Abstract
Promoter DNA methylation is a key epigenetic mechanism for stable gene silencing, but is correlated with expression when located in gene bodies. Maintenance and de novo DNA methylation by catalytically active DNA methyltransferases (DNMT1 and DNMT3A/B) require accessory proteins such as UHRF1 and DNMT3L. DNMT3B isoforms are widely expressed, although some do not have active catalytic domains and their expression can be altered during cell development and tumourigenesis, questioning their biological roles. Here, we show that DNMT3B isoforms stimulate gene body methylation and re-methylation after methylation-inhibitor treatment. This occurs independently of the isoforms’ catalytic activity, demonstrating a similar functional role to the accessory protein DNMT3L, which is only expressed in undifferentiated cells and recruits DNMT3A to initiate DNA methylation. This unexpected role for DNMT3B suggests that it might substitute for the absent accessory protein DNMT3L to recruit DNMT3A in somatic cells.
Similar content being viewed by others
Introduction
DNA methylation is a key epigenetic mechanism that participates in stable gene silencing in key biological processes, including the establishment and maintenance of tissue specific gene-expression patterns, X-chromosome inactivation, parasitic transposable elements silencing and genomic imprinting1,2,3,4. Although DNA methylation is critical for mammalian development, genome-wide studies show that aberrations of normal tissue DNA-methylation patterns are a hallmark of cancer and other diseases5,6.
Cytosines in a CpG context are methylated by the transfer of a methyl group from S-adenosylmethionine catalysed by DNA methyltransferases (DNMTs)7. DNMTs in mammals are comprised of four family members: DNMT1, DNMT3A, DNMT3B as well as DNMT3L (DNMT 3-like), which is required for the establishment of DNA-methylation patterns during development8. Maintenance of DNA methylation is carried out by DNMT1, which copies DNA-methylation patterns from the parental to the daughter strand during replication9. Both DNMT3A and DNMT3B serve as de novo methyltransferases during embryonic development but they also help maintain the DNA-methylation patterns in somatic cells in concert with DNMT1, since the latter cannot perform this function alone10,11,12. DNMT3A has 2 different isoforms, while DNMT3B has more than 30 isoforms13,14,15,16. Expression patterns of the latter are highly conserved between humans and mice, suggesting that these isoforms are biologically relevant17. Many studies have identified the specific roles of DNMTs in DNA methylation during development but the role of aberrant expression levels of DNMTs and isoforms, especially DNMT3B in cancer, leading to global DNA-methylation changes, is still unclear11,13,14,15,16,18,19,20,21,22.
Several studies have investigated the roles of DNMTs in more detail, demonstrating that DNMT3B participates in gene body methylation or re-methylation by targeting the H3K36me3 modification4,23,24,25,26,27,28. Disruption of the catalytic domains of all three DNMTs was recently characterized in human ES cells by CRISPR/Cas9 genome editing24. However, the disruption of DNMT3B1 left a truncated version very similar to the DNMT3B3 isoform in these cells24. Furthermore, this study also found that although DNMT3B1 is highly expressed in ES cells, DNMT3B1 expression decreases and DNMT3B3, which has a disrupted catalytic domain, becomes the dominant isoform expressed in somatic cells24. Thus the role of DNMT3B isoforms in ES and somatic cells is not well-characterized.
In this study, we sought to identify target sites of DNMT3B isoforms on a genome-wide level and their functional roles by characterizing a representative panel of DNMT3B isoforms and DNMT3L by restoring their expression in DNMT3B-deficient cells.
We confirm that transcribed regions of genes are the favoured de novo DNA-methylation target. We also show that isoforms of DNMT3B can influence DNA methylation in cells with decreased methylation and re-methylation in gene bodies after DNA-methylation-inhibitor treatment. Altogether, our results suggest that DNMT3B isoforms can act as accessory proteins that interact with catalytically active enzymes to re-establish DNA methylation and could be one of many key factors for initiation of de novo DNA methylation during tumourigenesis.
Results
Stable Reintroduction of DNMT Isoforms
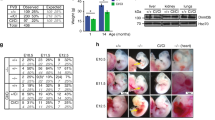
We elucidated the role of DNMT3B and its isoforms in DNA methylation by using the 3BKO and DKO8 derivatives of the HCT116 colon cancer cell line that have homozygous deletions for DNMT3B. DKO8 cells additionally have a markedly reduced protein level of a hypomorphic DNMT1 (DNMT1ΔE2-5) (refs 29, 30) allele. There is globally about 3% less DNA methylation in 3BKO cells and 22% less DNA methylation in DKO8 cells compared with the parental line HCT116 (Supplementary Fig. 1)29. We selected a representative panel of DNMT3B isoforms, as well as DNMT3L, which is catalytically inactive and expressed in ES cells but not in somatic cells8,31, to use as a positive accessory protein control (Fig. 1a). For DNMT3B isoforms, we chose DNMT3B1: the canonical full-length protein, DNMT3B3: a catalytically inactive isoform32 expressed in somatic and ES cells but overexpressed in some types of cancer33, DNMT3B4: a catalytically inactive isoform that interacts with DNMT3s and reduces DNA-binding ability32, DNMTΔ3B2: a N-terminal-truncated isoform mainly overexpressed in lung cancer15, DNMTΔ3B4: a N-terminal-truncated isoform missing the PWWP domain15 and DNMT3L: expressed only during embryonic development34. In addition, we engineered DNMT3B1-M and DNMTΔ3B2-M, which are catalytically inactive mutants containing a cysteine to serine inactivating point mutation at position 651 and 452, respectively35. After infection we obtained polyclonal cell populations expressing a specific DNMT3B isoform or DNMT3L in DKO8 and 3BKO cells.
(a) Schematic diagram of DNMT3A (3A), DNMT3B isoforms (3B1, 3B1-M, 3B3, 3B4, Δ3B2, Δ3B2-M and Δ3B4) and DNMT3L (3L) showing conserved PWWP (purple), PHD-like domain (green) and DNMT catalytic motifs (black). There are five catalytic domains (I, IV, VI, IX and X) in DNMT3A and DNMT3B, all of which are absent in DNMT3L. A red-coloured VI domain indicates inactivating mutations (Cys to Ser) of amino acids 651 and 452 in 3B1-M and Δ3B2-M, respectively. 3B4 has a frameshift with a unique protein sequence shown in orange. ^indicates alternative splicing. (b,c) mRNA expression level of endogenous DNMT3B in HCT116 cells, exogenous DNMT3B isoforms and DNMT3L assessed by qRT-PCR and normalized to the expression of the TATA Box Binding Protein (TBP) in DKO8 cells, 56 days post transfection and 3BKO cells, 14 days post transfection, respectively. Error bars indicate standard deviation from the mean of three biological replicates. The empty vector (EV) cell line is the transfection control. (d,e) Protein-expression levels of exogenous MYC-tagged DNMT isoforms by western blot analysis in DKO8 cells, 56 days post transfection and 3BKO cells, 14 days post transfection, respectively. β-ACTIN was used as a loading control.
Different RNA-expression levels were seen for each of the DNMTs but larger variations were apparent at the protein levels, possibly reflecting differential stabilities of the variable isoforms outside of the putative DNMT3A/3B complex (Fig. 1b–e). Endogenous DNMT3A and exogenous DNMT3B or DNMT3L RNA-expression levels were measured by qRT-PCR, following puromycin selection in both cell lines and before and after 5-Aza-CdR treatment in 3BKO cells (Supplementary Fig. 2). Consistent with our previous study35, we show the mutant form of DNMT3B1 was expressed at a higher protein level than its wild-type form; however, no significant differences in restoration of DNA methylation between the transfected constructs could be observed (Fig. 2b). We also know that excess DNMT3B protein is degraded when not bound to nucleosomes, which may explain the differences between the RNA and protein levels of these DNMTs35.
(a) Heatmap showing 54,911 CpG sites in DKO8 cells, expressing the indicated DNMT isoforms. CpGs were selected because they were targeted by at least one construct. CpG sites in genomic locations targeted by DNMT3Bs and 3L with respect to promoter (maroon), gene body (orange) and other regions, excluding promoters and gene bodies (grey), are shown in the left panel. CpG sites in CpG islands are represented in green in the left panel. Endogenous methylation levels in HCT116 and DKO8 cells are represented by a cold to warm colour scale (β-value 0–1, 0–100% methylated), where every row represents one CpG. DNA-methylation levels of DKO8 cells expressing a DNMT3B isoforms or DNMT3L are shown in the right panel. (b) Boxplots showing the distribution of DNA-methylation levels of 54,911 CpG sites for each indicated cell line. HCT116 cells’ methylation level is included for comparison with the derivative cell line DKO8 EV. A dashed red line indicates the median of DKO8 EV, outliers outside the 5th to 95th percentiles are not shown. (c) Distribution of genomic locations targeted by individual DNMT3Bs and 3L with respect to promoter (maroon), gene body (orange) and other regions excluding promoters and gene bodies (grey), from the 450 K array. (d) Distribution of genomic locations targeted by DNMT3Bs and 33LL in respect to CpG island (green) or non-CpG island (grey) from the 450 K array. Background distribution is the representative population of CpG sites that could be targeted by the individual DNMTs. Statistical enrichment was performed using a z-test comparing the targeted CpG sites for each DNMT isoform with the overall distribution of probes on the 450 K array, ***P<0.001.
DNA-methylation restoration by DNMT3B isoforms
Global DNA-methylation levels in DKO8 cells expressing DNMT3B isoforms or DNMT3L were measured 56 days post transfection. The results are compared with the parental HCT116 cell line and represented as boxplots (Supplementary Fig. 3a). CpG sites that showed an increase of 0.2 in β-value, compared with the control EV with any construct, were retained for further analysis and the resulting methylation values are shown as heatmaps, boxplots (Fig. 2a,b) or δ-methylation values (Supplementary Fig. 3b). As expected, DNMT3L could restore DNA methylation in the transfected cells and showed the strongest overall restoration of DNA methylation compared with the DNMT3B isoforms (Fig. 2a). Interestingly, the majority of sites showing increased DNA methylation were also methylated in the parental HCT116 cells (Fig. 2a). Most of these restored sites are located in gene bodies, which were found to be enriched in H3K36me3 in HCT116 cells, a characteristic of an actively transcribed region23. We previously showed that the H3K36me3 mark is not lost in the more severely demethylated derivative cell line DKO1, which has lost 90% of DNA methylation23. This finding is consistent with other observations that DNMT3B recognizes the H3K36me3 modification written by SETD2 (refs 25, 26). Individually, the sites targeted by each DNMT3B isoform which showed a 0.2 increase in β-value compared with the control EV are located in gene bodies and non-CpG islands regardless of the isoform when compared with the background distribution of CpG sites on the array (Fig. 2a,c,d) except for DNMT3B4 and DNMTΔ3B4. Biological replicates for 3 of the 9 DNMT constructs were performed, demonstrating that the CpG site-specific recruitment was consistent globally (Supplementary Fig. 4) and for the specific target sites (Supplementary Figs 5–7).
Furthermore, DNMT3B1 and its mutated form DNMT3B1-M show similar restoration of DNA methylation regardless of the latter being catalytically inactive, consistent with previous findings (Fig. 2a,b)35. In addition, although DNMT3B3 has been considered as a catalytically inactive isoform32, DNA methylation was strongly increased by its presence. The catalytically active DNMTΔ3B2 and inactive Δ3B2-M isoforms both lack the N-terminal domain and have similar decreased abilities to methylate DNA (Fig. 2a,b). DNMTΔ3B4 that has no PWWP domain had the lowest ability to induce re-methylation. DNMT3B4 was also weak at inducing re-methylation, confirming the finding of its decreased binding affinity with DNA32.
The abilities of DNMT3B and its isoforms to restore methylation are mainly dependent on the presence of an N-terminal but not a catalytic domain in most of the isoforms (Fig. 2a,b). The exception here is DNMT3B4, which loses the ability to restore DNA methylation and has previously been shown to cause hypomethylation by binding to active DNMT3s and reducing their DNA-binding affinities20,32. The loss of DNA or nucleosome-binding affinity of DNMT3B4 could be due to the specific truncated C-terminal structure of DNMT3B4, which should be further studied. However, our previous studies have demonstrated that the ability of DNMT3B1 to bind nucleosomes is important in maintaining DNA-methylation levels, while loss of the N-terminal domain could markedly decrease the binding ability and increase protein degradation35,36. Unexpectedly, most DNMT3B isoforms can restore DNA methylation at similar target regions independently of the presence of functional catalytic domains. These findings suggest that besides its functional role as a methyltransferase, DNMT3B may also act as an accessory protein to recruit DNMT3A and stimulate DNA methylation.
Re-methylation rate increased by DNMT3B isoforms
We recently showed that inhibiting gene body methylation by transient 5-Aza-CdR treatment decreases the expression of some genes, and that restoration of this methylation requires DNMT3B (ref. 23). We demonstrated that there are four groups (I–IV) of demethylated targets, based on the rate of re-methylation in HCT116 cells after treatment with 5-Aza-CdR (Supplementary Fig. 8a)23. The CpG sites of Group I were enriched in gene bodies having the H3K36me3 modifications and showed the fastest rates of re-methylation and were almost fully remethylated 42 days after treatment. The CpG sites in Group IV, however, showed the slowest rate of re-methylation and remained demethylated at day 42 (ref. 23).
To test whether DNMT3B isoforms have different functional roles during re-methylation, we stably introduced a subset of isoforms into the 3BKO cell line (Fig. 1a,c,e) and subsequently treated them with 5-Aza-CdR for 24 h. We observed DNA methylation decreases 5 days post treatment and DNA-methylation restoration 42 days post treatment, consistent with pervious studies (Fig. 3 and Supplementary Fig. 8)23. RNA-expression levels of endogenous DNMT3A and the exogenous DNMT3B or DNMT3L were analysed before and after 5-Aza-CdR treatment (Supplementary Fig. 2). Interestingly, DNMT3B4 and DNMT3L showed decreased RNA-expression levels after 5-Aza-CdR treatment, although DNMT3L was still able to stimulate the DNA-methylation levels after 42 days (Fig. 3 and Supplementary Fig. 2b). DNMT3L, DNMT3B1, 3B1-M and 3B3 were able to stimulate DNA re-methylation most strongly at the H3K36me3-enriched Group I CpGs (Fig. 3)23. DNMT3B4 did not influence the rebound methylation of this group as we expected, since it has decreased binding affinity to DNA32. As we predicted, Group IV CpG sites were also confirmed to be non-DNMT3B target regions, the same as for DNMT3L (Supplementary Fig. 8).
Heatmaps and Boxplots showing previously (Yang et al.23) defined H3K36me3-enriched and fast-rebounding Group I CpG sites in cell lines expressing different DNMTs before 24 h 5-Aza-CdR treatment and at Day 5 and Day 42 post treatment for 3BKO (a) and 3ABDKO (b) cells. Individual CpG sites falling in genomic locations targeted by DNMT3Bs and 3L with respect to promoter (maroon), gene body (orange) and other regions, excluding promoters and gene bodies (grey) are shown in the left panel. CpG sites in CpG islands are represented in green in the left panel. Endogenous methylation levels in cell lines are represented by a cold to warm colour scale (β-value 0–1, 0–100% methylation) where every row represents one CpG site in the three right panels.
To confirm that DNMT3A is actually the enzyme responsible for the DNA-methylation rebound, we used another HCT116 derivative cell line, 3ABDKO, which has homozygous deletions of DNMT3A and DNMT3B in addition to a single allele of DNMT1 (ref. 37). We stably introduced a subset of isoforms into the 3ABDKO cell line and subsequently treated them with 5-Aza-CdR for 24 h. In general, these isoforms are stably expressed up to 42 days after treatment (Supplementary Fig. 9). We observed that only DNMT3B1 could robustly restore DNA methylation, while the catalytically inactive forms, mutant DNMT3B1, DNMT3B3 and DNMT3L, did not elicit the re-methylation at a majority of targets in the absence of DNMT3A (Fig. 3b). Although methylation was restored at a small portion of the targets in cells with or without isoforms of DNMT3B or DNMT3L (Fig. 3b), this could be explained by the known de novo activity of DNMT1 at specific regions10,37. In addition, 86% of these sites also gained DNA methylation in the 3BKO cell lines (Fig. 3a). This result therefore not only confirmed the well-known functional role of DNMT3B1 that is mainly expressed in ES cells24, but also a completely novel role as accessory protein for DNMT3B especially the catalytically inactive DNMT3B isoforms, such as DNMT3B3, which is highly expressed in somatic cells24. As we expected, Group IV CpGs were not showing increases in DNA methylation (Supplementary Fig. 10). These results therefore confirm that DNMT3B can act as an accessory protein like DNMT3L to establish DNA methylation by recruiting DNMT3A in somatic cells.
Overexpression of DNMT3s in different cancer types
The role of DNMT3B in acting as an accessory protein may also contribute to aberrant de novo methylation during tumourigenesis. Since overexpression of DNMTs38, especially DNMT3s and their isoforms, is commonly seen in tumours12,19,29,39. Our previous work suggests that DNMT3A requires DNMT3B for restoration of methylation in somatic cells and DNMT3B is also required for re-methylation after 5-Aza-CdR treatment23,35; however, the functional role of DNMT3B and its isoforms in a complex with DNMT3A is still not clear. DNMTs are overexpressed in cancer cells19, although detailed analysis of the expression profiles has not been described. We took advantage of The Cancer Genome Atlas (TCGA) project, which includes RNA-seq data on eight primary tumour types, to study expression levels of DNMT3s in normal tissues and their corresponding tumours. We confirmed overexpression of DNMT3A in six out of eight cancer types and DNMT3B overexpression in all cancer types, compared with normal tissues (Fig. 4). In addition, multiple isoforms of DNMT3B were also significantly overexpressed in these types of cancers (Fig. 4), including catalytically inactive DNMT3B3. Delta isoforms, DNMTΔ3B2 and four were purposefully not included in this analysis because of the difficulty to distinguish these isoforms by RNA-seq data (Fig. 4).
Normalized read counts from RNA-Seq data were calculated for DNMT3A, DNMT3B and isoforms from matched normal and tumour tissue samples by expectation-maximization (RSEM). Expression fold change is shown as log2 (tumour/normal) for each cancer type. N indicates the number of sample pairs for each tumour type. Mann–Whitney’s unpaired statistical test was used to assess expression differences on log2 (RSEM) values between tumour and normal tissue. *P<0.05, **P<0.01 or ***P<0.001.
Discussion
In this study we investigated the DNA-methylation target sites of DNMT3B isoforms on a genome-wide level by restoring the expression of a representative panel of DNMT3B isoforms in two different DNMT3B-deficient cell lines. Genome-wide analysis of DNA-methylation changes reveals that catalytic activity is not required for the induced DNA methylation, indicating an accessory protein role for DNMT3B isoforms. The presence of the N-terminal and PWWP domains was found to be important for DNMT3B function and its role as an accessory protein, except for DNMT3B4. Preferential binding of DNMT3B isoforms to regions known to be enriched with the H3K36me3 modification was found in our earlier work, as well as in others23,25,26. In addition, we confirmed the aberrant expression of DNMT3B isoforms in various cancers, using publically available data sets. Taken altogether, we show that DNMT3B can act as an accessory protein to restore DNA methylation of gene bodies in differentiated cells.
Our results, and other studies, have shown that DNMT3Bs are able to bind to the active DNMT3A molecules in vivo and in vitro32. The data here clearly show that DNMT3B1 can act as an accessory protein even when it is catalytically inactive, although it seems to require intact N-terminal and PWWP domains with the exception of DNMT3B4, thus further studies are required. Since catalytically inactive DNMT3B1 can establish de novo DNA-methylation patterns in both DNMT1 and DNMT3B-deficient cells but not in DNMT3B and DNMT3A deficient cells, it seems most likely that DNMT3B acts as an accessory protein for DNMT3A rather than DNMT1 (ref. 35). Although DNMT3B3 catalytic activity is controversial, our study suggests that DNA methylation in the presence of the DNMT3B3 isoform could be caused by it acting as an accessory protein in differentiated cells. This interpretation is consistent with the results of Liao et al.24 which showed that DNMT3A was necessary for de novo methylation of gene bodies during ES cell differentiation.
The changing landscapes of catalytically active and inactive proteins included in ES and differentiated cells are depicted in Fig. 5. DNMT3L has been identified as a DNMT3A accessory protein to stimulate de novo methylation in ES cells8,40,41,42,43,44,45, while UHRF1 has also been recognized as a DNMT1 accessory protein to maintain DNA-methylation patterns in embryonic and differentiated cells46,47. Our findings, for the first time, suggest a role for DNMT3B as an accessory protein for DNMT3A to stimulate and restore DNA methylation. While DNMT3A alone cannot restore DNA methylation in 3BKO cells, DNMT3B1 can achieve this in 3ABDKO cells. This suggests that DNMT3B1 can have a dual role in acting as both a methyltransferase and an accessory protein. The latter role could be especially pronounced when different isoforms of DNMT3B are expressed simultaneously, interacting with each other to boost DNA methylation32. Furthermore, DNMT3L recruits DNMT3s to specifically bind to nucleosomes without H3K4 methylation and stimulate de novo methylation during embryonic development34,41. Our results showing overlapping target regions by DNMT3B and DNMT3L suggest that H3K36me3 could also be a target signal for DNMT3L but this requires further studies.
In ES cells the catalytically active CpG methyltransferases DNMT3A1 (3A1), DNMT3A2 (3A2) and DNMT3B1 (3B1) are expressed, as well as the accessory protein DNMT3L (3L) (refs 40, 44, 53, 54). Our work suggests that catalytically inactive DNMT3B3 (3B3) may also participate as an accessory protein in the establishment of DNA-methylation patterns. Following differentiation 3A2 and 3L are no longer expressed, while expression of 3B1 is decreased markedly, leaving 3A1 and the accessory protein 3B3 as potential mediators of methylation53,54,55. Our work also suggests that 3B3 preferentially targets 3A1 to gene bodies. Active methyltransferase enzymes and accessory proteins are shown in red and blue boxes, respectively.
In conclusion, we have confirmed that DNMT3B isoforms can act as accessory proteins to restore DNA methylation, specifically in gene bodies in differentiated cells. This finding also suggests that DNMT3B and its catalytically inactive isoforms play key roles in collaborating with DNMT3A to initiate and maintain DNA methylation in transcribed regions. DNMT3A on the other hand has a low capability of restoring DNA methylation by itself without involvement of DNMT3B in differentiated cells. Our findings might also help explain the presence of aberrant DNA-methylation patterns during tumourigenesis due to the overexpression of DNMT3B isoforms, which could be a driving force for de novo methylation.
Methods
Cell lines and drug treatment
HCT116 derivative cell lines DKO8, 3BKO and 3ABDKO were obtained from Dr Baylin’s laboratory and cultured in McCoy’s 5A medium containing 1% penicillin/streptomycin and 10% inactivated fetal bovine serum in a humidified and 5% CO2 containing atmosphere at 37 °C. All cell lines were verified to be free of Mycoplasma contamination before use. Cell lines were treated with 0.3 μM of 5-Aza-CdR (Sigma-Aldrich) for 24 h followed by a medium change.
DNMT isoform constructs
Human DNMT isoforms 3B1, Δ3B2, 3B3, Δ3B4 and 3L, containing a MYC-tagged DNA sequence ligated to the 5′- ends, were amplified from pIRESpuro/Myc constructs48 (a modified version of the pIRESpuro3 vector, Clontech), a gift from Dr Allen Yang (USC). Catalytically inactive mutants containing a cysteine to serine alteration in position 651 of DNMT3B1 and 452 of DNMTΔ3B2 proteins were established as previously described35. MYC-tagged DNMT sequences were cloned into pLJM1 lentivirus vector at EcoRI and AgeI sites using Infusion HD PCR Cloning Plus (Clontech) following the manufacturer’s protocol. To produce lentivirus for the specific constructs, the vesicular stomatitis virus envelope protein G-expression construct pMD.G1, the packaging vector pCMV ΔR8.91 and transfer vector pLJM1 were used as previously described49. All vectors were amplified and purified using the PureYield Plasmid Maxiprep system (Promega), according to the manufacturer’s instructions. The HCT116 derivative cell lines 3BKO, 3ABDKO and DKO8 were stably transfected with a lentivirus and selected with 2 μg ml−1 puromycin for 14, 14 and 21 days, respectively.
Protein extraction and western blot analysis
Cells were trypsinised, washed with PBS and resuspended in RIPA buffer (50 mM Tris-HCl, ph 8.0, 150 mM NaCl, 1% NP-40, 0.5% DOC, 0.1% SDS) with protease inhibitors (Roche, 04693132001). The lysed cells were then sonicated on ice and cellular debris were removed by centrifugation. In all, 2 μg of protein was mixed with SDS/β-mercaptoethanol loading buffer and resolved on a BioRad 4-15% gradient SDS/PAGE gel. Antibodies against the MYC-epitope tag (Millipore, 05-724, 1:1,000) and β-ACTIN (Sigma, A2228, 1:2,000) as loading control were used. Proteins were visualized using the ECL detection system (Thermo Scientific) and BioRad’s ChemiDoc system. Protein was extracted on day 56 for DKO8 cells and day 14 for 3BKO cells. The full-size images are shown in Supplementary Fig. 11.
RNA isolation and qRT-PCR
Puromycin-resistant polyclonal cells, stably transfected with DNMT isoforms, were used for total RNA extraction using the RNeasy Mini kit (Qiagen). In all, 1 μg of total RNA was treated with 10 U of DNaseI (Roche) at 25 °C for 20 min followed by 8 mM final concentration of EDTA and 10 min heat inactivation at 75 °C. Typically, 1 μg of DNase treated RNA was reverse transcribed using iScript Reverse Transcription Supermix (BioRad), according to the manufacturer’s instructions. PCR reactions were performed using KAPA SYBR FAST University 2 × qPCR Master Mix. Primer sequences used in qRT-PCR: TBP sense 5′-GCCCGAAACGCCGAATAT-3′, antisense 5′-CCGTGGTTCGTGGCTCTCT-3′; PCNA sense 5′-GCAGATGTACCCCTTGTTGTAGAGT-3′, antisense 5′-TCTTCATCCTCGATCTTGGGA-3′; DNMT3A sense 5′-CAATGACCTCTCCATCGTCAAC-3′, antisense 5′-CATGCAGGAGGCGGTAGAA-3′; DNMT3B sense 5′-CTGCCGGTGTTTCTGTGTGG-3′, antisense 5′-TGTAACAGCTCCAGGGCTCC-3′; DNMT3L sense 5′-TACGCACGGCCCAAGC-3′, antisense 5′-ACCAGATTGTCCACGAACATCC-3′. RNA-expression analysis was done on days 21 and 56 post transfection on DKO8 cells. RNA-expression analysis was done on day 14 post transfection on 3BKO and 3ABDKO cells and on days 0 and 42 post 5-Aza-CdR treatment.
Illumina Infinium HM450 DNA-methylation data processing
DNA was extracted after 56 days of DNMT re-expression. DNA methylation was assessed using Illumina’s Infinium HumanMethylation450 (HM450) BeadChip array and was performed at the USC Epigenome Center according to the manufacturer’s specifications. The array examines the DNA-methylation status of 482,421 CpG sites and each is reported as a β-value, ranging from 0 (unmethylated) to 1 (fully methylated). CpG probes with a detection P value >0.05, located within 15 base pairs of a single-nucleotide polymorphism or located in gene deserts, were excluded from further analysis in all samples leaving 385,826. Analysis and β-value calculations were performed as described elsewhere23,50,51. Changes of 0.2 (20%) β-value were considered target sites for analyses and are described elsewhere48. Statistical enrichment was performed using a z-test comparing the targeted CpG sites for each DNMT isoform with the overall distribution of probes on the 450 K array. DKO8 cells at day 56 were analysed for DNA methylation on the HM450 array, as well as 3BKO and 3 ABDKO cells on days 0, 5 and 42 post 5-Aza-CdR treatment.
RNA-seq data collection and analysis
RNASeqV2 Level 3 data from TCGA was obtained from the publically available data portal (http://cancergenome.nih.gov/dataportal/). Values for gene-expression values were aligned using MapSplice and quantification was performed using RSEM52.
Data availability
The genome-wide DNA-methylation data that support the findings of this study have been deposited in the Gene Expression Omnibus (GEO) database with the accession codes GSE51815 and GSE68344.
Additional information
Accession codes: The DNA-methylation data obtained from the Illumina’s Infinium HumanMethylation450 BeadChip have been deposited in the Gene Expression Omnibus database under the accession code GSE68344.
How to cite this article: Duymich, C. E. et al. DNMT3B Isoforms without catalytic activity stimulate gene body methylation as accessory proteins in somatic cells. Nat. Commun. 7:11453 doi: 10.1038/ncomms11453 (2016).
Accession codes
References
Bird, A. DNA methylation patterns and epigenetic memory. Genes Dev. 16, 6–21 (2002).
Jurkowska, R. Z., Jurkowski, T. P. & Jeltsch, A. Structure and function of mammalian DNA methyltransferases. Chembiochem 12, 206–222 (2011).
Esteller, M. Cancer epigenetics for the 21st century: what's next? Genes Cancer 2, 604–606 (2011).
Jones, P. A. Functions of DNA methylation: islands, start sites, gene bodies and beyond. Nat. Rev. Genet. 13, 484–492 (2012).
Jones, P. A. & Baylin, S. B. The epigenomics of cancer. Cell 128, 683–692 (2007).
Sharma, S., Kelly, T. K. & Jones, P. A. Epigenetics in cancer. Carcinogenesis 31, 27–36 (2010).
Goll, M. G. & Bestor, T. H. Eukaryotic cytosine methyltransferases. Annu. Rev. Biochem. 74, 481–514 (2005).
Gowher, H., Liebert, K., Hermann, A., Xu, G. & Jeltsch, A. Mechanism of stimulation of catalytic activity of Dnmt3A and Dnmt3B DNA-(cytosine-C5)-methyltransferases by Dnmt3L. J. Biol. Chem. 280, 13341–13348 (2005).
Chen, T. et al. Complete inactivation of DNMT1 leads to mitotic catastrophe in human cancer cells. Nat. Genet. 39, 391–396 (2007).
Liang, G. et al. Cooperativity between DNA methyltransferases in the maintenance methylation of repetitive elements. Mol. Cell. Biol. 22, 480–491 (2002).
Chen, T., Ueda, Y., Dodge, J. E., Wang, Z. & Li, E. Establishment and maintenance of genomic methylation patterns in mouse embryonic stem cells by Dnmt3a and Dnmt3b. Mol. Cell. Biol. 23, 5594–5605 (2003).
Jones, P. A. & Liang, G. Rethinking how DNA methylation patterns are maintained. Nat. Rev. Genet. 10, 805–811 (2009).
Ostler, K. R. et al. Cancer cells express aberrant DNMT3B transcripts encoding truncated proteins. Oncogene 26, 5553–5563 (2007).
Xie, S. et al. Cloning, expression and chromosome locations of the human DNMT3 gene family. Gene 236, 87–95 (1999).
Wang, J., Walsh, G., Liu, D. D., Lee, J. J. & Mao, L. Expression of Delta DNMT3B variants and its association with promoter methylation of p16 and RASSF1A in primary non-small cell lung cancer. Cancer Res. 66, 8361–8366 (2006).
Gopalakrishnan, S. et al. A novel DNMT3B splice variant expressed in tumor and pluripotent cells modulates genomic DNA methylation patterns and displays altered DNA binding. Mol. Cancer Res. 7, 1622–1634 (2009).
Okano, M., Xie, S. & Li, E. Cloning and characterization of a family of novel mammalian DNA (cytosine-5) methyltransferases. Nat. Genet. 19, 219–220 (1998).
Okano, M., Bell, D. W., Haber, D. A. & Li, E. DNA methyltransferases Dnmt3a and Dnmt3b are essential for de novo methylation and mammalian development. Cell 99, 247–257 (1999).
Robertson, K. D. et al. The human DNA methyltransferases (DNMTs) 1, 3a and 3b: coordinate mRNA expression in normal tissues and overexpression in tumors. Nucleic Acids Res. 27, 2291–2298 (1999).
Saito, Y. et al. Overexpression of a splice variant of DNA methyltransferase 3b, DNMT3b4, associated with DNA hypomethylation on pericentromeric satellite regions during human hepatocarcinogenesis. Proc. Natl Acad. Sci. USA 99, 10060–10065 (2002).
Varley, K. E. et al. Dynamic DNA methylation across diverse human cell lines and tissues. Genome Res. 23, 555–567 (2013).
Kulis, M. et al. Epigenomic analysis detects widespread gene-body DNA hypomethylation in chronic lymphocytic leukemia. Nat. Genet. 44, 1236–1242 (2012).
Yang, X. et al. Gene body methylation can alter gene expression and is a therapeutic target in cancer. Cancer Cell 26, 1–14 (2014).
Liao, J. et al. Targeted disruption of DNMT1, DNMT3A and DNMT3B in human embryonic stem cells. Nat. Genet. 47, 469–478 (2015).
Baubec, T. et al. Genomic profiling of DNA methyltransferases reveals a role for DNMT3B in genic methylation. Nature 520, 243–247 (2015).
Morselli, M. et al. In vivo targeting of de novo DNA methylation by histone modifications in yeast and mouse. Elife 4, e06205 (2015).
Jones, P. A., Rideout, W. M., Shen, J. C., Spruck, C. H. & Tsai, Y. C. Methylation, mutation and cancer. Bioessays 14, 33–36 (1992).
Maunakea, A. K. et al. Conserved role of intragenic DNA methylation in regulating alternative promoters. Nature 466, 253–257 (2010).
Rhee, I. et al. DNMT1 and DNMT3b cooperate to silence genes in human cancer cells. Nature 416, 552–556 (2002).
Egger, G. et al. Identification of DNMT1 (DNA methyltransferase 1) hypomorphs in somatic knockouts suggests an essential role for DNMT1 in cell survival. Proc. Natl Acad. Sci. USA 103, 14080–14085 (2006).
Chen, T. & Li, E. Establishment and maintenance of DNA methylation patterns in mammals. Curr. Top. Microbiol. Immunol. 301, 179–201 (2006).
Gordon, C. A., Hartono, S. R. & Chédin, F. Inactive DNMT3B splice variants modulate de novo DNA methylation. PLoS ONE 8, e69486 (2013).
Weisenberger, D. J. et al. Role of the DNA methyltransferase variant DNMT3b3 in DNA methylation. Mol. Cancer Res. 2, 62–72 (2004).
Lucifero, D. et al. Coordinate regulation of DNA methyltransferase expression during oogenesis. BMC Dev. Biol. 7, 36 (2007).
Sharma, S., De Carvalho, D. D., Jeong, S., Jones, P. A. & Liang, G. Nucleosomes containing methylated DNA stabilize DNA methyltransferases 3A/3B and ensure faithful epigenetic inheritance. PLoS Genet. 7, e1001286 (2011).
Jeong, S. et al. Selective anchoring of DNA methyltransferases 3A and 3B to nucleosomes containing methylated DNA. Mol. Cell. Biol. 29, 5366–5376 (2009).
Jair, K. W. et al. De novo CpG island methylation in human cancer cells. Cancer Res. 66, 682–692 (2006).
Kautiainen, T. L. & Jones, P. A. DNA methyltransferase levels in tumorigenic and nontumorigenic cells in culture. J. Biol. Chem. 261, 1594–1598 (1986).
Linhart, H. G. et al. Dnmt3b promotes tumorigenesis in vivo by gene-specific de novo methylation and transcriptional silencing. Genes Dev. 21, 3110–3122 (2007).
Bourc'his, D., Xu, G. L., Lin, C. S., Bollman, B. & Bestor, T. H. Dnmt3L and the establishment of maternal genomic imprints. Science 294, 2536–2539 (2001).
Ooi, S. K. et al. DNMT3L connects unmethylated lysine 4 of histone H3 to de novo methylation of DNA. Nature 448, 714–717 (2007).
Neri, F. et al. Dnmt3L antagonizes DNA methylation at bivalent promoters and favors DNA methylation at gene bodies in ESCs. Cell 155, 121–134 (2013).
Guo, X. et al. Structural insight into autoinhibition and histone H3-induced activation of DNMT3A. Nature 517, 640–644 (2015).
Jia, D., Jurkowska, R. Z., Zhang, X., Jeltsch, A. & Cheng, X. Structure of Dnmt3a bound to Dnmt3L suggests a model for de novo DNA methylation. Nature 449, 248–251 (2007).
Chen, Z. X., Mann, J. R., Hsieh, C. L., Riggs, A. D. & Chédin, F. Physical and functional interactions between the human DNMT3L protein and members of the de novo methyltransferase family. J. Cell. Biochem. 95, 902–917 (2005).
Bostick, M. et al. UHRF1 plays a role in maintaining DNA methylation in mammalian cells. Science 317, 1760–1764 (2007).
Sharif, J. et al. The SRA protein Np95 mediates epigenetic inheritance by recruiting Dnmt1 to methylated DNA. Nature 450, 908–912 (2007).
Choi, S. H. et al. Identification of preferential target sites for human DNA methyltransferases. Nucleic Acids Res. 39, 104–118 (2011).
Ou, C. Y., Kim, J. H., Yang, C. K. & Stallcup, M. R. Requirement of cell cycle and apoptosis regulator 1 for target gene activation by Wnt and beta-catenin and for anchorage-independent growth of human colon carcinoma cells. J. Biol. Chem. 284, 20629–20637 (2009).
De Carvalho, D. D. et al. DNA methylation screening identifies driver epigenetic events of cancer cell survival. Cancer Cell 21, 655–667 (2012).
Pandiyan, K. et al. Functional DNA demethylation is accompanied by chromatin accessibility. Nucleic Acids Res. 41, 3973–3985 (2013).
Wang, K. et al. MapSplice: accurate mapping of RNA-seq reads for splice junction discovery. Nucleic Acids Res. 38, e178 (2010).
Chen, T., Ueda, Y., Xie, S. & Li, E. A novel Dnmt3a isoform produced from an alternative promoter localizes to euchromatin and its expression correlates with active de novo methylation. J. Biol. Chem. 277, 38746–38754 (2002).
Aapola, U. et al. Isolation and initial characterization of a novel zinc finger gene, DNMT3L, on 21q22.3, related to the cytosine-5-methyltransferase 3 gene family. Genomics 65, 293–298 (2000).
Lei, H. et al. De novo DNA cytosine methyltransferase activities in mouse embryonic stem cells. Development 122, 3195–3205 (1996).
Acknowledgements
Funding for this work was provided by RO1CA082422 (P.A.J.), R01CA083867 (P.A.J. and G.L.). J.C. was funded by a postdoctoral AFR grant from the Fonds National de la Recherche Luxembourg (5777110). 3BKO, 3ABDKO and DKO8 cells were kindly provided by Drs Bert Vogelstein, Stephen B. Baylin and Yi Cai. We thank Dr Kimberly Siegmund for bioinformatic and statistical assistance, and TCGA for making the data publically available.
Author information
Authors and Affiliations
Contributions
C.E.D. performed all the experiments and data analysis. C.E.D., G.L. and P.A.J. conceived the experiments and wrote the manuscript. C.E.D., J.C. and X.Y. performed the drug-treatment experiments. All authors provided helpful guidance on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-11 and Supplementary Reference (PDF 7791 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Duymich, C., Charlet, J., Yang, X. et al. DNMT3B isoforms without catalytic activity stimulate gene body methylation as accessory proteins in somatic cells. Nat Commun 7, 11453 (2016). https://doi.org/10.1038/ncomms11453
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms11453
This article is cited by
-
Serine protease PRSS56, a novel cancer-testis antigen activated by DNA hypomethylation, promotes colorectal and gastric cancer progression via PI3K/AKT axis
Cell & Bioscience (2023)
-
Expression analysis suggests that DNMT3L is required for oocyte de novo DNA methylation only in Muridae and Cricetidae rodents
Epigenetics & Chromatin (2023)
-
Genomic targets and selective inhibition of DNA methyltransferase isoforms
Clinical Epigenetics (2022)
-
Mechanisms of chromatin-based epigenetic inheritance
Science China Life Sciences (2022)
-
DNA methyltransferase 3A controls intestinal epithelial barrier function and regeneration in the colon
Nature Communications (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.