepam, a positive allosteric modulator of the GABAA receptor and a common agent in anxiolytic pharmaceuticals, affected the migration behaviour of Atlantic salmon smolt. Migration of smolts exposed to oxazepam in water was quantified and compared with non-exposed smolt migration both in the laboratory and in the field, by tracking the downstream movement of each individual smolt using small implanted microchips. In the laboratory, the intensity (that is, speed) of the migration was quantified as the number of downstream laps a smolt migrated in a large migration mesocosm. In the field, the migration speed was measured as the time it took for a smolt to migrate out of a small river tributary.
Results
Downstream migration intensity
In both the laboratory and field study, smolts exposed to oxazepam migrated significantly faster than unexposed control fish (laboratory study: general linear mixed model (LME), F1,170=19.7, P<0.01, Fig. 1; field study: LME, F1,107=10.7, P=0.014, Fig. 2). The effect of treatment on migration intensity in the lab was dependent of time (that is, significant interaction term; LME, F1,2259=39, P<0.01), where migration intensity of exposed fish was significantly higher than of control fish during the first 10 h (post hoc Tukey, P<0.01, Fig. 3), but not during the last 10 h (that is, between 60 and 70 h; post hoc Tukey, P=0.53, Fig. 3) of the migration trials. Analysis of within-treatment changes in migration intensity over time showed no difference in migration intensity of control fish between the first 10 and the last 10 h of the migration trial (post hoc Tukey, P=0.96, Fig. 3). The migration intensity of exposed fish was steadily decreasing over time, which is in line with the elimination rate of oxazepam from fish muscle tissue (Fig. 4). The no-effect concentration occurred after approximately 60 h at when mean fish muscle concentration of oxazepam was about 7.0 μg kg−1 (Fig. 4).
Downstream migration intensity for Atlantic salmon smolt (mean and 95% confidence intervals) in control and oxazepam-exposed Atlantic salmon smolts in a large-scale migration mesocosm. Migration intensity is inferred from the number of laps registered in the migration pool, and values for each treatment (exposed and control) represents the mean number of laps per smolt individual completed in the migration pool during 70 h, and is based on five independent replicated trials, each containing 20 smolts per treatment.
Downstream migration intensity in control (dashed line) and oxazepam-exposed (solid line) Atlantic salmon smolt as a function of time (lower x axis) and concentration of oxazepam in fish muscle tissue (upper x axis). Values are fitted values from a Loess smoother, and dotted lines are 95% confidence interval bands.
Diurnal trends
There was a clear difference between exposed and control smolt in diurnal migration intensity, as revealed by LOESS smoothers fitted to diurnal migration data from the laboratory experiment (Fig. 5). However, based on model selection using Akaike information criterion, a fourth order polynomial regression model was judged the most parsimonious for both exposed and control smolt, indicating that both groups exhibited a bimodal diurnal migration rhythm, where migration occurred more intensively in the morning and the evening (Supplementary Table 1).
Diel migration activity for control (dashed line) and oxazepam-exposed (solid line) Atlantic salmon smolt. Migration intensity was measured as the mean number of downstream detections per hour per fish individual. The lines are embedded with 95% confidence interval bands, and are the fitted values from a Loess smoother.
Fish condition
There were no differences in mass, length, or Fulton condition factor (K) between control and exposed fish for neither 1- nor 2-year old smolt (one-way analysis of variance, all P>0.05).
Discussion
Salmon migration is one of the most intensely researched topics in fisheries science9,21. Yet, despite the great number of studies on the physiological and environmental effects underlying migration, considerable variation in migration behaviour remains unexplained4,6. Our study clearly shows that exposure to the GABAA receptor agonist oxazepam, an anxiolytic pharmaceutical, significantly increased migration intensity in salmon smolt. Although oxazepam also accentuated the diurnal migration intensity, it did not alter the diurnal migration pattern compared with the control fish or to the natural diurnal pattern seen in salmon in the adjacent Ume river system (Supplementary Fig. 1). The fact that the diurnal migration pattern in our study mimics that seen in wild salmon migrating in Ume River corroborates that we truly are measuring migration and not just activity.
Exposure to oxazepam is known to reduce anxiety in fish (reviewed in refs 17, 22), resulting in increased risk taking23,24, and the role of the GABAA receptor in regulating anxiety in salmon has recently been shown using gabazine, an antagonist to the GABAA receptor25. Hence, the intensified migration seen in our study could be explained by a reduction in anxiety that otherwise would constrain the intensity of risky activities such as migration. Downstream migration behaviour is crucial in the life cycle of salmon, and the timing and intensity of the migration is adaptive as it impacts survival and fitness of the fish6,26. An emotional adaptation27, where selection acts on anxiety phenotypes that generates a migration response matching the specific risk conditions prevailing in the river or estuary, is an intriguing and novel thought that complement current research stating migration as strongly governed by the cognitive ability and personality of fish11,28.
Any disruption in salmon migration behaviour can have unforeseen and severe ecological consequences6. Recently, the anxiolytic function of the GABAergic system has gained attention in ecotoxicology and animal conservation, as anthropogenically altered environmental conditions, such as pharmaceutical contamination and acidification, have been found to affect the GABAA receptor in fish with subsequent behavioural and potentially ecological effects17,29. Anxiolytic drugs targeting the GABAergic system enter wastewater effluents at concentrations high enough to affect behaviour in fish24,30. In our study, salmon smolts were exposed to concentrations of oxazepam within the range of concentrations that have been found in effluent water31,32, and we here show effects of dilute concentrations of pharmaceutical on behaviours linked to crucial non-reproductive life cycle events in fish. The use of anxiolytic pharmaceuticals is projected to increase globally33, and, as a consequence, concentrations of oxazepam in aquatic environments close to urbanized areas may more than double in the coming decades34. With such scenarios in mind, significant ecological effects of benzodiazepines in the environment seems plausible, especially as concentration of oxazepam in fish tissue may be magnitudes higher than in the surrounding water due to bioconcentration24,35. As such, this study highlights the importance of understanding the behavioural and ecological implications of such contamination to make proper ecotoxicological risk assessments36.
In addition to anxiolytic pharmaceuticals, recent research has shown water pH to influence the polarizing state of the GABAA receptor in fish, with behavioural alterations as a consequence29. In waters acidified by dissolved CO2, the action potential of the GABAA receptor is reduced, leading to reduced anxiety in fish, often expressed as an increase in risk taking37,38. Acidification of waters is emphasized as a major global threat to salmon, based on the negative impact on physiological development and survival39. Recently, climate-change related acidification, due to increased CO2 levels, has been suggested to be especially detrimental to salmon20, partly because it modifies risk-taking behaviour and anxiety during the freshwater phase25. Our study adds to this concern by suggesting that also migration behaviour could be affected. However, it is important to be aware that although oxazepam and CO2 exposure lead to the same endpoint (that is, modification of GABAA receptor function and altered anxiety levels), we still lack knowledge of comparable levels of effect concentrations for oxazepam and CO2 exposure. Further, CO2 exposure may also have considerably different effect dependent on environment and species. For example, fish species evolved in well-buffered systems such as the ocean may be more sensitive to variation in pH compared with fish in freshwater environments where seasonal fluctuations in pH may be significant. Nevertheless, pH effects on the GABAA receptor has recently been demonstrated for freshwater fish species, indicating that alterations of the carbonate buffer system in aquatic ecosystems might have broad-scale effects on fish behaviour40. However, more knowledge of potential consequences of GABAA-receptor-induced modification in migration intensity due to changes in pH following anthropogenic-induced acidification or liming is needed.
Although the ecotoxicological relevance of our findings is evident, the fact that we found significant increases in migration following exposure to oxazepam suggests direct applications in fisheries management. Enhancing weak salmonid populations by stocking hatchery-reared smolt is a common management practice worldwide41, but the success of such enhancement stocking is considered to be low42,43. Inadequate migration behaviour in hatchery smolt has been identified as an important reason for the low success44,45,46,47, as low migration intensity increases the risk of predation5,10. Consequently, large efforts have been made to improve migration intensity of hatchery fish, via, for example, enriched rearing environments48, intense exercising programs47, and dietary regimes49, but with so far modest success. It is noteworthy that by exposing the hatchery smolt to low concentrations of oxazepam, the smolt almost doubled its migration intensity. Such treatment, although unconventional, would not drug the fish permanently as elimination rate of oxazepam is relatively high as shown in this study. Future studies should investigate how oxazepam exposure affects migration success of hatchery smolt after release into rivers, using suitable telemetry techniques to track their behaviour and survival in large natural systems50.
Our study adds important new mechanistic understanding of migration behaviour in salmon by suggesting the involvement of the GABAA receptor for regulation of downstream migration intensity. The study showcase a novel method of using psychoactive drugs to investigate the role of anxiety for fish behaviour and ecology, and provides a new conceptual framework for understanding salmon migration by suggesting cognitive and emotional dimensions that complement the current view of migration as a purely environmental and physiological response. Hence, our findings have implications for aquatic ecotoxicology and conservation, as well as for applied fisheries management.
Methods
Experiment
All experimental protocols and animal handling in this study were approved by the Swedish Animal Ethics Board (Dnr: A-11-13). The laboratory study was conducted between 6th of May and 20th of June 2013 and the field study between 27th May and 1st of June 2015, at Norrfors compensatory hatchery (Vattenfall AB) (63°52′ N; 20°01′ E), located at the Ume River about 10 km from the city of Umeå, Sweden. The hatchery annually produces 80,000 Atlantic salmon smolts, which are released into the Ume River as compensation for the loss of wild smolt production due to hydroelectric damming of the river. The smolts are reared in large round pools (diameter 8 m, depth 1 m), supplied with continuously running water diverted from the nearby river, and are fed according to standard commercial protocols. The light conditions in the hatchery are set to follow the natural light regime for the region. The majority of the fish smoltify at the age of 1 or 2 years. During late spring, the young salmon (that is, parr) stops holding a position against the current and start to actively swim downstream. This change in behaviour signals the completion of the smolting51 and is used by the hatchery staff as a cue to initiate the release of salmon into the river52.
Laboratory study
In April, before the onset of the downstream migration, 80 2-year old and 120 1-year old salmon were randomly selected from the hatchery stock and transferred to 400 l holding tanks supplied with continuously running water. One tank per age group was used. In conjunction with the transfer, the parr were anaesthetized with MS222 and measured for length (mean±1 s.d.: 1-year old=114±8.5 mm, 2-year old=145±8.3 mm) and weight (mean±1s.d.: 1-year old=14.5±3.6 g, 2-year old=26.9±7.3 g), after which each fish was tagged with a 12-mm passive intergrated transponder (PIT) (Biomark Inc, BIO12.B.03/TX708HQ) into the abdominal cavity using a syringe. PIT tags are routinely used to track and monitor juvenile salmonids, with very little or no impact on the fish53.
Exposure (laboratory)
The experiment was run in a set of six trials, each trial including 96 h of exposure to oxazepam, and 70 h of migration test. Forty fish (20 that had been exposed to oxazepam and 20 unexposed controls) were tested in each of the first four trials, while 20 control fish were tested in the fifth trial, and 20 exposed in the sixth trial. Every third day, starting 1st of June, a new trial was initiated. The first two trials consisted of two-year old fish, and the last four trials of 1-year old fish. Exposure started by transferring fish from the holding tanks to 1 × 1-m cattle tanks filled with 280 l of standing water diverted from the river, keeping 20 fish per tank. Then, oxazepam dissolved in water was poured into one of the cattle tanks resulting in an oxazepam concentration of 1.9 μg l−1. Each cattle tank had an oxygen pump to compensate for the oxygen consumption by the fish. Water temperature was kept constant at 10 °C during the exposure period. Initial studies on the uptake and elimination of oxazepam in salmon smolt showed that salmon reach steady state with the surrounding water concentrations after 4 days (Supplementary Fig. S2), and the fish in this study were hence exposed for 96 h to ensure steady state. The same initial uptake study also concluded that salmon completely eliminated oxazepam from blood and tissue after 150 h if kept in clean water (Supplementary Fig. S2). After 60–70 h in clean water, tissue-concentrations of oxazepam are below 7.5 μg kg−1, which is the lowest concentration known to generate behavioural modifications in fish in earlier studies30. Based on this, each trial in the migration study (see below) was limited to 70 h. Water samples were taken continuously throughout the study to monitor oxazepam concentrations.
Laboratory based migration assay
After exposure, fish were moved into a migration study pool (diameter 8 m, depth 1 m, see Fig. 6). Two pools, one per treatment, were used and treatments were shifted between pools for every trial to avoid pool-generated effects. As with the rearing pools, the study pools were continuously supplied with water from the Ume River, keeping water temperatures identical to the natural regime of the season. The continuous flowthrough of water created a unidirectional water current in the pools, with water circulating alongside the walls. The centre of each pool was enclosed, restricting fish from entering the central area (Fig. 6). A ‘dent’ in the enclosed area created a back eddy, which served as a refuge for the fish from the current. At the narrow passage between the enclosed area and the wall of the pool, two PIT-tag antennae, 3 m apart, were installed and connected to a reader (Biomark/Allflex RM310). Fish passing through these antennae were registered on the reader. Each registration included the tag ID (unique for each fish) and the time and date of detection. The reader collected data non-stop during the study period and a higher number of detections indicate higher migration intensity. A similar circular migration-pool setup has successfully been used in earlier studies of migration behaviour in Atlantic salmon with regards to swimming intensity and speed, and motivation to migrate52,54,55,56,57. After the experiment, all fish were released into the Ume River.
The centre of the rearing tank (diameter 8 m) is enclosed, restricting fish from entering the area. At the narrowest passages between the enclosed area and the tank wall, two PIT-antennas were installed, connected to a reader station. A ‘dent’ in the enclosed area created a back eddy, serving as a refuge for the fish from the water current. Schematic drawing by G Hellström.
Analysis
Salmon were allowed to acclimatize for 30 min in the pools; only data recorded after the acclimation period were used in subsequent analysis. The difference in downstream migration intensity between exposed and control smolt was analysed using a general LME model, treating number of detections per fish as response variable and treatment (exposed/control) as a two-level factor. Trial nested in age was included as a random effect structure, to account for dependence between observations within age and trial. Gaussian error was used, as both the response variable and the model residuals were found to be normally distributed by visual inspection. Statistical inference on the treatment effect was done using Wald test. To investigate the dependency of time on the effect of Treatment on migration intensity, difference between exposed and control fish was tested for the first 10 h (0–10) and the last 10 h (60–70) using a similar model structure as above, but also including the interaction term ‘treatment × time’ (10 first/10 last hours). To investigate the continuous decline of migration intensity over time and in relation to the elimination rate of oxazepam from muscle tissue, a LOESS smoother58 was fitted for each treatment and graphically compared in a plot, having time (0–70 h) on the x axis and number of detections per hour (migration intensity) on the y axis. A LOESS smoother was also used to investigate difference between treatments in diurnal migration intensity using hour of day on the x axis and number of detections (migration intensity) on the y axis. To more formally determine the form of the relationship between migration intensity and hour of day, multi-order polynomial regression models were fitted to the data, and the most parsimonious of these models was determined using Akaike information criterion-based model-selection.
Potential differences between treatments in mass, length and Fultons condition index59 were assessed using one-way analysis of variance. All analyses were performed in the statistical program R60, using the package nlme61.
Field-based migration assay
The field study was carried out in a small stream (0.5 m3 s−1) that enters into Ume river close to the hatchery (63°52′ N; 20°01′ E, Supplementary Fig. S3). The procedure to select and tag smolt was identical to the laboratory study described above. For exposure 200 1-year old smolts (length mean±1 s.d.:=114±15 mm, weight mean±1 s.d.:=14.8±4.3 g) were evenly distributed into four 1 × 1-m tanks, resulting in 50 smolts per tank. Fish in two of the tanks were exposed to 1.9 μg l−1 oxazepam, while the other two were kept as controls containing unexposed fish in river water. After a seven-day exposure period, the smolts were released into a small stream pool 300 m upstream from where the stream enters the river. A migration barrier immediately upstream the pool prevented the fish from moving further upstream. Fifty smolts form each treatment were released at two occasions, at 09:30 and 11:30 on 27 May 2015. A PIT-tag antenna connected to a reader (Biomark HPR Plus) was installed 200 m downstream of the release pool one week before releasing the smolts (Supplementary Fig. S3). The antenna covered the entire width of the stream, and, hence, all migrating smolts had to pass through the antenna on their way downstream. The antenna was removed 3 days after the smolts were released, and during those three days 39% of the control and 71% of the exposed smolts had migrated pass the antenna. Smolts that had not been detected during that time were not considered in subsequent analysis.
Time from release to first detection on the antenna was treated as a normal distributed response variable in a general linear mixed effect model, having Treatment as a two-level fixed effect (exposed, control) and Release occasion as a two-level random effect (first and second release occasion).
Data Availability
The data supporting this study are available on request from the corresponding author
Additional information
How to cite this article: Hellström, G. et al. GABAergic anxiolytic drug in water increases migration behaviour in salmon. Nat. Commun. 7, 13460 doi: 10.1038/ncomms13460 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Noakes, D. in: Ecology of Atlantic Salmon and Brown Trout (eds Jonsson B., Jonsson N Springer (2011).
Windsor, M. in: Atlantic Salmon Ecology (eds Aas O., Einum S., Klemetsen A., Skurdal J. Wiley-Blackwell (2010).
Klemetsen, A. et al. Atlantic salmon Salmo salar L., brown trout Salmo trutta L. and Arctic charr Salvelinus alpinus (L.): a review of aspects of their life histories. Ecol. Freshw. Fish 12, 1–59 (2003).
McCormick, S. D., Hansen, L. P., Quinn, T. P. & Saunders, R. L. Movement, migration, and smolting of Atlantic salmon (Salmo salar). Can. J. Fish. Aquat. Sci. 55, 77–92 (1998).
Hindar, K., Hutchings, J., Diserud, O. & Fiske, P. in: Atlantic Salmon Ecology (eds Aas O., Einum S., Klementsen A., Skurdal J. 299–325Wiley-Blackwell (2011).
Thorstad, E. B. et al. A critical life stage of the Atlantic salmon Salmo salar: behaviour and survival during the smolt and initial post-smolt migration. J. Fish Biol. 81, 500–542 (2012).
McCormick, S. D. & Saunders, R. L. Preparatory physiological adaptations for marine life of salmonids: osmoregulation, growth, and metabolism. Am. Fish. Soc. Symp. 1, 1–229 (1987).
Stich, D. S., Kinnison, M. T., Kocik, J. F. & Zydlewski, J. D. Initiation of migration and movement rates of Atlantic salmon smolts in fresh water. Can. J. Fish. Aquat. Sci. 72, 1339–1351 (2015).
Thorstad, E. B., Whoriskey, F. G., Rikardsen, A. H. & Aarestrup, K. in: Atlantic Salmon Ecology (eds Aas O., Einum S., Klementsen A., Skurdal J. 1–23Wiley-Blackwell (2011).
Ward, D. & Hvidsten, N. in: Atlantic Salmon Ecology (eds Aas O., Einum S., Klementsen A., Skurdal J. 199–215Wiley-Blackwell (2011).
Chapman, B. B. et al. To boldly go: individual differences in boldness influence migratory tendency: boldness influences migratory tendency. Ecol. Lett. 14, 871–876 (2011).
Brown, J. S., Laundré, J. W. & Gurung, M. The ecology of fear: optimal foraging, game theory, and trophic interactions. J. Mammal. 80, 385–399 (1999).
Clinchy, M., Sheriff, M. J. & Zanette, L. Y. Predator-induced stress and the ecology of fear. Funct. Ecol. 27, 56–65 (2013).
Pritchett, D. B. et al. Importance of a novel GABAA receptor subunit for benzodiazepine pharmacology. Nature 338, 582–585 (1989).
Gunnarsson, L., Jauhiainen, A., Kristiansson, E., Nerman, O. & Larsson, D. J. Evolutionary conservation of human drug targets in organisms used for environmental risk assessments. Environ. Sci. Technol. 42, 5807–5813 (2008).
Bourin, M., Petit-Demoulière, B., Nic Dhonnchadha, B. & Hascöet, M. Animal models of anxiety in mice. Fundam. Clin. Pharmacol. 21, 567–574 (2007).
Brodin, T. et al. Ecological effects of pharmaceuticals in aquatic systems-impacts through behavioural alterations. Philos. Trans. R. Soc. B Biol. Sci 369, 20130580 (2014).
Braithwaite, V. A., Huntingford, F. & van den Bos, R. Variation in emotion and cognition among fishes. J. Agric. Environ. Ethics 26, 7–23 (2013).
Rose, J. D. et al. Can fish really feel pain? Fish Fish. 15, 97–133 (2014).
Munday, P. L. Climate change ecology: Salmon behaving badly. Nat. Clim. Change 5, 915–916 (2015).
Quinn, T. P. The Behavior and Ecology of Pacific Salmon and Trout UBC Press (2005).
Maximino, C. et al. Measuring anxiety in zebrafish: a critical review. Behav. Brain Res. 214, 157–171 (2010).
Richendrfer, H., Pelkowski, S. D., Colwill, R. M. & Creton, R. On the edge: pharmacological evidence for anxiety-related behavior in zebrafish larvae. Behav. Brain Res. 228, 99–106 (2012).
Brodin, T., Fick, J., Jonsson, M. & Klaminder, J. Dilute concentrations of a psychiatric drug alter behavior of fish from natural populations. Science 339, 814–815 (2013).
Ou, M. et al. Responses of pink salmon to CO2-induced aquatic acidification. Nat. Clim. Change 5, 950–955 (2015).
Todd, C. D. et al. Phenological and phenotypic changes in Atlantic salmon populations in response to a changing climate. ICES J. Mar. Sci. 69, 1686–1698 (2012).
Packard, A. & Delafield-Butt, J. T. Feelings as agents of selection: putting Charles Darwin back into (extended neo-) Darwinism. Biol. J. Linn. Soc. 112, 332–353 (2014).
Odling-Smee, L., Simpson, S. D. & Braithwaite, V. A. The role of learning in fish orientation. Fish Cogn. Behav. 166–185 (2011).
Nilsson, G. E. et al. Near-future carbon dioxide levels alter fish behaviour by interfering with neurotransmitter function. Nat. Clim. Change 2, 201–204 (2012).
Klaminder, J., Jonsson, M., Fick, J., Sundelin, A. & Brodin, T. The conceptual imperfection of aquatic risk assessment tests: highlighting the need for tests designed to detect therapeutic effects of pharmaceutical contaminants. Environ. Res. Lett. 9, 084003 (2014).
Loos, R. et al. EU-wide monitoring survey on emerging polar organic contaminants in wastewater treatment plant effluents. Water Res. 47, 6475–6487 (2013).
Meador, J. P., Yeh, A., Young, G. & Gallagher, E. P. Contaminants of emerging concern in a large temperate estuary. Environ. Pollut. 213, 254–267 (2016).
World Health Organization. in The World Medicines Situation (eds Hoebert, J., Laing, R. & Stephens, P.) 1–20 (Geneva, 2011).
Klaminder, J. et al. Long-term persistence of an anxiolytic drug (oxazepam) in a large freshwater lake. Environ. Sci. Technol. 49, 10406–10412 (2015).
Heynen, M., Fick, J., Jonsson, M., Klaminder, J. & Brodin, T. Effect of bioconcentration and trophic transfer on realized exposure to oxazepam in two predators, the dragonfly larvae ( Aeshna grandis ) and the Eurasian perch ( Perca fluviatilis ): species-specific bioconcentration and trophic transfer. Environ. Toxicol. Chem. 35, 930–937 (2016).
Arnold, K. E., Brown, A. R., Ankley, G. T. & Sumpter, J. P. Medicating the environment: assessing risks of pharmaceuticals to wildlife and ecosystems. Philos. Trans. R. Soc. B Biol. Sci 369, 11 (2014).
Hamilton, T. J., Holcombe, A. & Tresguerres, M. CO2-induced ocean acidification increases anxiety in Rockfish via alteration of GABA(A) receptor functioning. Proc. R. Soc. B Biol. Sci. 281, 7 (2014).
Munday, P. L., Cheal, A. J., Dixson, D. L., Rummer, J. L. & Fabricius, K. E. Behavioural impairment in reef fishes caused by ocean acidification at CO2 seeps. Nat. Clim. Change 4, 487–492 (2014).
Rosseland, B. O. & Kroglund, F. in: Atlantic Salmon Ecology (eds Aas O., Einum S., Klementsen A., Skurdal J. Wiley-Blackwell (2011).
Regan, M. D. et al. Ambient CO2, fish behaviour and altered GABAergic neurotransmission: exploring the mechanism of CO2-altered behaviour by taking a hypercapnia dweller down to low CO2 levels. J. Exp. Biol. 219, 109–118 (2016).
Cowx, I. G. Stocking and Introduction of Fish Fishing News Books (1998).
Romakkaniemi, A. Conservation of Atlantic Salmon by Supplementary Stocking of Juvenile Fish. PhD thesis University of Helsinki, (2008).
Bacon, P. J. et al. Can conservation stocking enhance juvenile emigrant production in wild atlantic salmon? Trans. Am. Fish. Soc. 144, 642–654 (2015).
Saltveit, S. J. The effects of stocking Atlantic salmon, Salmo salar, in a Norwegian regulated river. Fish. Manag. Ecol. 13, 197–205 (2006).
Kekäläinen, J., Niva, T. & Huuskonen, H. Pike predation on hatchery-reared Atlantic salmon smolts in a northern Baltic river. Ecol. Freshw. Fish 17, 100–109 (2008).
Lans, L. et al. The effects of ration size on migration by hatchery-raised Atlantic salmon (Salmo salar) and brown trout (Salmo trutta): effects of ration size on migration. Ecol. Freshw. Fish 20, 548–557 (2011).
Anttila, K., Jokikokko, E., Erkinaro, J., Järvilehto, M. & Mänttäri, S. Effects of training on functional variables of muscles in reared Atlantic salmon Salmo salar smolts: connection to downstream migration pattern. J. Fish Biol. 78, 552–566 (2011).
Hyvärinen, P. & Rodewald, P. Enriched rearing improves survival of hatchery-reared Atlantic salmon smolts during migration in the River Tornionjoki. Can. J. Fish. Aquat. Sci. 70, 1386–1395 (2013).
Norrgård, J. R., Bergman, E., Schmitz, M. & Greenberg, L. A. Effects of feeding regimes and early maturation on migratory behaviour of landlocked hatchery-reared Atlantic salmon Salmo salar smolts: migration of hatchery-reared salmo salar. J. Fish Biol. 85, 1060–1073 (2014).
Hellström, G., Klaminder, J., Jonsson, M., Fick, J. & Brodin, T. Upscaling behavioural studies to the field using acoustic telemetry. Aquat. Toxicol. 170, 384–389 (2015).
Jonsson, N. & Jonsson, B. Time and size at seaward migration influence the sea survival of Salmo salar. J. Fish Biol. 84, 1457–1473 (2014).
Martin, P. et al. Experimental study of the influence of photoperiod and temperature on the swimming behaviour of hatchery-reared Atlantic salmon (Salmo salar L.) smolts. Aquaculture 362, 200–208 (2012).
Ostrand, K., Zydlewski, G., Gale, W. & Zydlewski, J. in: Advances in Fish Tagging and Marking Technology (eds Jeremy M.et al. American Fisheries Society (2012).
Fangstam, H. Individual downstream swimming speed during the natural smolting period among young of baltic salmon (salmo-salar). Can. J. Zool. 71, 1782–1786 (1993).
Fangstam, H., Berglund, I., Sjoberg, M. & Lundqvist, H. Effects of size and early sexual maturity on downstream migration during smolting in baltic salmon (salmo-salar). J. Fish Biol. 43, 517–529 (1993).
Vainikka, A. et al. Food restriction prior to release reduces precocious maturity and improves migration tendency of Atlantic salmon ( Salmo salar ) smolts. Can. J. Fish. Aquat. Sci. 69, 1981–1993 (2012).
Pirhonen, J., Forsman, L., Soivio, A. & Thorpe, J. Movements of hatchery reared Salmo trutta during the smolting period, under experimental conditions. Aquaculture 168, 27–40 (1998).
Cleveland, W., Grosse, E. & Shyu, W. in: Statistical Models in S (eds Chambers J., Hastie T. Wadsworth & Brooks (1992).
Anderson, R. O. & Neumann, R. M. in: Fisheries techniques 2nd edition (eds Murphy B. R., Willis D. W. American Fisheries Society (1996).
R Core Team. R: A Language and Environment for Statistiscal Computing R Foundation for Statistical Computing (2015).
Pinheiro, J., Bates, D., DebRoy, S., Sarkar, D. & R, Develoment Core Team. nlme: Linear and Nonlinear Mixed Effects Models (2013).
Acknowledgements
Financial support was provided by the Foundation for Strategic Environmental Research (MISTRA) (J.F.), the Trygger foundation (T.B.), the Swedish Research Council Formas (T.B. - 2013-734), the Swedish Research Council VR (M.J.), Kempestiftelserna (J.K.) and the Knut and Alice Wallenberg foundation (J.K.). We also thank Dr Kjell Leonardsson, Swedish University of Agricultrual Sciences, for letting us use data on wild smolt migration.
Author information
Authors and Affiliations
Contributions
G.H. designed and conducted the study, and wrote the majority of the paper, under the supervision of J.K. and T.B. F.F. and L.P. assisted with the laboratory- and field-based behaviour assays, while J.F. conducted the chemical analyses. A.A., M.J. and L.P. contributed substantially during the writing process and the statistical analyses.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-3, Supplementary Table 1 (PDF 836 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Hellström, G., Klaminder, J., Finn, F. et al. GABAergic anxiolytic drug in water increases migration behaviour in salmon. Nat Commun 7, 13460 (2016). https://doi.org/10.1038/ncomms13460
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms13460
This article is cited by
-
Enhancing our understanding of fish movement ecology through interdisciplinary and cross-boundary research
Reviews in Fish Biology and Fisheries (2022)
-
Electrochemical degradation of psychotropic pharmaceutical compounds from municipal wastewater and neurotoxicity evaluations
Environmental Science and Pollution Research (2021)
-
Environmental risk assessment of psychoactive drugs in the aquatic environment
Environmental Science and Pollution Research (2019)
-
Studying behavioural variation in salmonids from an ecological perspective: observations questions methodological considerations
Reviews in Fish Biology and Fisheries (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.