Abstract
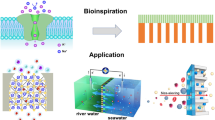
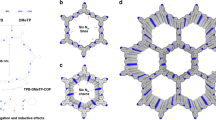
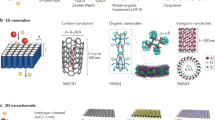
Transporting protons is essential in several biological processes as well as in renewable energy devices, such as fuel cells. Although biological systems exhibit precise supramolecular organization of chemical functionalities on the nanoscale to effect highly efficient proton conduction, to achieve similar organization in artificial systems remains a daunting challenge. Here, we are concerned with transporting protons on a micron scale under anhydrous conditions, that is proton transfer unassisted by any solvent, especially water. We report that proton-conducting systems derived from facially amphiphilic polymers that exhibit organized supramolecular assemblies show a dramatic enhancement in anhydrous conductivity relative to analogous materials that lack the capacity for self-organization. We describe the design, synthesis and characterization of these macromolecules, and suggest that nanoscale organization of proton-conducting functionalities is a key consideration in obtaining efficient anhydrous proton transport.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Williams, R. J. P. Proton circuits in biological energy interconversions. Annu. Rev. Biophys. Biophys. Chem. 17, 71–97 (1988).
Kreuer, K. D. Proton conductivity: materials and applications. Chem. Mater. 8, 610–641 (1996).
Sass, H. J. et al. Structural alterations for proton translocation in the M state of wild-type bacteriorhodopsin. Nature 406, 649–653 (2000).
Schnell, J. R. & Chou, J. J. Structure and mechanism of the M2 proton channel of influenza A virus. Nature 451, 591–560 (2008).
Stouffer, A. L. et al. Structural basis for the function and inhibition of an influenza virus proton channel. Nature 451, 596–600 (2008).
Carrette, L., Friedrich, K. A. & Stimming, U. Fuel cells – fundamentals and applications. Fuel Cells 1, 5–39 (2001).
Steele, B. C. H. & Heinzel, A. Materials for fuel-cell technologies. Nature 414, 345–352 (2001).
Mauritz, K. A. & Moore, R. B. State of understanding of Nafion, Chem. Rev. 104, 4535–4585 (2004).
Diat, O. & Gebel, G. Proton channels. Nature Mater. 7, 13–14 (2008).
Schmidt-Rohr, K. & Chen, Q. Parallel cylindrical water nanochannels in Nafion fuel-cell membranes. Nature Mater. 7, 75–83 (2008).
Elliott, J. A., Hanna, S., Elliott, A. M. S. & Cooley, G. E. Interpretation of the small-angle X-ray scattering from swollen and oriented perfluorinated ionomer membranes. Macromolecules 33, 8708–8713 (2000).
Hickner, M. A., Ghassemi, H., Kim, Y. S., Einsla, B. R. & McGrath, J. E. Alternative polymer systems for proton exchange membranes (PEMs). Chem. Rev. 104, 4587–4612 (2004).
Rikukawa, M. & Sanui, K. Proton-conducting polymer electrolyte membranes based on hydrocarbon polymers. Prog. Polym. Sci. 25, 1463–1502 (2000).
Li, Q., He, R., Jensen, J. O. & Bjerrum, N. J. Approaches and recent development of polymer electrolyte membranes for fuel cells operating above 100 °C. Chem. Mater. 15, 4896–4915 (2003).
Kreuer, K. D. Fast proton conductivity: a phenomenon between the solid and the liquid state? Solid State Ionics 94, 55–62 (1997).
Kreuer, K. D., Fuchs, A., Ise, M., Spaeth, M. & Maier, J. Imidazole and pyrazole-based proton conducting polymers and liquids. Electrochim. Acta 43, 1281–1288 (1998).
Scharfenberger, G. et al. Anhydrous polymeric proton conductors based on imidazole functionalized polysiloxane. Fuel Cells 6, 237–250 (2006).
Zhou, Z., Li, S. W., Zhang, Y. L., Liu, M. L. & Li, W. Promotion of proton conduction in polymer electrolyte membranes by 1H-1,2,3-triazole. J. Am. Chem. Soc. 127, 10824–10825 (2005).
Granados-Focil, S., Woudenberg, R. C., Yavuzcetin, O., Tuominen, M. T. & Coughlin, E. B. Water-free proton-conducting polysiloxanes: a study on the effect of heterocycle structure. Macromolecules 40, 8708–8713 (2007).
Persson, J. C. & Jannasch, P. Intrinsically proton-conducting benzimidazole units tethered to polysiloxanes. Macromolecules 38, 3283–3289 (2005).
Shogbon, C. B., Brousseau, J.-L., Zhang, H., Benicewicz, B. C. & Akpalu, Y. Determination of the molecular parameters and studies of the chain conformation of polybenzimidazole in DMAc/LiCl. Macromolecules 39, 9409–9418 (2006).
Subbaraman, R., Ghassemi, H. & Zawodzinski, T. A. Jr 4,5-Dicyano-1H-[1,2,3]-triazole as a proton transport facilitator for polymer electrolyte membrane fuel cells. J. Am. Chem. Soc. 129, 2238–2239 (2007).
Won, J. et al. Fixation of nanosized proton transport channels in membranes. Macromolecules 36, 3228–3234 (2003).
Serpico, J. M. et al. Transport and structural studies of sulfonated styrene–ethylene copolymer membranes. Macromolecules 35, 5916–5921 (2002).
Shi, Z. & Holdcroft, S. Synthesis and proton conductivity of partially sulfonated poly([vinylidene difluoride-co-hexafluoropropylene]-b-styrene) block copolymers. Macromolecules 38, 4193–4201 (2005).
Rubatat, L., Shi, Z., Diat, O., Holdcroft, S. & Frisken, B. J. Structural study of proton-conducting fluorous block copolymer membranes. Macromolecules 39, 720–730 (2006).
Cho, B. K., Jain, A., Gruner, S. M. & Wiesner, U. Mesophase structure – mechanical and ionic transport correlations in extended amphiphilic dendrons. Science 305, 1598–1601 (2004).
Kishimoto, K. et al. Nano-segregated polymeric film exhibiting high ionic conductivities. J. Am. Chem. Soc. 127, 15618–15623 (2005).
Wanakule, N. S. et al. Ionic conductivity of block copolymer electrolytes in the vicinity of order–disorder and order–order transitions. Macromolecules 42, 5642–5651 (2009).
Savariar, E. N., Aathimankandan, S. V. & Thayumanavan, S. Supramolecular assemblies from amphiphilic homopolymers: testing the scope. J. Am. Chem. Soc. 128, 16224–16230 (2006).
Basu, S., Vutukuri, D. R. & Thayumanavan, S. Homopolymer micelles in heterogeneous solvent mixtures. J. Am. Chem. Soc. 127, 16794–17695 (2005).
Wintersgill, M. C. & Fontanella, J. J. Complex impedance measurements on Nafion. Electrochim. Acta 43, 1533–1538 (1998).
Sanders, E. H. et al. Characterization of electrosprayed Nafion films. J. Power Sources 129, 55–61 (2004).
Ruotsalainen, T. et al. Structural hierarchy in flow-aligned hexagonally self-organized microphases with parallel polyelectrolytic structures. Macromolecules 36, 9437–9442 (2003).
Roy, A. et al. Influence of chemical composition and sequence length on the transport properties of proton exchange membranes. J. Polym. Sci. B 44, 2226–2239 (2006).
Schuster, M. F. H. & Meyer, W. H. Anhydrous proton-conducting polymers. Annu. Rev. Mater. Res. 33, 233–261 (2003).
Acknowledgements
This work was supported by the National Science Foundation through the Fueling the Future Center for Chemical Innovation at the University of Massachusetts Amherst (CHE-0739227). We thank W. de Jeu for discussions on the X-ray scattering results.
Author information
Authors and Affiliations
Contributions
S.T. and Y.C. conceived the molecular design. S.T., R.H. and Mark T. planned the project. Y.C, Michael T., S.C. and C.V. carried out the experiments and analysed the data. Y.C. and A.P. synthesized the discussed compounds, Michael T. and C.V. measured ionic conductivities, and S.C. performed SAXS. Results were discussed by R.H., Mark T. and S.T. All authors contributed to writing the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Chen, Y., Thorn, M., Christensen, S. et al. Enhancement of anhydrous proton transport by supramolecular nanochannels in comb polymers. Nature Chem 2, 503–508 (2010). https://doi.org/10.1038/nchem.629
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.629
This article is cited by
-
Gyroid nanostructured soft membranes formed by controlling the degree of crosslinking polymerization of bicontinuous cubic liquid-crystalline monomers
Polymer Journal (2021)
-
Co-tri MOF-impregnated Aquivion® composites as proton exchange membranes for fuel cell applications
Ionics (2021)
-
High proton conductivity polybenzimidazole proton exchange membrane based on phosphotungstic acid-anchored nano-Kevlar fibers
Journal of Materials Science (2019)
-
Novel proton-conductive nanochannel membranes with modified SiO2 nanospheres for direct methanol fuel cells
Journal of Solid State Electrochemistry (2018)
-
Nanocrack-regulated self-humidifying membranes
Nature (2016)