Abstract
Identification of positive regulators of T-cell immunity induced during autoimmune diseases is critical for developing novel therapies. The endoplasmic reticulum resident ubiquitin ligase Hrd1 has recently emerged as a critical regulator of dendritic cell antigen presentation, but its role in T-cell immunity is unknown. Here we show that genetic deletion of Hrd1 in mice inhibits T-cell proliferation, production of IL-2, and differentiation of Th1 and Th17 cells, and consequently protects mice from experimental autoimmune encephalomyelitis. Hrd1 facilitates T-cell proliferation by the destruction of cyclin-dependent kinase inhibitor p27kip1, and deletion of p27kip1 in Hrd1-null T-cells rescues proliferative capacity but not the production of cytokines, including IL-2, IFN-γ and IL-17. T-cell expression of Hrd1 is higher in patients with multiple sclerosis than in healthy individuals, and knockdown of Hrd1 in human CD4+ T cells inhibits activation and differentiation to Th1 and Th17 cells. Our study identifies Hrd1 as a previously unappreciated positive regulator of T cells and implies that Hrd1 is a potential therapeutic target for autoimmune diseases.
Similar content being viewed by others
Introduction
T-cell activation is initiated by the binding of antigenic peptides presented by the major histocompatibility complex (MHC) to the T-cell receptor (TCR)/CD3 complex, which results in T-cell proliferation and interleukin-2 (IL-2) production1,2. In addition to antigen-specific interaction with the TCR, full-scale T-cell activation requires a co-stimulatory signal provided by engagement of the T-cell co-receptor CD28 with its ligand, B7, on antigen-presenting cells2. Stimulation of TCR and CD28 drives T cells to proliferate by increasing the expression and activity of positive regulators and suppressing the expression of negative regulators through the activation of several transcription factors, including AP-1, NF-κB and NF-AT, and through epigenetic regulation2. For example, the expression of genes that promote cell cycle progression, including cyclins and cyclin-dependent kinases (CDKs), is quickly induced on TCR/CD28 stimulation, both in vitro and in vivo. By contrast, the expression of genes that inhibit cell cycle progression, including the CDK inhibitors and retinoblastoma protein, diminishes within a few hours of T-cell activation3,4. After clonal expansion, an efficient T-cell immune response relies on the differentiation of T cells into their functional subsets; CD4+ helper T (Th) cells differentiate into Th1, Th2, Th17 or regulatory T cells (Tregs). The differentiation programs resulting in these Th-cell subsets are influenced by a number of factors, including the dose and form of antigen and the antigen-presenting cells and/or co-stimulatory molecules; however, the dominant regulators of Th-cell differentiation are cytokines, often produced by innate immune cells such as dendritic cells. Commitment to each Th subset is regulated by a variety of mechanisms, including differential cytokine signalling, exclusive cytokine receptor expression, differential expression of transcription factors and/or differential chromatin remodelling of lineage-specific genes5. Although substantial progress has been made, the molecular mechanisms that govern T-cell activation and differentiation are incompletely understood.
Hrd1 is an E3 ubiquitin ligase that spans the endoplasmic reticulum (ER) membrane and was initially identified as a human homologue of yeast Hrd1p/Der3p (refs 6, 7). Hrd1 forms a complex with its stabilizing factor SEL1L, a human homologue of yeast Hrd3p (ref. 8). The best-characterized function of Hrd1 is to catalyse the degradation of misfolded/unfolded proteins, a process called ER-associated degradation (ERAD)9,10. MHC I heavy chain is an endogenous substrate for Hrd1, and Hrd1 suppression leads to the accumulation of glycosylated MHC I in the ER membrane11. ERAD-mediated MHC I degradation probably has an important role in the homeostatic regulation of MHC class I assembly and expression by dendritic cells11,12. The Hrd1 gene has been renamed SYVN1 (Synoviolin), owing to induced expression by synovial fibroblasts from patients with rheumatoid arthritis (RA), a disease in which Hrd1 suppresses synovial cell apoptosis13,14. We and others have demonstrated that pro-inflammatory cytokines, including IL-1β, IL-6, tumour necrosis factor-α (TNF-α) and IL-17, which have important pathogenic roles in synovitis development, induce Hrd1 expression in RA15,16,17. A body of evidence now indicates that Hrd1 also has a variety of important ERAD-independent physiological and pathological functions. p53 was the first identified non-ERAD substrate of Hrd1, and p53 ubiquitination and degradation negatively regulate Hrd1 expression and functions, including gene transcription, cell cycle regulation and apoptosis18. In addition to p53, the transcription factor Nrf2 is a substrate of Hrd1 in hepatocytes, with ubiquitination leading to attenuation of the Nrf2-mediated anti-oxidative stress response during liver cirrhosis19. Moreover, we have shown that Hrd1 programs dendritic cells for CD4+ T-cell activation during inflammation by directly targeting the zinc-finger transcription suppressor Blimp1 for ubiquitination and degradation. As Blimp1 suppresses the transcription of MHC class II, dendritic cell Hrd1 promotes CD4+ T-cell priming by inducing MCH II expression20.
In the current study, we conditionally delete the Hrd1 gene in developing thymocytes by crossing floxed Hrd1 and CD4-Cre mice. By analysing the phenotype of the resulting T-cell-specific Hrd1 conditional knockout (cKO) mice, we show that Hrd1 functions are required for T-cell homeostasis, activation and differentiation. Targeted Hrd1 gene deletion reduced T-cell numbers, inhibited T-cell clonal expansion and attenuated CD4+ T-cell differentiation to Th1, Th17 and Treg lineages. At the molecular level, we identify p27Kip1 as a target of the Hrd1 E3 ubiquitin ligase, as Hrd1 interacts with p27kip1 and promotes its degradation in T cells. Deletion of p27kip1 in Hrd1 cKO T cells rescues proliferation but not differentiation of T cells. Therefore, we identify Hrd1 as a positive regulator of T-cell immunity.
Results
Mice with T-cell-specific Hrd1 deletion are lymphocytopenic
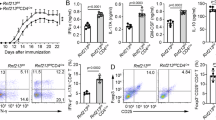
To study the role of Hrd1 in regulating the T-cell immune response, first we analysed Hrd1 expression in mouse CD4+ T cells. Hrd1 messenger RNA (mRNA) expression was relatively low in naive CD4 T cells compared with B cells (Supplementary Fig. 1a). Stimulation with anti-CD3/CD28 significantly (P<0.01) enhanced both Hrd1 mRNA and protein expression in CD4 T cells (Supplementary Fig. 1b,c), suggesting that Hrd1 expression is regulated in T cells by TCR/CD28 signalling. We then generated a strain of T-cell-specific Hrd1 knockout (Hrd1 cKO) mice by breeding mice with Hrd1 alleles (Hrd1fl/fl)20 with CD4-Cre transgenic mice (Supplementary Fig. 1d). Immunoblot analysis confirmed the complete elimination of Hrd1 protein expression in purified CD4+ T cells from the resulting Hrd1fl/flCD4-Cre mice (Hrd1 cKO mice; Supplementary Fig. 1e). By analysing cell surface CD4 and CD8 expression in the thymocytes of the Hrd1 cKO mice, we observed a slight, but statistically significant reduction in both CD4+CD8− and CD4−CD8+ thymocytes compared with Hrd1+/+CD4-Cre (wild type, WT) control mice (Supplementary Fig. 2a–c), implying that Hrd1 function is involved in both CD4-positive and CD8-positive T-cell development. Consistent with this notion, we also saw a marked reduction in the frequencies, as well as the absolute numbers, of either total CD3+ or CD4+ and CD8+ T cells in the spleens of Hrd1 cKO mice compared with WT mice (Supplementary Fig. 2g–h). Despite an increase in the frequency of γδ T cells in Hrd1 cKO mice, their absolute numbers were not altered (Fig. 1i). Therefore, the increased frequency of γδ T cells is likely a consequence of the reduction of αβ T cells in the Hrd1 cKO mice. In addition, the percentage of Tregs (CD25+FoxP3+) was not different, but the absolute cell numbers of Tregs decreased significantly, in cKO mice compared with WT controls (Supplementary Fig. 2f,j), which appears to be due to a lower total number of T cells in the spleens of cKO mice. Hrd1 conditional deletion in T cells therefore resulted in perturbed development of thymocytes and led to lymphocytopenia in both the thymus and spleen.
(a–e) Naive CD4 T cells were purified from the spleens of WT and Hrd1 cKO mice, and stained with CFSE. Cells were then cultivated in the presence of anti-CD3 plus anti-CD28 antibodies (1 mg ml−1 each). Cell proliferation (a,b) and apoptosis (c,d) at each indicated time point were analysed by flow cytometry. The representative data (a,c) and the averages (b,d) from five independent experiments are shown. (e) The absolute numbers of dividing cells from five independent experiments are indicated. (f) CFSE-stained cells were adoptively transferred into B6/SJL congenic mice bearing a CD45.1 marker. Ten days after the transfer, receipt mice were killed and the CD45.2+ CD4 T cells were gated for their CFSE dilution. Representative images (left panels) and the averages (right panels) from three pairs of mice were shown. (g–m) Naive CD4 T cells from WT and Hrd1 cKO mice were cultivated with anti-CD3 plus anti-CD28 (g–i) or at each polarization condition (j–m). IL-2 production and Th-cell differentiation were analysed by intracellular staining as described in the materials and methods (g,h). IL-2 in the culture supernatants 3 days after anti-CD3 plus anti-CD28 stimulation was analysed by enzyme-linked immunosorbent assay (ELISA). (i) The representative images (g,j,l) and the mean±s.d. (h,i,k,m) from five independent experiments are shown. The Mann–Whitney test was used for the statistical analysis. NS, no significant difference; *P<0.05, **P<0.01 and ***P<0.001.
Hrd1 is required for T-cell activation and differentiation
As TCR/CD28 signalling induced Hrd1 protein and mRNA expression in T cells (Supplementary Fig. 1b,c), we hypothesized that upregulation of Hrd1 in activated T cells might play an important role in T-cell function. Indeed, Hrd1 cKO T cells showed markedly lower proliferation on in vitro stimulation with anti-CD3/anti-CD28 antibodies compared with WT T cells because the carboxyfluorescein succinimidyl ester (CFSE) dilution was slowed down in Hrd1-null T cells (Fig. 1a,b), and the numbers of proliferative cells were reduced (Supplementary Fig. 3a). Because Hrd1 has been identified as an anti-apoptotic factor that prevents cells from ER stress-induced apoptosis, we further determined whether Hrd1 deletion affects CD4 T-cell survival during in vitro activation. The percentages of annexin V-positive cells were indistinguishable between WT and Hrd1 cKO T cells (Fig. 1c,d), suggesting that the observed reduction in T-cell growth was not due to the increased apoptosis. By contrast, the cell numbers of proliferative Hrd1-null T cells were significantly reduced, confirming that Hrd1 deficiency impairs T-cell growth upon in vitro TCR/CD28 stimulation (Fig. 1e). The in vivo analysis by adoptive transfer of CFSE-stained Hrd1 cKO CD4+ T cells into congenic recipient mice confirmed the reduction in Hrd1 cKO T-cell homeostatic proliferation compared with WT T cells (Fig. 1f; Supplementary Fig. 3b). A similar proliferative defect was further confirmed in Hrd1-null CD8 T cells as the CFSE fluorescence intensity of Hrd1-null T cells were less diluted (Supplementary Fig. 3c,d). Next, we assessed the production of IL-2 in Hrd1 cKO CD4 T cells and WT littermate controls after stimulation with anti-CD3 or anti-CD3 plus anti-CD28 antibodies. We observed that Hrd1 cKO T cells failed to secrete IL-2 as measured by both intracellular staining (Fig. 1g,h) and enzyme-linked immunosorbent assay (Fig. 1i), indicating that Hrd1 function is required for both T-cell clonal expansion and IL-2 production. As IL-2 is a known T-cell growth factor, one possible explanation for the reduced proliferation of Hrd1 cKO T cells is impaired IL-2 production; however, addition of exogenous IL-2 failed to rescue Hrd1 cKO CD4 T-cell proliferation (Fig. 1a, bottom panel, and Fig. 1b). Therefore, reduced IL-2 production appears not to be responsible for the impaired proliferation of Hrd1 cKO T cells.
Hrd1 gene deletion impairs T-cell activation is unlikely due to the altered expression levels of cell surface receptors, because the expression of TCR complex including TCRβ and CD3ɛ and the co-stimulatory receptor CD28 are indistinguishable between WT and Hrd1-null T cells from the spleen and draining lymph nodes (Supplementary Fig. 4a–c). Similarly, the cell surface expression levels of the early activation markers CD25 and CD69 were not altered by Hrd1 gene deletion in naive CD4 T cells and even after 24-h stimulation (Supplementary Fig. 5a–d). Antigen recognition by TCR in the presence of co-stimulation through CD28 triggers the phosphorylation of upstream signalling molecules during early phase of T-cell activation. Loss of Hrd1 functions appears not to impair the overall tyrosine phosphorylation patterns in CD4 T cells on either TCR stimulation alone or TCR plus CD28 stimuli (Supplementary Fig. 6a). Moreover, the activation of the mitogen-activated protein kinases, including Erk1/2 and JNK1, was unaltered by Hrd1 deficiency during T-cell activation (Supplementary Fig. 6b). Therefore, Hrd1 is unlikely to promote T-cell activation through activating the upstream TCR/CD28 signalling pathway.
After clonal expansion, and depending on the cytokine environment, CD4+ T cells differentiate into a variety of effector subsets, including Th1, Th2, Th17 and induced Tregs5. To determine whether Hrd1 affects CD4 T-cell differentiation in vitro, we cultured CD4+CD25− naive T cells from Hrd1 cKO and WT mice under different cytokine environments to direct them into different Th subsets, as reported previously21. Although Th2 differentiation was normal (Fig. 1j,k), Hrd1 cKO Th0 and Th1 cells exhibited lower production of IFN-γ compared with cells differentiated from WT controls. Meanwhile, Hrd1 cKO Th17 cells produced less IL-17 (Fig. 1j,k). While the FoxP3-positive Treg percentage at lower transforming growth factor-β (TGF-β) concentration was significantly reduced, Hrd1 deficiency did not affect Treg differentiation at a higher TGF-β concentration (Fig. 1l,m). Taken together, these data indicate that Hrd1 is a positive regulator of T-cell proliferation, activation and Th-cell differentiation in vitro. To further determine whether the defect in Th1 and Th17 cytokine production by Hrd1-null CD4 T cells is due to the impaired proliferation, we analysed the cell cycle-based production of IFN-γ and IL-17 by Hrd1 cKO CD4 T cells. As shown in Fig. 7, the overall IL-2, IFN-γ and IL-17 production by Hrd1-null CD4 T cells, as well as their cell cycle division, were reduced, confirming our initial observation that Hrd1 functions are required for the optimal T-cell proliferation and cytokine production. Importantly, Hrd1 cKO T cells produced a markedly less IFN-γ and IL-17 comparing with those of WT CD4 T cells even at the same cycle of cell division (Supplementary Fig. 7), indicating that the reduced cytokine production by Hrd1 cKO T cells is uncoupled with the cell cycle progression.
WT and Hrd1 cKO mice were immunized with 50 μg KLH-NP in CFA on day 1 followed by a boosted immunization with 50 μg KLH-NP in IFA on day 8. Mice were killed on day 14. (a–c) Total splenocytes were stained with CFSE and cultivated with 100 ng ml−1 of KLH-NP for 5 days. The proliferation (a,b) and apoptosis (c) of CD4+ T cells were analysed. The representative images (a,c) and data from five pairs of mice (b) are shown. (d–g) The production of IL-2 in CD4 T cells (d), and TNF-α, IFN-γ and FoxP3+ Tregs in CD4 (e) and CD8 (f) cells were analysed. The representative data (d–f) and the mean±s.d. (g) from five pairs of mice are shown. (h) The levels of NP-specific antibodies in the sera of immunized Mann–Whitney test mice were analysed by enzyme-linked immunosorbent assay (ELISA). Error bars represent data from five pairs of mice (mean±s.d.). The Mann–Whitney test was used for the statistical analysis. NS, no significant difference; *P<0.05, **P<0.01 and ***P<0.001.
WT and Hrd1 cKO CD4 T cells were stimulated with anti-CD3 plus anti-CD28 (2 μg ml−1) for 24 h. (a,b) Cell cycle was analysed by DNA staining and flow cytometry. Representative images (a) and data from five independent experiments (b) are shown. (c–e) p27kip1 protein (c,d) and mRNA (e) levels were analysed by immunoblotting and real-time PCR with reverse transcription. Representative images (c) and data from five independent experiments (d) are shown. (f) HEK293 cells were transfected with Myc-tagged Hrd1 and Flag-p27. The interaction of Hrd1 with p27kip1 in the transiently transfected cells was determined by co-immunoprecipitation with anti-Flag and the bound Hrd1 was detected by immunoblotting with anti-Myc antibody (top panel). The same membrane was re-blotted with anti-Flag antibody (middle panel) and the expression levels of Hrd1 in the whole-cell lysates were confirmed by immunoblotting (bottom panel). (g) CD4 T cells from WT and p27kip1 KO mice were stimulated with anti-CD3 plus anti-CD28 for 24 h. The interaction of p27kip1 with Hrd1 was analysed by immunoprecipitation with anti-p27 and immunoblotting with anti-Hrd1 antibody (top panel). The expression levels of p27 (middle panel) and Hrd1 (bottom panel) in whole-cell lysate was confirmed by immunoblotting. (h) CD4 T cells were stimulated with anti-CD3 or anti-CD3 plus anti-CD28 for 30 min, and Hrd1 interaction with p27kip1 was determined as in g. Error bars (b,d,e) represent data from five pairs of mice (mean+s.d.). Mann–Whitney test was used for the statistical analysis. NS, no significant difference; *P<0.05, **P<0.01 and ***P<0.001.
(a,b) Flag-p27 and HA-Ub expression plasmids were co-transfected with Myc-Hrd1 or its CA mutant. p27kip1 protein ubiquitination was determined by immunoprecipitation of p27kip1 protein and immunoblotting with anti-HA antibodies (top panels). The same membranes were re-probed with anti-Flag (middle panels) and Hrd1 expression in the whole-cell lysates was confirmed by immunoblotting with anti-Myc antibody (bottom panels). (c) The interaction of p27 with both WT and Hrd1/CA mutant in transiently transfected 293 cells were analysed. (d,e) WT and Hrd1 cKO CD4 T cells were cultured with anti-CD3 plus anti-CD28 overnight and then chased by cycloheximide (CHX) for the indicated times. The protein expression levels of p27, Hrd1, SKP2 and the loading control β-actin were examined by immunoblotting using each specific antibody. Representative images (d) and quantifications of five independent experiments (e) are shown. (f–h) The protein expression levels of p27kip1, Hrd1, p53 and β-actin in WT and Hrd1 cKO CD4 T cells were determined by immunoblotting (f), and the average levels from five pairs of mice are indicated (g). Total RNA was extracted from stimulated cells and the expression levels of p21 and PUMA were analysed by real-time PCR with reverse transcription (h). Mann–Whitney test was used for the statistical analysis. NS, no significant difference; *P<0.05, **P<0.01 and ***P<0.001.
(a,b) Thymus (a) and spleen (b) were isolated from 8–10-week-old WT, Hrd1 cKO, p27kip1 KO and double KO (dKO) mice. Representative images (top panels) and the weights in milligrams (mg; bottom panels) are shown. (c,d) Single-cell suspensions from the thymus and spleen were analysed by cell surface staining of CD4 and CD8. Representative flow data (c) and the absolute numbers of each indicated population from five sets of mice (d) are shown. (e–h) Naive CD4 T cells from WT, Hrd1 cKO, p27kip1 KO and dKO mice were stained with CFSE and cultured with anti-CD3 plus anti-CD28. Cell proliferation (e,f) and IL-2 production (g,h) were determined. Representative images (e,g) and data from five independent experiments are shown (f,h). (i) Naive CD4 T cells from WT, Hrd1 cKO, p27kip1 KO and dKO mice under Th1, Th17 or Treg polarization conditions. The production of each indicated cytokines or FoxP3 expression was analysed by intracellular staining. Representative images are shown here and the results from five independent experiments are indicated in Supplementary Fig. 8. Mann–Whitney test was used for the statistical analysis. NS, no significant difference; *P<0.05, **P<0.01 and ***P<0.001.
(a,b) EAE was induced in 8–10-week-old WT and Hrd1 cKO mice, and the symptoms were scored daily. The average symptom scores (a) and disease incidence (b) from 15 pairs of mice are shown. (c–j) The spinal cords were isolated 12–15 days after EAE induction. Representative images of H&E staining are shown; scale bars, 200 μm (top); 10 μm (bottom; c). The splenocytes (d) and infiltrated lymphocytes were isolated and analysed by flow cytometry for each of the indicated cell surface markers (d,e) and apoptosis markers (g). The cytokine production was determined by intracellular staining (h,j). Error bars represent data from five independent isolation (each purification was from spinal cords pooled from three mice; f,h,j). (i) Purified naive CD4 T cells from WT and Hrd1 cKO mice were adoptively transferred into T-cell-null mice. Two days after transfer, EAE induction was performed and the mice were scored for disease development. Error bars represent data from five pairs of recipient mice. Mann–Whitney test was used for the statistical analysis. *P<0.05, **P<0.01 and ***P<0.001. The P value is <0.05 in (a,k) at each scored time point unless specifically indicated.
(a) The levels of Hrd1 expression in freshly isolated CD4 T cells from healthy donors (N=12) or MS patients (N=16) by real-time PCR. (b–d) CD4 T cells freshly isolated CD4 T cells from healthy donors or MS patients (N=5) were lysed and subjected to western blotting analysis for the protein levels of Hrd1 and p27kip1 using GAPDH as a loading control (b), and the relative band intensities were quantified (c). The mRNA levels of p27kip1 in samples of (b) were determined by real-time PCR using β-actin as a control (d). (e–k) Purified CD4 T cells from healthy donors were infected with lentivirus carrying control or Hrd1-specific shRNA. (e,f), green fluorescent protein (GFP)-positive cells were sorted, the expression levels of Hrd1, p27kip1 and GAPDH were examined by western blotting (e), and p27kip1 ubiquitination was determined as in Fig. 4a (f). (g,h) Cells were stained with far-red cell trace and cultivated for 3 days with TCR/CD28 stimulation. GFP-positive cells were gated and the proliferation was analysed for CFSE dilution. (h,i) Cells were cultivated under Th1, Th17 and Treg polarization conditions and the gated GFP+ cells were analysed. Representative images (g,i,j) and the percentages from three independent experiments (h,k) are shown. Mann–Whitney test was used for the statistical analysis in a,c,d,h and k.
Hrd1 deficiency impairs antigen-specific T-cell immunity
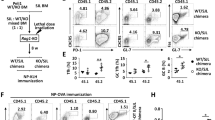
To determine the effect of Hrd1 deficiency on T-cell activation in vivo, we immunized 8–10-week-old Hrd1 cKO mice and their WT littermate controls with 4-hydroxy-3-nitrophenyl acetyl-keyhole limpet haemocyanin (NP-KLH) in complete Freund’s adjuvant (CFA) on day 1 and a boosted immunization of NP-KLP in incomplete Freund’s adjuvant (IFA) on day 8. Immunized mice were killed on day 14 and the NP-specific T-cell immune response was analysed. Consistent with our in vitro observations, there was a significant decrease in the proliferation of Hrd1 cKO T cells on cultivation with NP antigen (Fig. 2a,b). This reduction in NP-specific T-cell growth was not due to an increase in activation-induced cell death, because WT and Hrd1 cKO CD4 T cells derived from splenocytes showed similar amounts of apoptosis (Fig. 2c). Furthermore, IL-2, TNF-α and IFN-γ produced by CD4+ T cells and TNF-α and IFN-γ produced by CD8+ T cells were markedly decreased in Hrd1 cKO mice compared with controls (Fig. 2d–g), suggesting that Hrd1 functions as a regulator required for antigen-specific T-cell activation in vivo. In addition, similar to those observed in mice under naive conditions, the percentage of CD25+FoxP3+ Tregs was not altered in the spleens of immunized mice (Fig. 2e,g)
To determine the effects of Hrd1 on the T-cell-dependent humoral immune response, we measured NP-specific antibody levels in sera from mice 7 days after the first immunization with NP-KLH/CFA (the primary immune response) and again after the boosted immunization with NP-KLP/IFA. As shown in Fig. 2h, Hrd1 cKO mice exhibited lower antigen-specific antibodies of IgG1, IgG2a, IgG2b and IgG3 in the primary and secondary responses compared with WT controls. In contrast, the levels of NP-specific IgM were not altered. These results indicate that Hrd1 is required for the T-cell-dependent humoral response.
Hrd1 interacts with p27kip1 in T cells
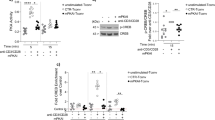
Our finding that Hrd1 deficiency impaired T-cell proliferation without increasing activation-induced cell death in vitro and in mice implies an important role for Hrd1 in regulating T-cell cycle entry during clonal expansion. To test this hypothesis, we analysed the cell cycle progression of Hrd1 cKO and WT T cells on TCR/CD28 stimulation. As indicated in Fig. 3a,b, Hrd1 deficiency resulted in a partial cell cycle arrest at the G1–S transition because of a significant increase in G1/G0 phase cells and a reduction in cells at S and G2/M phases. p27kip1 has been shown to negatively regulate the transition from G1 to S phase by inhibiting the activity of CDK2 (refs 22, 23, 24). As a really interesting new gene (RING) finger-containing E3 ubiquitin ligase, Hrd1 often downregulates the expression levels of proteins through ubiquitination-mediated degradation18,19,20. We speculated that Hrd1 may regulate CD4 T-cell proliferation by ubiquitinating p27kip1 for subsequent destruction. We found that p27kip1 protein expression (Fig. 3c,d), but not mRNA level (Fig. 3e), was significantly increased in Hrd1 cKO T cells, implying that Hrd1 promotes cell cycle progression through p27kip1 protein destruction. To determine whether Hrd1 is an E3 ligase of p27kip1, we first asked whether Hrd1 physically interacts with p27kip1. Co-immunoprecipitation and immunoblot analysis detected a physical interaction between Hrd1 and p27kip1 proteins; Hrd1 protein was detected in the immunoprecipitates from HEK293 cells transiently transfected with both Hrd1 and p27kip1 expression plasmids, but not from those transfected with Hrd1 alone (Fig. 3f). The interaction between endogenous Hrd1 and p27kip1 in mouse primary CD4 T cells was confirmed by detection of the Hrd1 protein in the anti-p27kip1 immunoprecipitates from WT but not p27kip1-null T cells, clearly demonstrating that p27kip1 is an interacting protein of Hrd1 in T cells (Fig. 3g). The interaction of Hrd1 with p27kip1 occurred at low levels in naive T cells, and stimulation with anti-CD3 for 30 min resulted in a significant increase in their interaction, which was further enhanced by addition of anti-CD28 (Fig. 3h). These results indicate that Hrd1/p27kip1 interaction is regulated by TCR/CD28 signalling.
Hrd1 is an E3 ubiquitin ligase of p27kip1 in T cells
We next examined whether Hrd1 catalyses ubiquitination of p27kip1. Anti-HA antibody detected bands with gradually increasing molecular weights in anti-p27kip1 immunoprecipitates from cells transiently transfected with p27kip1 and HA-ubiquitin expression plasmids, indicating that p27kip1 is ubiquitinated. Hrd1 co-expression led to a substantial increase in p27kip1 ubiquitination (Fig. 4a). The E3 ligase activity of Hrd1 is required for p27kip1 ubiquitination, because co-expression of Hrd1 containing an alanine point mutation at the critical cysteine within the RING E3 ligase region (Hrd1/CA)25 failed to enhance p27kip1 ubiquitination (Fig. 4b). Interaction between p27kip1 with Hrd1 and the Hrd1/CA mutant was comparable (Fig. 4c). Together, these results indicate that Hrd1 is an E3 ubiquitin ligase that targets p27kip1.
Our observation that p27kip1 protein but not its mRNA levels were elevated in Hrd1 cKO T cells suggests that Hrd1-mediated p27kip1 ubiquitination is likely to promote p27kip1 protein destruction. To test this notion, we measured the half-life of p27kip1 in CD4 T cells chased with the translational inhibitor cycloheximide (CHX). p27kip1 was gradually degraded in Hrd1+/+ CD4 T cells following CHX treatment (Fig. 4d), with a half-life of ∼1.5 h (Fig. 4e). In contrast, the protein expression levels of p27kip1 were significantly higher in Hrd1 cKO CD4 T cells following CHX, with a half-life extended to beyond 4 h (Fig. 4e), indicating that Hrd1 E3 ligase is responsible for p27kip1 protein degradation in T cells. The S-phase kinase-associated protein 2 (SKP2), an F-box protein, has been well characterized as an E3 ubiquitin ligase targeting p27kip1 for ubiquitination and degradation26,27,28. It is possible that Hrd1 controls p27kip1 protein stability through SKP2 in T cells, with decreased expression of SKP2 in Hrd1 cKO CD4 T cells leading to p27kip1 accumulation. However, SKP2 protein levels in WT and Hrd1 cKO T cells were indistinguishable (Fig. 4d), excluding the possibility that p27kip1 protein accumulation in Hrd1-null T cells is due to the reduced SKP2 expression. In addition, p53 has been identified as a substrate of Hrd1, and it is possible that loss of Hrd1 results in T-cell hypo-proliferation through p53 protein accumulation18. To test this possibility, we examined the expression level of p53 protein in WT and Hrd1 cKO mice. Neither the expression level of p53 protein nor the transcription levels of its target genes, p21 and PUMA29,30, were altered by Hrd1 gene deletion in T cells (Fig. 4f–h). Collectively, our study identifies Hrd1 as a novel E3 ubiquitin ligase of p27kip1 in T cells.
Hrd1 promotes T-cell activation through p27kip1
p27kip1 plays a critical role in regulating T-cell clonal expansion, differentiation and memory development, and its upregulation is known as an important molecular mechanism in T-cell peripheral tolerance induction4,22,31,32. On the basis of our discoveries that Hrd1 is an E3 ubiquitin ligase of p27kip1 and loss of Hrd1 leads to p27kip1 protein accumulation in T cells, we speculated that Hrd1 may regulate T-cell immune response through p27kip1 degradation. We predicted that further deletion of p27kip1 in Hrd1 cKO T cells would rescue T-cell activation and differentiation from the Hrd1 deficiency defect. Therefore, we crossed Hrd1−/−CD4-cre mice with p27kip1−/− KO mice to create Hrd1−/−p27kip1−/−CD4-cre double-knockout (DKO) mice. Consistent with the previous observations, both the thymus and spleen sizes were enlarged in p27kip1 KO mice. Notably, in contrast to a slight reduction in the thymus and spleen sizes in Hrd1 cKO mice, the DKO animals exhibited a phenotype identical to that of p27kip1 KO mice, with the enlarged thymus and spleens (Fig. 5a,b), and increased total cell numbers, frequencies and absolute cell numbers of CD4 T cells in both the spleen and thymus (Fig. 5c,b). Moreover, while DKO mice showed a modest but statistically significant decrease in CD8 absolute cell numbers in the spleen compared with p27kip1 KO mice (Fig. 5d), indicating that p27kip1 deletion resulted in a partial rescue of the reduction in CD8 T cells. Importantly, DKO T cells exhibited the same proliferation rate as p27kip1 KO T cells on TCR/CD28 stimulation (Fig. 5e,f) as the numbers of proliferative p27kip1 KO and p27kip1Hrd1 DKO cells are comparable (Supplementary Fig. 8a), clearly indicating that Hrd1 promotes T-cell growth in a p27kip1-dependent manner. Collectively, our observations indicate that Hrd1 regulates T-cell development and proliferation through catalysing p27kip1 protein degradation.
Intracellular staining analysis detected comparable levels of IL-2 production in Hrd1 cKO and Hrd1/p27kip1 DKO T cells on in vitro TCR/CD28 stimulation (Fig. 5g,h), indicating that Hrd1 promotes IL-2 expression in a p27kip1-independent manner. Similarly, the levels of both IFN-γ and IL-17 production under Th1 and Th17 polarization conditions, as well as the percentages of FoxP3-positive Tregs, were indistinguishable between Hrd1 cKO and Hrd1/p27kip1 DKO T cells (Fig. 5i; Supplementary Fig. 8b). Consistent with previous studies31,32, we confirmed a marked reduction in IL-2 production and Th1, Th17 and Treg differentiation of p27kip1 KO CD4 T cells (Fig. 5g–i; Supplementary Fig. 8). Together, these data indicate that Hrd1-mediated p27kip1 protein degradation, while necessary for T-cell development and differentiation, is dispensable for IL-2 production and differentiation into Th1, Th17 and Tregs.
T-cell-specific Hrd1 deletion protects mice from EAE
Our findings that Hrd1 is a positive regulator of the T-cell activation and differentiation prompted us to test its therapeutic potential in autoimmune disease. We induced an experimental form of autoimmune encephalomyelitis (EAE), a model of human multiple sclerosis, in WT and Hrd1 cKO mice, and assessed the effect of genetic Hrd1 suppression in T cells, as reported previously33. When immunized with myelin oligodendrocyte glycoprotein peptide (MOG35–55), WT C57/B6 mice developed clinical symptoms of EAE starting on day 7 after immunization that peaked around day 14, followed by a partial recovery phase. In contrast, disease onset in Hrd1 cKO mice was delayed for at least 2 days, with a statistically dramatic reduction in symptoms (Fig. 6a). In addition, 100% of WT mice developed EAE disease by day 10 after MOG immunization, while only 80% of Hrd1 cKO mice developed disease and 20% of mice were fully protected from MOG-induced autoimmune disease (Fig. 6b). Histological analysis of the central nervous system (CNS) tissues isolated from MOG-immunized mice on day 14 showed fewer infiltrated lymphocytes into the spinal cord in Hrd1 cKO mice (Fig. 6c), indicating that genetic Hrd1 deletion suppresses the MOG-induced autoimmune response in mice.
We then purified the infiltrated lymphocytes in brain and spinal cord tissues isolated from MOG-immunized mice during peak EAE symptoms. As expected, Hrd1 cKO mice showed a significant decrease in the percentage and absolute number of CD4+ and CD8+ T cells in the spleen (Fig. 6d) and CNS (Fig. 6e,f). Again, the reduction in infiltrated T cells during disease development is not likely to be due to an elevation in apoptosis, because the percentages of annexin V-positive CD4+ T cells isolated from CNS tissues of Hrd1 cKO mice and their WT control littermates were comparable (Fig. 6g,h). The Hrd1 cKO mice exhibited a significant decrease in the proportion of TNF-α+, IFN-γ+ and IL-17+CD4+ cells, but no difference in the percentage of CD4+FoxP3+ cells compared with WT control mice (Fig. 6i,j). Consistent with the results observed in naive mice, a significant reduction in both CD4 and CD8 T cells in the spleens of Hrd1 cKO mice were detected after MOG immunization (Fig. 6d), raising the possibility that the decreased number of T cells in Hrd1 cKO mice is responsible for protecting mice from the disease. To test this, we purified CD4 T cells from WT and Hrd1 cKO mice, and adoptively transferred an equal number of them to T-cell-null mice. One day after transfer, EAE induction was performed. As shown in Fig. 6k, Hrd1 cKO T-cell recipient mice showed resistance to EAE as indicated by modest symptoms and delayed onset of disease, suggesting that Hrd1 deletion in T cells protected the mice from MOG-induced EAE. Taken together, these results support the notion that Hrd1 is a potential target for autoimmune disease treatment.
Hrd1 potentially promotes human autoimmunity
The fact that Hrd1 expression is upregulated during T-cell activation and that Hrd1 enhances autoimmune response in mice promoted us to test whether Hrd1 expression is associated with human autoimmune diseases. Indeed, analysis the expression levels of Hrd1 detected a significant increase in Hrd1 mRNA expression levels CD4 T cells from the multiple sclerosis (MS) patients than that of healthy controls (Fig. 7a), suggesting a pathogenic role of Hrd1 elevation in human MS. We then further confirmed the marked increase in Hrd1 protein expression levels in CD4 T cells from MS patients (Fig. 7b,c). Notably, in contrast to the elevated Hrd1 protein expression levels, p27kip1 protein, but not its mRNA expression levels are decreased in CD4 T cells from MS patients comparing with healthy controls (Fig. 7b–d), suggesting that Hrd1 regulates p27kip1 protein stability in human CD4 T cells and that Hrd1 promotes CD4 T-cell autoimmunity through p27kip1 protein destruction. To support this conclusion, we used a short hairpin RNA (shRNA) and knocked down Hrd1 expression in human CD4 T cells, and demonstrated that Hrd1 knockdown resulted in a significant increase in p27kip1 protein expression (Fig. 7e). In addition, knockdown of Hrd1 expression largely inhibited p27kip1 ubiquitination in human CD4 T cells (Fig. 7f). These results confirm that Hrd1 is an E3 ubiquitin ligase in human T cells. We then examined the effect of Hrd1 suppression on the activation and differentiation of human CD4 T cells. As expected, Hrd1 knockdown in human CD4 T cells inhibited their proliferation, IL-2 production, Th1 and Th17 differentiation (Fig. 7g–k; Supplementary Fig. 8c). Moreover, similar to that in mouse CD4 T cells, Hrd1 suppression slightly impaired Treg polarization at low but not high concentrations of TGF-β (Fig. 7j,k). Taken together, these studies demonstrate that Hrd1 is a positive regulator of human CD4 T-cell activation and differentiation, and that the elevated Hrd1 expression is involved in human autoimmune disease.
In summary, our study here reveals Hrd1 as a previously unappreciated positive regulator of T-cell immunity (Fig. 8). At the molecular level, Hrd1 functions as an E3 ubiquitin ligase of the CDK inhibitor p27kip1 and promotes the cell cycle progression through p27kip1 destruction during T-cell clonal expansion. However, this Hrd1-mediated p27kip1 degradation is unlikely responsible for Hrd1-mediated IL-2 production nor for Th1 and Th17 differentiation, because further deletion of p27kip1 in Hrd1-null T cells rescued T-cell growth, but not IL-2, IFN-γ and IL-17 production.
Discussion
Our study shows that the ER membrane-spanning E3 ubiquitin ligase Hrd1 is a previously unidentified positive regulator of T cells. This conclusion is supported by the following observations: (i) targeted deletion of the Hrd1 gene in developing thymocytes resulted in a dramatic reduction in the mature T-cell population in the thymus and spleens; (ii) Hrd1 functions were required for the proliferation and differentiation of CD4 T cells into Th1, Th17 and Tregs both in vitro and in mice during antigen-specific immune response; (iii) loss of Hrd1 functions led to cell cycle arrest and the accumulation of p27kip1 protein, an inhibitor of CDK, in T cells, and Hrd1 acted an E3 ubiquitin ligase of p27kip1; (iv) further genetic deletion of p27kip1 in Hrd1-null T cells rescued T-cell proliferation but not CD4 T-cell differentiation; (v) genetic Hrd1 suppression in T cells protected mice from MOG-induced experimental autoimmune EAE; and (vi) Hrd1 is a positive regulator of human CD4 T-cell activation and differentiation, and that the elevated Hrd1 expression are involved in human autoimmune disease multiple sclerosis.
It has been well established that downregulation of p27kip1 is essential for T-cell clonal expansion, the failure of which leads to T-cell anergy31,32,34,35,36. In the current study, we discovered that Hrd1-mediated ubiquitination and degradation of p27kip1 is a previously unappreciated molecular mechanism regulating cell cycle progression during the clonal expansion of T cells, as loss of Hrd1 functions led to cell cycle arrest at G1/G0 and p27kip1 protein accumulation. Importantly, Hrd1 interacted with p27kip1 in T cells and catalysed p27kip1 ubiquitination through its E3 ligase activity. In Hrd1-null T cells, p27kip1 protein accumulation does not appear to be related to the activity of SKP2, a well-characterized E3 ubiquitin ligase of p27kip1 (refs 26, 27, 28), because protein expression levels of SKP2 were not altered in Hrd1 cKO T cells. In addition to p27kip1, Hrd1 has been shown to catalyse ubiquitination and degradation of p53, a transcription factor involved in regulating gene transcription, cell cycle regulation and apoptosis18, raising the possibility that Hrd1 may positively regulate T-cell immunity through p53 degradation. However, the level of p53 protein and the transcription levels of its target genes, p21 and PUMA, were not affected by Hrd1 gene deletion in T cells. Together with our findings that Hrd1 suppression did not lead to an increase in apoptosis upon in vitro TCR/CD28 stimulation or in mice immunized with specific antigens, p53 is unlikely to be a Hrd1 target, and p53 degradation is likely not involved in Hrd1-mediated promotion of T-cell proliferation. Therefore, our study identifies Hrd1 as an E3 ligase of p27kip1 and establishes that Hrd1-mediated p27kip1 degradation plays an important role in T-cell immunity.
In addition to its functions in positively regulating T-cell growth, Hrd1 appears to play important roles in CD4 T-cell differentiation, since the targeted deletion of Hrd1 in T cells impaired in vitro Th1, Th17 and Treg differentiation. In support of this notion, we confirmed a reduction in Th1 and Th17 cells in Hrd1 cKO mice immunized with specific antigens or during EAE development. Despite comparable percentages of FoxP3-positive Treg populations in WT and Hrd1 cKO mice, there was a significant reduction in the absolute T-cell numbers in Hrd1 cKO mice, both under the naive condition and during inflammation. However, Hrd1-mediated p27Kip1 ubiquitination is unlikely to be the only molecular mechanism underlying Hrd1 function in CD4 differentiation because further deletion of p27kip1 in Hrd1 cKO T cells failed to rescue the production of IL-2 and differentiation of Th1, Th17 and Treg. We recently discovered that Hrd1 is an E3 ubiquitin ligase of Blimp1, a transcriptional suppressor known to inhibit IL-2 production and Th1/Th17 differentiation37,38,39, raising the possibility that Hrd1 enhances IL-2 production and the differentiation of Th1 and Th17 by CD4 T cells20. Therefore, Hrd1-mediated Blimp1 ubiquitination and degradation to increase IL-2 production could be another possible molecular mechanism by which Hrd1 modulates CD4 T-cell differentiation. It is well established that one E3 ubiquitin ligase often targets multiple substrates to achieve its biological functions40, it will not be a surprise that additional substrates of Hrd1 exist in T cells to regulate the different aspects of T-cell functions such as cytokine production. Future studies are needed to illuminate whether Hrd1 functions as a positive regulator of T-cell differentiation through Blimp1 and if not, to identify the additional substrates/pathways that are regulated by Hrd1.
Hrd1 has been identified as an anti-apoptotic factor that protects cells from ER stress-induced apoptosis41,42, raising the possibility that loss of Hrd1 might cause T-cell death. In our study, the percentages of annexin V-positive apoptotic cells were indistinguishable between WT and Hrd1 cKO T cells on in vitro TCR/CD28 stimulation or in mice during autoimmune disease development. We recently reported that Hrd1 can promote degradation of IRE1α14, suggesting a possible role of Hrd1-mediated IRE1 in regulating T-cell activation and differentiation. Indeed, we detected a significant increase in the expression levels of IRE1 protein but not mRNA in Hrd1 cKO T cells, further confirming our previous finding that Hrd1 is an E3 ubiquitin ligase of IRE1. However, neither genetic nor pharmacological IRE1α inhibition had an effect on CD4 T-cell proliferation and differentiation into Th1, Th17 and Tregs21, excluding the possibility that Hrd1-mediated ubiquitination contributes to these processes. In addition to Hrd1, ER stress sensors and chaperons have been shown to be involved in regulation a variety of immune functions43,44,45.
Our study indicates that Hrd1 is a potential therapeutic target for autoimmune disease treatment because genetic suppression of Hrd1 in T cells protected mice from MOG-induced EAE. The reduction in T-cell numbers in the peripheral lymphoid organs of Hrd1 cKO mice might have contributed to disease protection; however, the impaired T-cell activation and differentiation into Th1 and Th17, without reducing the percentage of Tregs, is likely to be responsible for the protective effect in Hrd1 cKO mice. Recipient mice that received an equal number of transfer CD4 T cells showed fewer symptoms of EAE after MOG immunization. In addition, we previously showed that Hrd1 programs dendritic cells to prime CD4 T cells by facilitating MHC II transcription, and that suppression of dendritic cell Hrd1 functions is also protective against EAE development20. Importantly, our data show that Hrd1 upregulation appears to be associated with human autoimmunity and that suppression of Hrd1 expression in human T cells inhibited their activation and differentiation. Since TCR/CD28 stimulation induces Hrd1 expression, the possibility that this elevated Hrd1 expression in T cells is a consequence of an increase in auto-reactive T-cell activation during MS cannot be fully excluded and further studies are needed. Nevertheless, we demonstrated that this elevated Hrd1 expression is associated with the reduced p27Kip1 protein but not mRNA levels. Together with our observations that Hrd1 knockdown resulted in the elevated p27Kip1 expression and reduced p27Kip1 ubiquitination, our studies demonstrate that Hrd1-mediated p27Kip1 protein degradation as a novel molecular mechanism in human T-cell proliferation. In addition, Hrd1 suppression by shRNA-mediated knockdown inhibited human T-cell proliferation, IL-2 production, and Th1 and Th17 differentiation. Therefore, targeting Hrd1 may be beneficial for the treatment of autoimmune diseases, in particular those mediated by Th1 and Th17 cells, such as multiple sclerosis and RA. Notably, a specific small-molecule inhibitor, LS-102, has been identified recently that suppresses the E3 ubiquitin ligase catalytic activity of Hrd1 (ref. 46). Administration of LS-102 significantly reduced the symptoms of arthritis in mice immunized with collagen by inducing the apoptosis of synovial fibroblasts during synovitis46. It will be interesting to further investigate whether LS-102, in addition to its effects on inducing synovial fibroblast apoptosis, suppresses T-cell activation and CD4 T-cell differentiation into Th1 and Th17 in mice during collagen-induced arthritis as well as MOG-induced EAE.
Methods
Mice
The Hrd1 floxed mice were used as reported20 and the T-cell-specific Hrd1-null mice (Hrd1 cKO) were generated by breeding Hrd1 floxed mice with CD4-Cre transgenic mice (Jackson laboratory, Bar Harbor, ME, catalogue no. 017336). Both p27kip1 KO (catalogue no. 002781) and T-cell-null mice (catalogue no. 002118) were purchased from Jackson Laboratory. All the mice used in this study are in the C57/B6 genetic background and were maintained/used at the Northwestern University mouse facility under pathogen-free conditions according to institutional guidelines and using animal study proposals approved by the Institutional Animal Care and Use Committees at Northwestern University.
Flow cytometry and cytokine production analysis
Single-cell suspensions of thymocytes and splenocytes were used for staining using specific antibodies against CD3 (catalogue no. 100204), CD4 (catalogue no. 100408), CD8 (catalogue no. 100733), CD25 (catalogue no. 102215), CD44 (catalogue no.103011) and CD62L (catalogue no. 104412), all of which were purchased from Biolegend (San Diego, CA), for 30 min on ice. The stained cells were washed with ice-cold PBS, fixed and analysed by flow cytometry as reported33. For intracellular staining, cells were fixed and permeabilized using reagents from Biolegend and stained with fluorescent-labelled antibodies against FoxP3 or each specific cytokine.
In vitro T-cell activation and polarization
Naive CD4 T cells were purified using the anti-CD4 antibody-coated magnetic beads (Life Technology, Grand Island, NY). For proliferation, cells were stained with 5 μM of CFSE at room temperature for 15 min followed by adding fetal calf serum (to a final of 2%) to terminate the reaction. CFSE-stained T cells were cultivated with soluble anti-CD3 (eBiosience, 16-0031-86) plus anti-CD28 (eBiosience 16-0281-86; 5 μg ml−1) for 3 days and the cell division was analysed by flow cytometry. For T-cell polarization, CD4+ T cells were cultivated under each polarization conditions for 5 days. The polarized T cells were analysed by intracellular staining of IFN-γ (Th1; catalogue no. 506908), IL-4 (Th2; catalogue no. 504105), IL-17 (Th17; catalogue no. 506916) and FoxP3 (Treg; catalogue no. 602107), which were purchased from Biolegend, as described33.
Antigen-specific T-cell immune response in mice
Eight- to 12-week-old WT and Hrd1 cKO mice were immunized with 50 μg NP-KLH in 100 μl of CFA on day 1 followed by a boosted immunization with NP-KLH/IFA on day 8. Sera were collected on day 7 and 14 for the detection of NP-specific antibody production by enzyme-linked immunosorbent assay, as described20. Immunized mice were killed on day 14 and the total splenocytes were isolated. Antigen-specific T-cell proliferation was analysed by CFSE (Life Technologies, Boston, MA, catalogue no. C34554) staining, and the production of cytokines by CD4 T cells was determined by intracellular staining and flow cytometry analysis.
Intracellular cytokine staining
In vitro stimulated T-cells under either non-polarization or each indicated polarization condition, or cells from the draining spleens of immunized mice were stimulated with phorbol myristate acetate (10 ng ml−1; Sigma-Aldrich, P1585), ionomycin (1 μg ml−1; Sigma-Aldrich, I0634) and monensin (10 μg ml−1; Affymetrix Inc., catalogue no 00-4505-51) for 4 h, and the intracellular staining was performed using an eBioscience kit (San Diego, CA) following a protocol provided by the manufactures.
Real-time quantitative PCR with reverse transcription
WT and Hrd1 cKO CD4 T cells were activated with or without anti-CD3 plus anti-CD28 for 4 h. Total RNA was extracted with Trizol reagent according to the manufacturer’s instructions (Thermo Fisher Scientific, catalogue no. 15596018). Complementary DNA was synthesized using the qScript cDNA Synthesis kit (Quanta Biosciences, Gaithersburg, MD, catalogue no. 95047-100). iQ5 and SYBRGreen Detection system (Bio-Rad, Hercules, CA) were used for quantitative PCR as described47,48. Data were normalized to the expression of β-actin in each sample. Primers used to detect the expression of Hrd1, p27kip1 (Cell Signalling, catalogue no. 3688S), p53 (Abcam, catalogue no. ab61241) and Skp2 (Cell Signalling, catalogue no. 4313S) transcripts are shown in Supplementary Table 1.
Co-immunoprecipitation and western blotting
Cells were lysed in cold radio immunoprecipitation assay buffer (Life Technologies). The cell lysate was pre-cleaned with protein-G sepharose (GE Healthcare, catalogue no. 17-0618-02) for 30 min and subjected to immunoprecipitation with each indicated antibodies (1 μg), incubated for 1 h on ice and then 25 μl of protein-G sepharose beads were added for additional 4–6 h. The beads were then washed for four times, boiled with 15 ml of 2 × SDS sample buffer for 5 min and the proteins were separated on 10% SDS–PAGE gels and transferred to polyvinylidene difluoride membranes. The membranes were blocked in 5% fat-free dried milk in Tris-buffered saline with 0.5% Tween 20 (TBST) for 2 h. The membranes were then incubated in appropriate primary antibodies overnight at 4 °C. Membranes were washed in TBST and then incubated in horseradish peroxidase (HRP)-conjugated secondary antibodies (EMD Millipore Corp, goat anti-rabbit IgG antibody, HRP conjugate, catalogue no. 12–348; goat anti-mouse IgG antibody, HRP conjugate, catalogue no. 12–349). Membranes were washed in TBST, and the signals were visualized using enhanced chemiluminescence substrate (Thermo Scientific, Waltham, MA) and quantified using the Bio-Rad Image software. Representative uncropped blots are shown in Supplementary Fig. 9.
EAE induction and disease characterization
Eight- to 10-week-old WT and Hrd1 cKO mice in the C57/BL6 genetic background were immunized with MOG35–55 peptide (emulsified with CFA (200 μg per mouse)). Mice were also given pertussis toxin (200 ng per mouse) on days 0 and 2 via tail vein injection. All mice were weighed and examined daily for symptoms and assigned scores on a scale of 0–5 as follows: 0, no overt signs of disease; 1, limp tail; 2, limp tail and partial hindlimb paralysis; 3, complete hindlimb paralysis; 4, complete hindlimb and partial forelimb paralysis; and 5, moribund state or death. For the adoptive transfer, 5 × 106 WT or Hrd1 cKO CD4 T cells were adoptively transferred into T-cell-null (TCRβ/δ DKO mice by intravenous injection. Two days after the transfer, EAE induction was performed and scored.
Isolation and characterization of human CD4 T cells
About 10 ml fresh blood was collected from each MS patient or healthy donor and centrifuged. After lysis of the red blood cells, total white blood cells were subjected to CD4 T-cell isolation using a negative selection isolation kit (Miltenyi Biotech, San Diego, CA) following the instruction. Purified CD4 T cells were utilized for the following experiments: for RNA purification and real-time PCR analysis, isolated cells were suspended in Trizol reagent and total RNA was isolated according to the manufacturer’s instructions (Invitrogen). For the shRNA knockdown experiment, CD4 T cells were infected with lentivirurs carrying Hrd1-specific or control shRNA. Green fluorescent protein-positive cells were either gated or sorted for analysis by intracellular staining or western blotting. The study has been approved by the Institutional Review Board.
Statistic analysis
All data are indicated as mean±s.d. The unpaired, nonparametric Student’s t-test (Mann–Whitney test) was used for the statistic analysis. P<0.05 was considered as the significant difference.
Data availability
The data that support the findings of this study are available from the corresponding author on request.
Additional information
How to cite this article: Xu, Y. et al. The ER membrane-anchored ubiquitin ligase Hrd1 is a positive regulator of T-cell immunity. Nat. Commun. 7:12073 doi: 10.1038/ncomms12073 (2016).
References
Rossjohn, J. et al. T cell antigen receptor recognition of antigen-presenting molecules. Ann. Rev. Immunol. 33, 169–200 (2014).
Smith-Garvin, J. E., Koretzky, G. A. & Jordan, M. S. T-cell activation. Ann. Rev. Immunol. 27, 591–619 (2009).
Furukawa, Y., Piwnica-Worms, H., Ernst, T. J., Kanakura, Y. & Griffin, J. D. cdc2 gene expression at the G1 to S transition in human T lymphocytes. Science 250, 805–808 (1990).
Wells, A. D. & Morawski, P. A. New roles for cyclin-dependent kinases in T cell biology: linking cell division and differentiation. Nat. Rev. Immunol. 14, 261–270 (2014).
Zhu, J., Yamane, H. & Paul, W. E. Differentiation of effector CD4 T cell populations (*). Ann. Rev. Immunol. 28, 445–489 (2010).
Kaneko, M., Ishiguro, M., Niinuma, Y., Uesugi, M. & Nomura, Y. Human HRD1 protects against ER stress-induced apoptosis through ER-associated degradation. FEBS Lett. 532, 147–152 (2002).
Bordallo, J., Plemper, R. K., Finger, A. & Wolf, D. H. Der3p/Hrd1p is required for endoplasmic reticulum-associated degradation of misfolded lumenal and integral membrane proteins. Mol. Biol. Cell 9, 209–222 (1998).
Neuber, O., Jarosch, E., Volkwein, C., Walter, J. & Sommer, T. Ubx2 links the Cdc48 complex to ER-associated protein degradation. Nat. Cell Biol. 7, 993–998 (2005).
Gardner, R. G. et al. Endoplasmic reticulum degradation requires lumen to cytosol signalling. Transmembrane control of Hrd1p by Hrd3p. J. Cell Biol. 151, 69–82 (2000).
Friedlander, R., Jarosch, E., Urban, J., Volkwein, C. & Sommer, T. A regulatory link between ER-associated protein degradation and the unfolded-protein response. Nat. Cell Biol. 2, 379–384 (2000).
Burr, M. L. et al. MHC class I molecules are preferentially ubiquitinated on endoplasmic reticulum luminal residues during HRD1 ubiquitin E3 ligase-mediated dislocation. Proc. Natl Acad. Sci. USA 110, 14290–14295 (2013).
Arteaga, M. F., Wang, L., Ravid, T., Hochstrasser, M. & Canessa, C. M. An amphipathic helix targets serum and glucocorticoid-induced kinase 1 to the endoplasmic reticulum-associated ubiquitin-conjugation machinery. Proc. Natl Acad. Sci. USA 103, 11178–11183 (2006).
Amano, T. et al. Synoviolin/Hrd1, an E3 ubiquitin ligase, as a novel pathogenic factor for arthropathy. Genes Dev. 17, 2436–2449 (2003).
Gao, B. et al. Synoviolin promotes IRE1 ubiquitination and degradation in synovial fibroblasts from mice with collagen-induced arthritis. EMBO Rep. 9, 480–485 (2008).
Gao, B., Calhoun, K. & Fang, D. The proinflammatory cytokines IL-1beta and TNF-alpha induce the expression of Synoviolin, an E3 ubiquitin ligase, in mouse synovial fibroblasts via the Erk1/2-ETS1 pathway. Arthritis Res. Ther. 8, R172 (2006).
Corvaisier, M. et al. IL-26 is overexpressed in rheumatoid arthritis and induces proinflammatory cytokine production and Th17 cell generation. PLoS Biol. 10, e1001395 (2012).
Toh, M. L. et al. Role of interleukin 17 in arthritis chronicity through survival of synoviocytes via regulation of synoviolin expression. PLoS ONE 5, e13416 (2010).
Yamasaki, S. et al. Cytoplasmic destruction of p53 by the endoplasmic reticulum-resident ubiquitin ligase ‘Synoviolin’. EMBO J. 26, 113–122 (2007).
Wu, T. et al. Hrd1 suppresses Nrf2-mediated cellular protection during liver cirrhosis. Genes Dev. 28, 708–722 (2014).
Yang, H. et al. Hrd1-mediated BLIMP-1 ubiquitination promotes dendritic cell MHCII expression for CD4 T cell priming during inflammation. J. Exp. Med. 211, 2467–2479 (2014).
Kemp, K. L. et al. The serine-threonine kinase inositol-requiring enzyme 1alpha (IRE1alpha) promotes IL-4 production in T helper cells. J. Biol. Chem. 288, 33272–33282 (2013).
Nourse, J. et al. Interleukin-2-mediated elimination of the p27Kip1 cyclin-dependent kinase inhibitor prevented by rapamycin. Nature 372, 570–573 (1994).
Polyak, K. et al. Cloning of p27Kip1, a cyclin-dependent kinase inhibitor and a potential mediator of extracellular antimitogenic signals. Cell 78, 59–66 (1994).
Toyoshima, H. & Hunter, T. p27, a novel inhibitor of G1 cyclin-Cdk protein kinase activity, is related to p21. Cell 78, 67–74 (1994).
Qiu, Q. et al. Toll-like receptor-mediated IRE1alpha activation as a therapeutic target for inflammatory arthritis. EMBO J. 32, 2477–2490 (2013).
Yu, Z. K., Gervais, J. L. & Zhang, H. Human CUL-1 associates with the SKP1/SKP2 complex and regulates p21(CIP1/WAF1) and cyclin D proteins. Proc. Natl Acad. Sci. USA 95, 11324–11329 (1998).
Carrano, A. C., Eytan, E., Hershko, A. & Pagano, M. SKP2 is required for ubiquitin-mediated degradation of the CDK inhibitor p27. Nat. Cell Biol. 1, 193–199 (1999).
Tsvetkov, L. M., Yeh, K. H., Lee, S. J., Sun, H. & Zhang, H. p27(Kip1) ubiquitination and degradation is regulated by the SCF(Skp2) complex through phosphorylated Thr187 in p27. Curr. Biol. 9, 661–664 (1999).
Kaeser, M. D. & Iggo, R. D. Chromatin immunoprecipitation analysis fails to support the latency model for regulation of p53 DNA binding activity in vivo. Proc. Natl Acad. Sci. USA 99, 95–100 (2002).
Seoane, J., Le, H. V. & Massague, J. Myc suppression of the p21(Cip1) Cdk inhibitor influences the outcome of the p53 response to DNA damage. Nature 419, 729–734 (2002).
Wells, A. D., Walsh, M. C., Sankaran, D. & Turka, L. A. T cell effector function and anergy avoidance are quantitatively linked to cell division. J. Immunol. 165, 2432–2443 (2000).
Rowell, E. A., Wang, L., Hancock, W. W. & Wells, A. D. The cyclin-dependent kinase inhibitor p27kip1 is required for transplantation tolerance induced by costimulatory blockade. J. Immunol. 177, 5169–5176 (2006).
Zhang, J. et al. The type III histone deacetylase Sirt1 is essential for maintenance of T cell tolerance in mice. J. Clin. Invest. 119, 3048–3058 (2009).
Verdoodt, B. et al. The cyclin-dependent kinase inhibitors p27Kip1 and p21Cip1 are not essential in T cell anergy. Eur. J. Immunol. 33, 3154–3163 (2003).
Boussiotis, V. A. et al. p27kip1 functions as an anergy factor inhibiting interleukin 2 transcription and clonal expansion of alloreactive human and mouse helper T lymphocytes. Nat. Med. 6, 290–297 (2000).
Powell, J. D., Lerner, C. G. & Schwartz, R. H. Inhibition of cell cycle progression by rapamycin induces T cell clonal anergy even in the presence of costimulation. J. Immunol. 162, 2775–2784 (1999).
Choi, Y. S. et al. ICOS receptor instructs T follicular helper cell versus effector cell differentiation via induction of the transcriptional repressor Bcl6. Immunity 34, 932–946 (2011).
Nutt, S. L., Fairfax, K. A. & Kallies, A. BLIMP1 guides the fate of effector B and T cells. Nat. Rev. Immunol. 7, 923–927 (2007).
Bandyopadhyay, S., Valdor, R. & Macian, F. Tle4 regulates epigenetic silencing of gamma interferon expression during effector T helper cell tolerance. Mol. Cell. Biol. 34, 233–245 (2014).
Liu, Y. C., Penninger, J. & Karin, M. Immunity by ubiquitylation: a reversible process of modification. Nat. Rev. Immunol. 5, 941–952 (2005).
Carvalho, P., Goder, V. & Rapoport, T. A. Distinct ubiquitin-ligase complexes define convergent pathways for the degradation of ER proteins. Cell 126, 361–373 (2006).
Denic, V., Quan, E. M. & Weissman, J. S. A luminal surveillance complex that selects misfolded glycoproteins for ER-associated degradation. Cell 126, 349–359 (2006).
Hur, K. Y. et al. IRE1alpha activation protects mice against acetaminophen-induced hepatotoxicity. J. Exp. Med. 209, 307–318 (2012).
Staron, M. et al. Heat-shock protein gp96/grp94 is an essential chaperone for the platelet glycoprotein Ib-IX-V complex. Blood 117, 7136–7144 (2011).
Liu, B. et al. Folding of Toll-like receptors by the HSP90 paralogue gp96 requires a substrate-specific cochaperone. Nat. Commun. 1, 79 (2010).
Yagishita, N. et al. RING-finger type E3 ubiquitin ligase inhibitors as novel candidates for the treatment of rheumatoid arthritis. Int. J. Mol. Med. 30, 1281–1286 (2012).
Lee, S. M., Gao, B., Dahl, M., Calhoun, K. & Fang, D. Decreased FoxP3 gene expression in the nasal secretions from patients with allergic rhinitis. Otolaryngol. Head Neck Surg. 140, 197–201 (2009).
Lee, S. M., Gao, B. & Fang, D. FoxP3 maintains Treg unresponsiveness by selectively inhibiting the promoter DNA-binding activity of AP-1. Blood 111, 3599–3606 (2008).
Acknowledgements
We thank Fang lab members for critical reading of the manuscript and constructive suggestions during our research. This work was supported by National Institutes of Health R01 grants (AI079056, AI108634 and AR006634) to D.F. and by National Natural Science Grants and Shanghai Committee of Science and Technology Grant (no. 14DZ2260300) to H.L. (nos 81271054 and 81470673) to H.L.
Author information
Authors and Affiliations
Contributions
Y.X., F.Z., Q.Q., J.W., K.C., Q.K., B.G. and J.M.-C. performed the experiments and analysed the data. B.Z., Jin.Z., Jia.Z., J.S., Y.F. and D.D.Z. contributed critical reagents and in experimental design. Jin. Z., Y.X., H.L. and D.F. designed the study, analysed the data and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-9 and Supplementary Table 1. (PDF 5480 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Xu, Y., Zhao, F., Qiu, Q. et al. The ER membrane-anchored ubiquitin ligase Hrd1 is a positive regulator of T-cell immunity. Nat Commun 7, 12073 (2016). https://doi.org/10.1038/ncomms12073
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms12073
This article is cited by
-
Overexpression of Synoviolin and miR-125a-5p, miR-19b-3p in peripheral blood of rheumatoid arthritis patients after treatment with conventional DMARDs and methylprednisolone
Clinical Rheumatology (2024)
-
A potential immunotherapy target for breast cancer: parenchymal and immune-stromal expression of the NLRP3 inflammasome pathway
BMC Cancer (2023)
-
SYT7 regulates the progression of chronic lymphocytic leukemia through interacting and regulating KNTC1
Biomarker Research (2023)
-
SEL1L–HRD1 endoplasmic reticulum-associated degradation controls STING-mediated innate immunity by limiting the size of the activable STING pool
Nature Cell Biology (2023)
-
E3 ubiquitin ligase SYVN1 is a key positive regulator for GSDMD-mediated pyroptosis
Cell Death & Disease (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.