Abstract
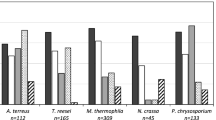
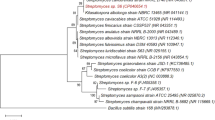
Wood biomass is the most abundant feedstock envisioned for the development of modern biorefineries. However, the cost-effective conversion of this form of biomass into commodity products is limited by its resistance to enzymatic degradation. Here we describe a new family of fungal lytic polysaccharide monooxygenases (LPMOs) prevalent among white-rot and brown-rot basidiomycetes that is active on xylans—a recalcitrant polysaccharide abundant in wood biomass. Two AA14 LPMO members from the white-rot fungus Pycnoporus coccineus substantially increase the efficiency of wood saccharification through oxidative cleavage of highly refractory xylan-coated cellulose fibers. The discovery of this unique enzyme activity advances our knowledge on the degradation of woody biomass in nature and offers an innovative solution for improving enzyme cocktails for biorefinery applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Field, C.B., Behrenfeld, M.J., Randerson, J.T. & Falkowski, P. Primary production of the biosphere: integrating terrestrial and oceanic components. Science 281, 237–240 (1998).
Himmel, M.E. et al. Biomass recalcitrance: engineering plants and enzymes for biofuels production. Science 315, 804–807 (2007).
Biely, P., Singh, S. & Puchart, V. Towards enzymatic breakdown of complex plant xylan structures: state of the art. Biotechnol. Adv. 34, 1260–1274 (2016).
Simmons, T.J. et al. Folding of xylan onto cellulose fibrils in plant cell walls revealed by solid-state NMR. Nat. Commun. 7, 13902 (2016).
Loqué, D., Scheller, H.V. & Pauly, M. Engineering of plant cell walls for enhanced biofuel production. Curr. Opin. Plant Biol. 25, 151–161 (2015).
Hibbett, D.S. & Donoghue, M.J. Analysis of character correlations among wood decay mechanisms, mating systems, and substrate ranges in homobasidiomycetes. Syst. Biol. 50, 215–242 (2001).
Lombard, V., Golaconda Ramulu, H., Drula, E., Coutinho, P.M. & Henrissat, B. The carbohydrate-active enzymes database (CAZy) in 2013. Nucleic Acids Res. 42, D490–D495 (2014).
Riley, R. et al. Extensive sampling of basidiomycete genomes demonstrates inadequacy of the white-rot/brown-rot paradigm for wood decay fungi. Proc. Natl. Acad. Sci. USA 111, 9923–9928 (2014).
Vaaje-Kolstad, G. et al. An oxidative enzyme boosting the enzymatic conversion of recalcitrant polysaccharides. Science 330, 219–222 (2010).
Quinlan, R.J. et al. Insights into the oxidative degradation of cellulose by a copper metalloenzyme that exploits biomass components. Proc. Natl. Acad. Sci. USA 108, 15079–15084 (2011).
Kracher, D. et al. Extracellular electron transfer systems fuel cellulose oxidative degradation. Science 352, 1098–1101 (2016).
Johansen, K.S. Discovery and industrial applications of lytic polysaccharide mono-oxygenases. Biochem. Soc. Trans. 44, 143–149 (2016).
Hemsworth, G.R., Henrissat, B., Davies, G.J. & Walton, P.H. Discovery and characterization of a new family of lytic polysaccharide monooxygenases. Nat. Chem. Biol. 10, 122–126 (2014).
Vu, V.V., Beeson, W.T., Span, E.A., Farquhar, E.R. & Marletta, M.A. A family of starch-active polysaccharide monooxygenases. Proc. Natl. Acad. Sci. USA 111, 13822–13827 (2014).
Lo Leggio, L. et al. Structure and boosting activity of a starch-degrading lytic polysaccharide monooxygenase. Nat. Commun. 6, 5961 (2015).
Couturier, M. et al. Enhanced degradation of softwood versus hardwood by the white-rot fungus Pycnoporus coccineus. Biotechnol. Biofuels 8, 216 (2015).
Frandsen, K.E. et al. The molecular basis of polysaccharide cleavage by lytic polysaccharide monooxygenases. Nat. Chem. Biol. 12, 298–303 (2016).
Peisach, J. & Blumberg, W.E. Structural implications derived from the analysis of electron paramagnetic resonance spectra of natural and artificial copper proteins. Arch. Biochem. Biophys. 165, 691–708 (1974).
Garajova, S. et al. Single-domain flavoenzymes trigger lytic polysaccharide monooxygenases for oxidative degradation of cellulose. Sci. Rep. 6, 28276 (2016).
Bennati-Granier, C. et al. Substrate specificity and regioselectivity of fungal AA9 lytic polysaccharide monooxygenases secreted by Podospora anserina. Biotechnol. Biofuels 8, 90 (2015).
Herpoël-Gimbert, I. et al. Comparative secretome analyses of two Trichoderma reesei RUT-C30 and CL847 hypersecretory strains. Biotechnol. Biofuels 1, 18 (2008).
Westereng, B. et al. Enzymatic cellulose oxidation is linked to lignin by long-range electron transfer. Sci. Rep. 5, 18561 (2015).
Villares, A. et al. Lytic polysaccharide monooxygenases disrupt the cellulose fibers structure. Sci. Rep. 7, 40262 (2017).
Frommhagen, M. et al. Discovery of the combined oxidative cleavage of plant xylan and cellulose by a new fungal polysaccharide monooxygenase. Biotechnol. Biofuels 8, 101 (2015).
Fanuel, M. et al. The Podospora anserina lytic polysaccharide monooxygenase PaLPMO9H catalyzes oxidative cleavage of diverse plant cell wall matrix glycans. Biotechnol. Biofuels 10, 63 (2017).
McCartney, L. et al. Differential recognition of plant cell walls by microbial xylan-specific carbohydrate-binding modules. Proc. Natl. Acad. Sci. USA 103, 4765–4770 (2006).
Nieduszynski, I. & Marchessault, R.H. Structure of β-D-(1→4′)xylan hydrate. Nature 232, 46–47 (1971).
Miyauchi, S. et al. Visual comparative omics of fungi for plant biomass Deconstruction. Front. Microbiol. 7, 1335 (2016).
Altschul, S.F., Gish, W., Miller, W., Myers, E.W. & Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 215, 403–410 (1990).
Edgar, R.C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Katoh, K. & Standley, D.M. MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evol. 30, 772–780 (2013).
Talavera, G. & Castresana, J. Improvement of phylogenies after removing divergent and ambiguously aligned blocks from protein sequence alignments. Syst. Biol. 56, 564–577 (2007).
Price, M.N., Dehal, P.S. & Arkin, A.P. FastTree 2--approximately maximum-likelihood trees for large alignments. PLoS One 5, e9490 (2010).
Huson, D.H. & Scornavacca, C. Dendroscope 3: an interactive tool for rooted phylogenetic trees and networks. Syst. Biol. 61, 1061–1067 (2012).
Vos, R.A., Caravas, J., Hartmann, K., Jensen, M.A. & Miller, C. BIO:Phylo-phyloinformatic analysis using perl. BMC Bioinformatics 12, 63 (2011).
Haon, M. et al. Recombinant protein production facility for fungal biomass-degrading enzymes using the yeast Pichia pastoris. Front. Microbiol. 6, 1002 (2015).
Kittl, R., Kracher, D., Burgstaller, D., Haltrich, D. & Ludwig, R. Production of four Neurospora crassa lytic polysaccharide monooxygenases in Pichia pastoris monitored by a fluorimetric assay. Biotechnol. Biofuels 5, 79 (2012).
Englyst, H.N. & Cummings, J.H. Improved method for measurement of dietary fiber as non-starch polysaccharides in plant foods. J. Assoc. Off. Anal. Chem. 71, 808–814 (1988).
Westbye, P., Svanberg, C. & Gatenholm, P. The effect of molecular composition of xylan extracted from birch on its assembly onto bleached softwood kraft pulp. Holzforschung 60, 143–148 (2006).
Larsson, P.T., Wickholm, K. & Iversen, T. A CP/MAS 13C NMR investigation of molecular ordering in celluloses. Carbohydr. Res. 302, 19–25 (1997).
Kabsch, W. XDS. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Winn, M.D. et al. Overview of the CCP4 suite and current developments. Acta Crystallogr. D Biol. Crystallogr. 67, 235–242 (2011).
Cowtan, K. The Buccaneer software for automated model building. 1. Tracing protein chains. Acta Crystallogr. D Biol. Crystallogr. 62, 1002–1011 (2006).
Emsley, P., Lohkamp, B., Scott, W.G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Murshudov, G.N., Vagin, A.A. & Dodson, E.J. Refinement of macromolecular structures by the maximum-likelihood method. Acta Crystallogr. D Biol. Crystallogr. 53, 240–255 (1997).
Chen, V.B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010).
Berman, H., Henrick, K. & Nakamura, H. Announcing the worldwide Protein Data Bank. Nat. Struct. Biol. 10, 980 (2003).
Stoll, S. & Schweiger, A. EasySpin, a comprehensive software package for spectral simulation and analysis in EPR. J. Magn. Reson. 178, 42–55 (2006).
Acknowledgements
We thank the European Synchrotron Radiation Facility (Grenoble), and the synchrotron Soleil (Gif-sur-Yvette) for beamtime allocation and assistance. We thank S. Tapin (Centre Technique du Papier, France) for providing cellulose fibers, E. Bonnin and J. Vigouroux for compositional analyses, G. Toriz and P. Gatenholm (Chalmers University of Technology, Sweden) for providing purified wood xylan, L. Foucat and X. Falourd for their valued assistance with treatments of the NMR data, E. Perrin for the excellent technical support for TEM images, B. Seantier for the access and assistance to AFM facilities, D. Hartmann and E. Bertrand for their help with enzyme production in bioreactor, D. Gillet (Mahtani Chitosan, India) for providing chitin, and D. Navarro and G. Anasontzis for insightful discussions. M.C. was funded by a Marie Curie International Outgoing Fellowship within the 7th European Community Framework Program (328162). S.L., M.-N.R. and J.-G.B. were funded by the Microbio-E A*MIDEX project (ANR-11-IDEX-0001-02). This work was supported in part by the CNRS and the French Infrastructure for Integrated Structural Biology (FRISBI) ANR-10-INSB-05-01. N.L. and B.H. were supported by Agence Française de l'Environnement et de la Maîtrise de l'Energie (1201C102). P.H.W., G.J.D. and L.C. thank the UK Biotechnology and Biological Sciences Research Council (BB/L001926/1 and BB/L021633/1) for funding. G.J.D. is the Royal Society Ken Murray Research Professor.
Author information
Authors and Affiliations
Contributions
M.C. identified the new enzymes and performed biochemical characterization. M.-N.R. was in charge of transcriptomic and proteomic analyses. M.C., S.L., S.G., I.H.-G. and M.H. performed production of proteins in flasks and bioreactors. F.C. performed ICP-MS analysis. S.L. and S.G. performed synergy assays with xylanase and protein crystallization. S.L. and G.S. solved the crystal structure of PcAA14B. B.H. and N.L. performed bioinformatic analyses. M.C., S.L., S.G. performed HPAEC analyses. M.F., D.R. and H.R. identified oxidized products using mass spectrometry. M.C., S.G. and I.H.-G. performed saccharification assays. A.V., C.M. and B.C. carried out microscopy and NMR analyses. L.C. performed the EPR study under the direction of P.H.W. and G.J.D. J.-G.B. supervised the work and organized the data. The manuscript was written by J.-G.B. with contributions from B.H. and P.H.W. All authors made comments on the manuscript and approved the final version. Figures were prepared by J.-G.B., K.E.F., A.L., N.L., S.L., L.C., M.F., S.G. and I.H.-G.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Tables 1–4 and Supplementary Figures 1–13 (PDF 3061 kb)
Supplementary Data Set 1
Number of AA14 genes in fungal genomes (XLSX 27 kb)
Rights and permissions
About this article
Cite this article
Couturier, M., Ladevèze, S., Sulzenbacher, G. et al. Lytic xylan oxidases from wood-decay fungi unlock biomass degradation. Nat Chem Biol 14, 306–310 (2018). https://doi.org/10.1038/nchembio.2558
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.2558
This article is cited by
-
A novel AA14 LPMO from Talaromyces rugulosus with bifunctional cellulolytic/hemicellulolytic activity boosted cellulose hydrolysis
Biotechnology for Biofuels and Bioproducts (2024)
-
Expanding the catalytic landscape of metalloenzymes with lytic polysaccharide monooxygenases
Nature Reviews Chemistry (2024)
-
Enzymatic debranching is a key determinant of the xylan-degrading activity of family AA9 lytic polysaccharide monooxygenases
Biotechnology for Biofuels and Bioproducts (2023)
-
Visible light-exposed lignin facilitates cellulose solubilization by lytic polysaccharide monooxygenases
Nature Communications (2023)
-
Histidine oxidation in lytic polysaccharide monooxygenase
JBIC Journal of Biological Inorganic Chemistry (2023)