Abstract
Microscopy of biological specimens often requires low light levels to avoid damage. This yields images impaired by shot noise. An improved measurement accuracy at the Heisenberg limit can be achieved exploiting quantum correlations. If sample damage is the limiting resource, an equivalent limit can be reached by passing photons through a specimen multiple times sequentially. Here we use self-imaging cavities and employ a temporal post-selection scheme to present full-field multi-pass polarization and transmission micrographs with variance reductions of 4.4±0.8 dB (11.6±0.8 dB in a lossless setup) and 4.8±0.8 dB, respectively, compared with the single-pass shot-noise limit. If the accuracy is limited by the number of detected probe particles, our measurements show a variance reduction of 25.9±0.9 dB. The contrast enhancement capabilities in imaging and in diffraction studies are demonstrated with nanostructured samples and with embryonic kidney 293T cells. This approach to Heisenberg-limited microscopy does not rely on quantum state engineering.
Similar content being viewed by others
Introduction
Quantum enhanced metrology allows sub-shot noise measurements by exploiting quantum correlations between probe particles1. This has been demonstrated in microscopy in scanning configurations applying N00N states2,3 or squeezed light4. Full-field shadow imaging was demonstrated using entangled photons from parametric down-conversion5. Experimentally, these studies relied on postselection and the reduction in variance was <3.3 dB, mainly due to the difficulties in creating the necessary correlations between the photons. On the other hand, it has also been shown that, under conditions of equivalent sample damage, a single probe particle that interacts m times sequentially with the sample can be used to reach the same (Heisenberg) noise limit, and that this represents an optimal parameter estimation strategy6,7,8. In this way a variance reduction of >10 dB was achieved in a phase-shift measurement9. Contrast enhancement in full-field double-pass transmission microscopy was demonstrated using a phase-conjugated mirror to pass light twice through a sample10.
In the following, we generalize these techniques to full-field multi-pass microscopy by placing a sample in a self-imaging cavity11,12,13. The setup allows us to form an image of enhanced contrast by re-imaging a pulse of light m times onto the sample. Although continuous wave cavity-enhanced techniques have increased measurement resolution in various fields of science, recently, for example, at LIGO14 or in a scanning cavity microscope15, counting the exact number of interactions allows for a precise parameter estimation and an enhanced sensitivity also for phase shifts larger than  . At a constant number of photon sample interactions and employing a temporal postselection scheme, we show both retardance and transmission measurements with a sensitivity beyond the single-pass shot-noise limit, which we define as the resolution obtained in our setup using only a single pass through the sample. It is limited by the shot noise on the number of detected photons. We show micrographs of nanostructured and biological samples, as well as the signal-enhancing capabilities in diffraction studies.
. At a constant number of photon sample interactions and employing a temporal postselection scheme, we show both retardance and transmission measurements with a sensitivity beyond the single-pass shot-noise limit, which we define as the resolution obtained in our setup using only a single pass through the sample. It is limited by the shot noise on the number of detected photons. We show micrographs of nanostructured and biological samples, as well as the signal-enhancing capabilities in diffraction studies.
Results
The setup
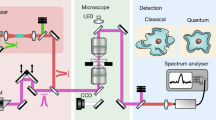
Our setup is depicted in Fig. 1. A pulse of light (see Methods) is coupled into the cavity via the in-coupling mirror Mi. Four lenses L1–4 form a microscope on either side of the sample. After the first light–sample interaction, an image is formed on the out-coupling mirror Mo. Most of the light is reflected back onto the sample, which is now illuminated by an image of itself. An image of enhanced contrast will then be formed on Mi and will again be reflected onto the sample. This process is repeated multiple times. Every time an image is formed on Mo, a fraction of the light is out-coupled and imaged onto a gated intensified CCD (charge-coupled device) camera. The gating time of the CCD camera is much shorter than the cavity roundtrip time (<500 ps and 2.7 ns, respectively), such that light can be postselected that interacted with the sample exactly m times.
A pulse of light (indicated in magenta) from a single-mode fibre (SMF) is collimated and enters the self-imaging cavity through the in-coupling mirror Mi. The lenses L1 and L2 (L3 and L4, focal length f) form a microscope on the left (right) side of the sample S. Light scattered in the sample plane is indicated in turquoise. After each interaction an image is formed on either Mi or Mo and reimaged onto the sample. At Mo, a fraction of the light is out-coupled and imaged using a microscope objective (Obj.) onto a gated camera (ICCD). Diffraction patterns can be imaged in the Fourier plane F. For polarization microscopy crossed polarizers are added (Pi and Po). The blue trace shows the integrated detected intensity as a function of delay, overlaid with simulated images corresponding to the respective intensity peaks.
Polarization microscopy
To demonstrate contrast enhancement and sub-shot-noise imaging, a wedged quartz-silica depolarizer is placed in the sample plane S. Every interaction with the quartz crystal leads to a position-dependent rotation of the polarization vector on the Poincaré sphere. For a properly cut and oriented quartz crystal (see Methods), the detected number of photons in a cross-polarized setup is expected to be  , where Nm,0(x, y) is the number of photons detected without the polarization analyser Po. η(x) is the retardance of the sample, which is proportional to the local thickness of the wedged quartz crystal. Although on a single interaction the transmitted intensity varies slowly across the field of view, more rapid signal oscillations are observed for m=3, 13 and 29 interactions (Fig. 2a). It is noteworthy that the ambiguity that arises from having m fringes instead of a single fringe can be resolved by acquiring images for different numbers of interactions.
, where Nm,0(x, y) is the number of photons detected without the polarization analyser Po. η(x) is the retardance of the sample, which is proportional to the local thickness of the wedged quartz crystal. Although on a single interaction the transmitted intensity varies slowly across the field of view, more rapid signal oscillations are observed for m=3, 13 and 29 interactions (Fig. 2a). It is noteworthy that the ambiguity that arises from having m fringes instead of a single fringe can be resolved by acquiring images for different numbers of interactions.
(a) Micrographs after m=1,3,13 and 29 interactions (scale bar, 250 μm) with the quartz-silica depolarizer. The inset data are taken without crossed polarizer and shows a dark spot, probably a piece of dust, reimaged onto itself m times. (b) The measured number of photons (normalized) per column (left) agrees well with the expected signal (right). (c) Experimental and calculated (solid lines) variance reduction in multipass microscopy for different numbers of collected photons (see text). The error bars give the s.d. of the variance (see Methods).
For each image in Fig. 2a, a total number of Nm=∑x∑yNm(x, y)=1.4 × 106 photons were detected. Figure 2b compares the measured (left) and calculated (right) ratio ∑yNm(x, y)/∑yNm,0(x, y), where the summation is carried out in between the dashed lines in Fig. 2a. The visibility of the intensity modulation is reduced from 0.98 after 1 interaction to 0.52 after 29 interactions. As a figure of merit (FOM) for multi-pass microscopy, we define the reduction in variance of the retardance measurement  , where Δηm is the s.d. of ηm, which can be obtained from the measured s.d. of Nm(x)=∑yNm(x, y) via error propagation as
, where Δηm is the s.d. of ηm, which can be obtained from the measured s.d. of Nm(x)=∑yNm(x, y) via error propagation as  . The FOM at
. The FOM at  is plotted in Fig. 2c (grey data points) and shows a variance reduction by 25.9±0.9 dB after 27 interactions. This is slightly lower than the theoretically expected value FOM=m2 (grey line, see Methods) due to residual birefringence of the optical setup, reflections and misalignment. This variance reduction has been obtained for a constant number of detected photons. It is the relevant FOM if the accuracy of a measurement is limited by a finite detection rate, for example, due to the dead time of a detector. Assuming a low loss setup, it is also the relevant FOM if the number of detected photons is limited by either the collection and detection efficiency or by the number of available probe particles.
is plotted in Fig. 2c (grey data points) and shows a variance reduction by 25.9±0.9 dB after 27 interactions. This is slightly lower than the theoretically expected value FOM=m2 (grey line, see Methods) due to residual birefringence of the optical setup, reflections and misalignment. This variance reduction has been obtained for a constant number of detected photons. It is the relevant FOM if the accuracy of a measurement is limited by a finite detection rate, for example, due to the dead time of a detector. Assuming a low loss setup, it is also the relevant FOM if the number of detected photons is limited by either the collection and detection efficiency or by the number of available probe particles.
Multi-pass microscopy also offers an advantage when the number of probe particle–sample interactions has to be limited due to probe particle-induced damage. Under this constant damage condition, assuming a lossless cavity and sample, a FOM=m is expected (red line, see Methods). The total number of detected photons now depends on the number of roundtrips  such that the total number of interactions is independent of m. This implies an m-fold damage reduction if Δη is kept constant. αPo,m gives the fraction of light blocked by the polarization analyser Po and is close to
such that the total number of interactions is independent of m. This implies an m-fold damage reduction if Δη is kept constant. αPo,m gives the fraction of light blocked by the polarization analyser Po and is close to  .The measurements (red data points) show a maximum variance reduction of 11.6±0.8 dB, which is observed after 25 interactions. It is noteworthy that under the assumption of constant damage, the measurement error for a single multi-passed particle is equivalent to the error obtained in a Heisenberg-limited measurement with m entangled particles and a single pass.
.The measurements (red data points) show a maximum variance reduction of 11.6±0.8 dB, which is observed after 25 interactions. It is noteworthy that under the assumption of constant damage, the measurement error for a single multi-passed particle is equivalent to the error obtained in a Heisenberg-limited measurement with m entangled particles and a single pass.
Photon loss due to absorption, reflection or scattering in the sample (αs=1−ts=0.039 per interaction) or in the cavity (αc=1−tc=0.189 per interaction) reduce the efficiency of multi-pass microscopy, as only a total number of  photons will be detected for operation at constant damage. Here, t=tcts=1−α and mα<m is the mean number of interactions of a photon with the sample before it is either lost or the mth interaction is reached (see Methods). Taking this into account, a maximum variance reduction of 4.4±0.8 dB after three interactions is observed (blue data). This could be improved significantly in an optimized setup, as the losses in the cavity optics are mainly due to the finite reflectivity of the in- and out-coupling mirror, which, for this proof-of-principle experiment, were chosen to out-couple a considerable fraction of the beam intensity after every interaction.
photons will be detected for operation at constant damage. Here, t=tcts=1−α and mα<m is the mean number of interactions of a photon with the sample before it is either lost or the mth interaction is reached (see Methods). Taking this into account, a maximum variance reduction of 4.4±0.8 dB after three interactions is observed (blue data). This could be improved significantly in an optimized setup, as the losses in the cavity optics are mainly due to the finite reflectivity of the in- and out-coupling mirror, which, for this proof-of-principle experiment, were chosen to out-couple a considerable fraction of the beam intensity after every interaction.
Transmission measurements and diffraction studies
Transmission measurements can also benefit from multi-passing as long as the total losses due to the sample and the cavity are small (see Methods). Measuring the optical density of a spatially homogeneous sample shows signal amplification and a variance reduction by 4.8±0.8 dB (Fig. 3a, again at constant damage). Owing to the photon losses in the sample and in the cavity no further decrease in variance is observed when the number of interactions is increased to beyond 7.
(a) Photon counting measurements of an optical density (OD) filter show a linear growth of the OD with m and a variance reduction at constant damage of up to 4.8±0.8 dB (see Methods). (b) Contrast enhancement in multi-pass micrographs of a micro-structured carbon membrane (normalized to  ) as well as in the respective diffraction patterns (c) (normalized to the central peak). (d) Multi-pass micrographs of embryonic kidney 293T cells. Scale bar, 20 μm, 10 mm−1 and 20 μm for b–d, respectively.
) as well as in the respective diffraction patterns (c) (normalized to the central peak). (d) Multi-pass micrographs of embryonic kidney 293T cells. Scale bar, 20 μm, 10 mm−1 and 20 μm for b–d, respectively.
The imaging and contrast-enhancement capabilities of the setup are further exemplified with microfabricated grating structures and with embryonic kidney 293T cells. Figure 3b shows multi-pass (m=1, 3, 5) micrographs of a hexagonal hole pattern in a carbon membrane. The resolution of the microscope is ∼5 μm due to the finite numerical aperture of the employed lenses (f=50 mm). The photon loss due to the carbon membrane is amplified by multiple passes, which leads to a significant contrast enhancement. This also becomes apparent in the images shown in Fig. 3c, which were taken in the Fourier plane of the imaging optics. Although the single pass image is dominated by the diffraction pattern caused by the four-sided copper support structure of the carbon membrane, the hexagonal symmetry of the holes in the membrane becomes clearly visible after multiple interactions. In addition, for embryonic kidney 293T cells a clear contrast enhancement is observed (Fig. 3d). Although cells and other phase objects are hardly visible in single pass bright-field microscopy, the outline of single cells becomes clearly visible in multi-pass microscopy images. Both absorption and phase shifts need to be taken into account in the analysis of such images and future high-resolution multi-pass microscopy studies will aim at exploring fine details in the interior of cells.
Discussion
Multi-pass microscopy is a technique for signal amplification and allows for optimal parameter estimation in the presence of noise sources that are not significantly amplified by multi-passing (such as shot noise or read noise). For the images acquired at constant damage, the technique presented above relies on temporal post-selection of a few percent of the light that interacted with the sample. Although this degree of postselection is orders of magnitude less stringent than the postselection required in comparable N00N-state microscopy experiments, we note that there are at least two technologically feasible ways of overcoming this need for postselection: on the one hand, an optical switch could be incorporated into the cavity, which would allow for out-coupling all light at once. This could be achieved using a Pockels cell and a polarizing beam splitter. On the other hand, even without an out-coupling switch a metrological advantage can be gained, if a detector is used that records the detection time, and thus the number of interactions, of each individual photon.
Postselection free multi-pass microscopy will benefit applications that are sensitive to photo-induced damage, such as live-cell microscopy16, the label-free detection of single proteins17, the detection of single molecules via their absorption18 or low damage imaging of exotic quantum states of matter19. Apart from increasing the detectability of such weak signals17,18, an improved signal-to-noise ratio would also lead to better spatial accuracy in super-resolution techniques that rely on these weak signals. Increased accuracy is also obtained if a measurement is limited by the number of detectable particles, for example, at wavelengths where there are no high-intensity light sources, or also in measurements that involve massive particles as probe particles (electron or ion microscopy, measurements involving anti-matter). Multi-pass electron microscopy especially seems worth considering, as sample damage limits the spatial resolution achieved in imaging biological specimens20,21.
Methods
Experimental setup
Two different laser systems were used for the experiments:
For the data in Fig. 3, a titanium sapphire laser was used (Venteon, 10 fs pulse width, 5 nJ per pulse, 100 MHz repetition rate, spectrally centred at λ=780 nm). The pulses are spectrally filtered (Semrock LL01-810-12.5) to reduce the effect of chromatic aberrations in the self-imaging cavity. This inevitably broadens the temporal width of the pulses; however, the pulses are still short compared with the cavity round trip time. A resonantly driven electro-optic modulator is used to reduce the laser repetition rate to 50 MHz with an extinction ratio of about 16 dB.
A lower repetition rate laser was required for the data in Fig. 2, to allow for more interactions before the advent of the consecutive laser pulse. The laser diode (783 nm) was driven with fast voltage pulses. Pulse widths <1 ns at a peak power of about 100 mW were achieved. The repetition rate was controlled using a direct digital synthesizer (Novatech 409B) and set to 25 MHz.
The alignment of the self-imaging cavity is done element by element (starting from the out-coupling mirror) by maximizing the light that is back-reflected into the single mode fibre.
Jones matrix representation of quartz-silica depolarizer
The quartz-silica depolarizer consists of a wedged plate of optical quartz cemented to a wedged plate of synthetic fused silica (OptoSigma DEQ 2S). The quartz crystal is cut and oriented such that it has the fast axis at a 45° angle with respect to the polarization of the incoming beam. The fused silica wedge has negligible birefringence and prevents beam deviation. The Jones matrix of the quartz crystal can be written as  , where η is the phase retardance due to the birefringence of the crystal. The retardance is proportional to the thickness of the crystal, which varies spatially, as the crystal is cut with a wedge angle of β∼2°: η∼βΔxΔn/λ, where Δn∼0.009 is the difference in index of refraction of light polarized along the fast or slow axis of the crystal. In the experiment horizontally polarized light
, where η is the phase retardance due to the birefringence of the crystal. The retardance is proportional to the thickness of the crystal, which varies spatially, as the crystal is cut with a wedge angle of β∼2°: η∼βΔxΔn/λ, where Δn∼0.009 is the difference in index of refraction of light polarized along the fast or slow axis of the crystal. In the experiment horizontally polarized light  enters the multi-pass microscope (I0=|E0|2 is the intensity of the incoming light), interacts with the quartz crystal m times and is finally projected onto the vertical polarization axis and detected. This can be written as
enters the multi-pass microscope (I0=|E0|2 is the intensity of the incoming light), interacts with the quartz crystal m times and is finally projected onto the vertical polarization axis and detected. This can be written as  .
.
Measurement error in retardance measurements
The error in retardance measurements is obtained from error propagation as  . For a constant number of detected particles the s.d.
. For a constant number of detected particles the s.d.  is independent of m, whereas the slope
is independent of m, whereas the slope  increases linearly with m. This leads to a
increases linearly with m. This leads to a  , which scales with the square of the number of roundtrips. If the number of probe particle–sample interactions is to be kept constant, for a lossless setup and sample,
, which scales with the square of the number of roundtrips. If the number of probe particle–sample interactions is to be kept constant, for a lossless setup and sample,  and
and  . The slope
. The slope  would still increase linearly with m, leading to a
would still increase linearly with m, leading to a  . For
. For  , the setup could be adapted to reach optimal sensitivity3.
, the setup could be adapted to reach optimal sensitivity3.
Measurement error in absorption measurements
Assume N0,m photons are used to probe the transmission of a sample. After m interactions, all photons are out-coupled from the cavity and detected with a photon counting detector. Without the sample,  photons will be detected. Uncorrelated repetitions of the measurement of Nwo will give a s.d. of
photons will be detected. Uncorrelated repetitions of the measurement of Nwo will give a s.d. of  . With a partially absorptive sample in the setup,
. With a partially absorptive sample in the setup,  photons will be detected with a s.d. of
photons will be detected with a s.d. of  . It follows that
. It follows that  and error propagation yields a s.d. of the measured sample transmission of
and error propagation yields a s.d. of the measured sample transmission of  . We are interested in the s.d. as a function of m at constant damage. This implies that the number of in-coupled photons N0,m will be a function of m, such that the number of absorbed photons Dm is independent of m. For a symmetric setup, in which the cavity losses are the same on both sides of the sample, we get
. We are interested in the s.d. as a function of m at constant damage. This implies that the number of in-coupled photons N0,m will be a function of m, such that the number of absorbed photons Dm is independent of m. For a symmetric setup, in which the cavity losses are the same on both sides of the sample, we get  and Dm=D1 yields
and Dm=D1 yields  and
and

For mα<<1, this yields a FOM of multi-pass absorption microscopy  that scales linearly with m, just as it did for the retardance measurements.
that scales linearly with m, just as it did for the retardance measurements.
When studying living samples, it might be more important to look at the damage reduction at constant s.d. (Δts,1=Δts,m, but Dm≠D1), which yields

and scales with m for mα<<1.
Image acquisition and noise analysis
To assess the single-pass shot-noise limit, the spatially resolved photon counting capabilities of the ICCD camera are exploited. it is noteworthy that they are not required to benefit from multipass microscopy.
For the retardance measurements in Fig. 2, the exposure is chosen such that ∼140 photons are detected per image. There are fewer than one dark counts per image. For each value of m, 10,000 images were acquired. This total data set, containing roughly 1.4 × 106 detected photons, is divided into Np=100 images, each of them containing Nm photons, with N1=1.4 × 104, which were randomly picked without replacement. For each of these Np images, the  , where
, where  , is calculated and plotted in Fig. 2c. For this calculation, the slope
, is calculated and plotted in Fig. 2c. For this calculation, the slope  is obtained from a sinusoidal fit to the sum of all Np images. The above equation can be rewritten as
is obtained from a sinusoidal fit to the sum of all Np images. The above equation can be rewritten as  and, as the images are independent of each other,
and, as the images are independent of each other,  . This allows us to calculate22 the s.d. of the variance as
. This allows us to calculate22 the s.d. of the variance as  , with μ2 and μ4 being the second and fourth central moment of the distribution of values
, with μ2 and μ4 being the second and fourth central moment of the distribution of values  . The s.d. of the
. The s.d. of the  is then obtained from propagating the errors in (Δηm)2 and (Δη1)2.
is then obtained from propagating the errors in (Δηm)2 and (Δη1)2.
The equations for the calculation of Nm that are given in the main text assume a symmetric setup, with equal loss αc on both sides of the sample plane. For the data and theory shown in Fig. 2, the asymmetry due to the unequal reflectance of Mi and Mo is taken into account when calculating Nm and mα. With αi=1−ti and αo=1−to being the total losses on the in-coupling and out-coupling side of the sample, respectively, we get  and
and 
 , respectively. As the images in Fig. 2 are centred at
, respectively. As the images in Fig. 2 are centred at  , the ratio
, the ratio  , with a maximum measured deviation of 0.04. For the theoretical curves in Fig. 2c,
, with a maximum measured deviation of 0.04. For the theoretical curves in Fig. 2c,  was assumed for even numbers of interactions, for which αPo,m could not be determined experimentally.
was assumed for even numbers of interactions, for which αPo,m could not be determined experimentally.
For the optical density measurements in Fig. 3 the process is similar. At m=1, 100 images are recorded, each with a total integration time τ1. For m>1, the integration time is τm=τ1/mα to operate at constant damage. For each image, the total number of detected photons is integrated. The process is repeated without the sample and the transmission  and its variance are calculated. The error bars in Fig. 3 corresponds to the s.d. of the FOM as obtained from error propagation. For the theory curve, the asymmetry of the setup is again taken into account, which can result in kinks in the FOM between odd and even interaction numbers.
and its variance are calculated. The error bars in Fig. 3 corresponds to the s.d. of the FOM as obtained from error propagation. For the theory curve, the asymmetry of the setup is again taken into account, which can result in kinks in the FOM between odd and even interaction numbers.
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
Additional information
How to cite this article: Juffmann, T. et al. Multi-pass microscopy. Nat. Commun. 7:12858 doi: 10.1038/ncomms12858 (2016).
References
Giovannetti, V., Lloyd, S. & Maccone, L. Quantum-enhanced measurements: beating the standard quantum limit. Science 306, 1330–1336 (2004).
Israel, Y., Rosen, S. & Silberberg, Y. Supersensitive polarization microscopy using NOON states of light. Phys. Rev. Lett. 112, 103604 (2014).
Ono, T., Okamoto, R. & Takeuchi, S. An entanglement-enhanced microscope. Nat. Commun. 4, 2426 (2013).
Taylor, M. A. et al. Biological measurement beyond the quantum limit. Nat. Photonics 7, 229–233 (2013).
Brida, G., Genovese, M. & Ruo Berchera, I. Experimental realization of sub-shot-noise quantum imaging. Nat. Photonics 4, 227–230 (2010).
Giovannetti, V., Lloyd, S. & Maccone, L. Advances in quantum metrology. Nat. Photonics 5, 222–229 (2011).
Giovannetti, V., Lloyd, S. & Maccone, L. Quantum metrology. Phys. Rev. Lett. 96, 010401 (2006).
Luis, A. Phase-shift amplification for precision measurements without nonclassical states. Phys. Rev. A 65, 025802 (2002).
Higgins, B. L., Berry, D. W., Bartlett, S. D., Wiseman, H. M. & Pryde, G. J. Entanglement-free Heisenberg-limited phase estimation. Nature 450, 393–396 (2007).
Pégard, N. C. & Fleischer, J. W. in CLEO:2011-Laser Applications to Photonic Applications CThW6 Optical Society of America.
Arnaud, J. A. Degenerate optical cavities. Appl. Opt. 8, 189–196 (1969).
Kolobov, M. I. Quantum Imaging Springer: New York, USA, (2007).
Gigan, S., Lopez, L., Treps, N., Maître, A. & Fabre, C. Image transmission through a stable paraxial cavity. Phys. Rev. A 72, 023804 (2005).
Collaboration, L. S. et al. Observation of gravitational waves from a binary black hole merger. Phys. Rev. Lett. 116, 061102 (2016).
Mader, M., Reichel, J., Hänsch, T. W. & Hunger, D. A scanning cavity microscope. Nat. Commun. 6, 7249 (2015).
Frigault, M. M., Lacoste, J., Swift, J. L. & Brown, C. M. Live-cell microscopy—tips and tools. J. Cell Sci. 122, 753–767 (2009).
Piliarik, M. & Sandoghdar, V. Direct optical sensing of single unlabelled proteins and super-resolution imaging of their binding sites. Nat. Commun. 5, 4495 (2014).
Celebrano, M., Kukura, P., Renn, A. & Sandoghdar, V. Single-molecule imaging by optical absorption. Nat. Photonics 5, 95–98 (2011).
Andrews, M. R. et al. Direct, nondestructive observation of a bose condensate. Science 273, 84–87 (1996).
Egerton, R. F. Choice of operating voltage for a transmission electron microscope. Ultramicroscopy 145, 85–93 (2014).
Glaeser, R. M. How good can cryo-EM become? Nat. Methods 13, 28–32 (2016).
Kenney, J. F. in Mathematics of statistics ed. Keeping E. S. Van Nostrand: New York, USA, (1947).
Acknowledgements
We thank Gernot Neumayer for the preparation of the cell sample. P.H. thanks the Austrian Science Fund (FWF): J3680. This research is funded by the Gordon and Betty Moore Foundation, and by work supported under the Stanford Graduate Fellowship.
Author information
Authors and Affiliations
Contributions
T.J., B.K., P.H. and M.K. conceived the experiment. B.K. and T.J. carried out the experiment. B.K., T.J. and M.K. analysed the data and prepared the manuscript. T.F. analysed alignment constraints for the setup.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Juffmann, T., Klopfer, B., Frankort, T. et al. Multi-pass microscopy. Nat Commun 7, 12858 (2016). https://doi.org/10.1038/ncomms12858
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms12858
This article is cited by
-
Distributed quantum phase estimation with entangled photons
Nature Photonics (2021)
-
Electro-optic imaging enables efficient wide-field fluorescence lifetime microscopy
Nature Communications (2019)
-
Super sensitivity and super resolution with quantum teleportation
npj Quantum Information (2019)
-
Full-field interferometric imaging of propagating action potentials
Light: Science & Applications (2018)
-
Multi-pass transmission electron microscopy
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.