Abstract
3-Deoxysugars are important constituents of complex carbohydrates. For example, 2-keto-3-deoxy-D-manno-octulosonic acid (KDO) is an essential component of lipopolysaccharides in Gram-negative bacteria, 2-keto-3-deoxy-D-glycero-D-galacto-nonulosonic acid (KDN) is widely found in carbohydrates of the bacterial cell wall and in lower vertebrates, and sialic acid is a common cap of mammalian glycoproteins. Although ready access to such sugars would benefit the creation of vaccine candidates, antibiotics and small-molecule drugs, their chemical synthesis is difficult. Here we present a simple chemoenzymatic method for preparing differentially protected 3-deoxysugar derivatives from readily available starting materials. It exploits the promiscuous aldolase activity of the enzyme macrophomate synthase (MPS) to add pyruvate enolate diastereoselectively to a wide range of structurally complex aldehydes. A short synthesis of KDN illustrates the utility of this approach. Enzyme promiscuity, which putatively fosters large functional leaps in natural evolution, has great promise as a source of synthetically useful catalytic transformations.
Similar content being viewed by others
Main
Recent mechanistic work on the putative Diels-Alderase macrophomate synthase (MPS) revealed the enzyme to be a proficient aldolase. It promotes the reaction of a variety of aromatic and aliphatic aldehydes with pyruvate enolate, that is generated in situ by decarboxylation of oxaloacetate1. Of particular note is the enzyme's remarkable tolerance to substrate size and shape. Moreover, although simple substrates are converted with only modest selectivities, protected glyceraldehyde derivatives afford synthetically useful diastereoselectivities, suggesting that MPS might provide a mild and practical route to various 3-deoxysugars. Indeed, as shown here, a host of differentially protected 3-deoxysugar derivatives is accessible through the promiscuous MPS-catalysed pyruvate aldol reaction.
A number of routes to 3-deoxysugars have been described2. Because they generally involve circuitous, multistep procedures, direct addition of pyruvate enolate to an appropriate aldose, known as the Cornforth synthesis3, would be an attractive alternative. However, this process suffers from poor scope, low yields, irreproducibility and harsh reaction conditions. A catalytic Ni2+ work-up has been developed to increase reproducibility and yields4, but a general solution has proven elusive. This challenge has spurred the development of enzymatic processes5, such as those based on KDO aldolase6 or sialic acid aldolase7, which work well for unprotected sugars but are unproven with other substrates8. In contrast, an MPS-catalysed Cornforth reaction potentially offers dual advantages. First, unlike the present indirect chemical methods2, unprotected pyruvate generated in situ serves as the nucleophile. Second, in a departure from other enzymatic protocols8, the starting aldehydes are differentially protected sugars, which yield products that can be selectively manipulated downstream in a chemical synthesis.
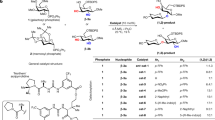
The MPS-catalysed Cornforth reaction with differentially protected trioses and tetroses such as 5, 8a and 8b (Fig. 1) confirmed that the enzyme tolerates functionally complex substrates. Racemic triose 5 was selected to examine the tolerance of MPS to synthetically useful protecting groups before committing to more complex substrate syntheses. It gave the triose aldol product 6, which contains both a methoxymethyl ether (MOM) group at the C5 hydroxyl and a selectively cleavable silyl protecting group at the C6 hydroxyl. Strategic release of the latter allows selective cyclization to the 3-deoxysugar (see 6 to 7 in Fig. 2a). Both tetroses 8a and 8b were converted to product in good yield, with the matched case (that is, 8a) (ref. 9) affording very high diastereoselectivity. The thioacetal group of aldehyde 8 serves as a useful handle for subsequent elongation and is readily accommodated at the enzyme active site. MPS appears to enforce stereoinduction regardless of the configuration of other stereocentres in the molecule. Thus enantiomers 8a and 8b gave diastereomeric products with the new stereocentre in the same configuration. The stereocentres of the products 9a and 9b are tentatively assigned the S configuration based on analogy to stereoproofs in the triose1, pentose (see Supplementary Information) and hexose series (see below).
a, Triose aldehyde 5 is readily converted to the differentially protected 3-deoxysugar derivative 6. b, Both enantiomers of thioacetal tetrose 8 are accepted as substrates by MPS. The reactions proceed with good to excellent diastereoselectivity for the mismatched (R,R) and matched (S,S) isomers, respectively. (TEOC: trimethylsilylethoxy-carbonate, d.r.: diastereomeric ratio).
Reactions with diverse pentoses further illustrate the scope of the MPS-catalysed pyruvate aldol reaction. Fully protected aldehydes of various pentoses were prepared by ozonolysis of the corresponding oximes (full details provided in Supplementary Information)10 with the goal of mapping the substrate range and protecting group tolerance of the enzyme active site. Diisopropylidene-protected sugars work efficiently in both the l- and D-sugar series (see, for example, 10a and 10b in Table 1). In contrast to 8, where the mismatched substrate enantiomer gave only a 4:1 diastereomeric ratio (see 8b to 9b in Fig. 1), the homologated sugar 10 led to the formation of products 16a, 16b and 16c in high diastereoselectivity irrespective of the aldehyde configuration. The configuration of the new stereocentre for 16a, 16b, and 17 was rigorously established as S by comparison to a known compound (see Supplementary Fig. S2), and the others (16c, 19, 20 and 21) are assigned by analogy. MPS tolerates ether, acetal, thioacetal, allyl, benzyl, silyl and ester protecting groups, as well as combinations thereof, in differentially protected sugar aldehydes (Table 1). In the cases where the aldehyde starting material was stable enough to be purified (for example 8a and 8b in Fig. 1 and 13a and 13b in Table 1) the enzymatic reaction was clean. The moderate yields of 30–65% seen for substrates 10–12 in Table 1 can likely be ascribed to the impurity and instability of the aldehydes used directly following oxime ozonolysis (see Supplementary Information for a full discussion). Positioning of an orthogonal protecting group at the C3 hydroxyl (aldehyde numbering) in pentoses 13a–c and hexose 15b allows strategic control of ring closure to the 3-deoxysugar derivative (see, for example, Fig. 2b). Although the enzyme active site can accommodate sterically bulky and hydrophobic protecting groups, the reaction yield suffers when polar groups are close to the aldehyde centre. For example, the peracetylated aldehyde derived from D-arabinose was not converted by MPS. Substrates of low solubility in aqueous buffer, including multiply benzylated or silylated aldehydes, were similarly ineffective, producing no detectable product.
MPS also provides access to C9 sugar derivatives from the corresponding hexoses (substrates 14 and 15a–b in Table 1). Diisopropylidene-protected glucosamine aldehyde 14 was synthesized and subjected to MPS and oxaloacetate. Although the conversion was only 50%, stereoselectivity was excellent. Aldehydes 15a and 15b, which were synthesized in four steps from D-mannose, were also good substrates in the aldol reaction. The products of these reactions, α-keto acids 21a and 21b, are in the same stereoisomeric series as KDN. Indeed, acidic deprotection of 21a afforded a product identical to a commercial sample of KDN as judged by high-resolution mass spectrometry and 1H NMR spectroscopy (see Fig. 2c and Supplementary Information). The short (six steps overall) synthesis of KDN, a valuable member of the higher sugar family11, from D-mannose once again demonstrates the preference of MPS for S stereoinduction in the carbon–carbon bond-forming step.
The seemingly general preference of MPS for inducing the S configuration regardless of substrate bias suggests a selective aldehyde binding mode. The recently determined X-ray structure of MPS containing a pyruvate molecule bound to the active site magnesium ion12 enables rationalization of this preference. Figure 3a shows the active site formed at the interface of two subunits. The wide entrance leading to the deeply buried binding pocket is consistent with the broad substrate tolerance observed. The preferred binding mode of the aldehyde, shown in Fig. 3b,c, is most likely set by three factors. First, proximity to the positively charged arginine residue (Arg101) is needed to polarize the carbonyl group and stabilize the incipient negative charge created in the aldol reaction. Second, the aldehyde should be aligned antiperiplanar to the enolate, as is preferred for open transition states in aldol reactions13. Last, the aldehyde should be positioned such that its alkyl group is facing the large entrance to the active site. The binding mode illustrated in Fig. 3 meets all three of the binding conditions and predicts the S stereochemistry of the product. Closed Zimmerman–Traxler-type transition states14 were not considered as a synclinal arrangement through a magnesium chelate would be sterically crowded around the magnesium ion and because steric clashes between the alkyl group of the aldehyde and a number of active site residues would preclude the observed S stereoselectivity.
a, View into the MPS active site12 with the S,S-thioaldehyde substrate 4a docked proximal to the magnesium-bound pyruvate enolate (substrate carbons are shown in teal, magnesium in magenta). The wide entrance to the binding pocket can readily accommodate a wide range of structurally complex aldehydes. b, The dimensions of the binding pocket adjacent to the magnesium-bound pyruvate dictate the preferred orientation of the aldehyde substrate. Placement of the bulky alkyl substituent in the entry chamber allows the carbonyl group to penetrate deeply into the active site and contact Arg101, which can orient and activate it for attack by the nearby enolate. The modelled antiperiplanar arrangement of the substrates rationalizes the observed formation of the S-configured alcohol in the course of the reaction. A synclinal arrangement of aldehyde and enolate is precluded by clashes with the protein backbone. c, Stick representation of the interactions illustrated in b.
The promiscuous MPS-catalysed reaction between oxaloacetate and protected aldoses is both efficient and stereoselective, providing ready access to natural and unnatural derivatives of valuable higher sugars such as KDN and sialic acid. The enzyme's broad substrate tolerance and its use of pyruvate enolate rather than a surrogate provide an attractive complement to existing methods for preparing differentially protected sugars. Such molecules represent unique building blocks for oligosaccharide assembly and their rapid synthesis should facilitate the development of synthetic vaccines and carbohydrate-based antibiotics. Because the stereochemical preferences of MPS favour formation of 4S-configured alcohols, discovery and/or engineering of new enzymes for the Cornforth-type synthesis of 3-deoxysugars like KDO that are R-configured at this position represents a challenge for the future. In this context, design through evolution offers one possible strategy to identify enantiocomplementary MPS mutants15,16.
Methods
Buffers were prepared with ultrapure water. All chemicals were purchased from Sigma-Aldrich, Fluka or Acros and used as received. Macrophomate synthase was produced as previously described1. For MPS-catalysed reactions a buffer of 50 mM NaPi, 5 mM MgCl2 at pH 7.5 was used. The aldol reaction for substrate 8b is described here as an example. Aldehyde 8b (37 mg, 0.14 mmol) was dissolved in a minimum amount of acetonitrile (MeCN) and transferred quantitatively to a 50 ml round-bottom flask (note: in cases where the substrate forms large oil bubbles the yields are low unless the product is removed with a pipette, redissolved in additional MeCN and added again to the reaction mixture). Enough buffer was then added such that the final oxaloacetate concentration (including enzyme solution) was 15 mM (13.1 ml). MPS (925 µl of a 56 µM stock solution to give a 5 µM final [MPS]) was then added by pipette. Oxaloacetate was added in four portions (4 × 6.9 mg, 0.21 mmol) as a solid powder over 4 h, and the reaction was shaken (or stirred slowly) for an additional 1 h. After 5 h in total the mixture was frozen and the water was removed by lyophilization. The crude residue was suspended in methanol and stirred for 15 minutes. The resulting suspension was filtered (with washings) and the methanol was evaporated from the filtrate. 1H NMR of the crude product was used to assess the diastereoselectivity by integrating the C3 methylene proton peaks at 2.88 and 3.08 ppm. The product was then purified by reversed-phase high-performance liquid chromotography under neutral conditions to prevent elimination. A linear gradient of eluant from 5% to 40% CH3CN in water was used and the product 9b (40 mg, 0.11 mmol, 76%) was obtained as a white powder after lyophilization.

References
Serafimov, J. M., Gillingham, D., Kuster, S. & Hilvert, D. The putative Diels-Alderase macrophomate synthase is an efficient aldolase. J. Am. Chem. Soc. 130, 7798–7799 (2008).
Li, L. S. & Wu, Y. L. Recent progress in syntheses of higher 3-deoxy-2-ulosonic acids and their derivatives. Curr. Org. Chem. 7, 447–475 (2003).
Cornforth, J. W., Firth, M. E. & Gottschalk, A. The synthesis of N-acetylneuraminic acid. Biochem. J. 68, 57–61 (1958).
Shirai, R. & Ogura, H. Improved syntheses of two 3-deoxyald-2-ulosonic acids (KDN, KDO) by condensation of oxalacetic acid with aldoses followed by Ni2+ catalyzed decarboxylation. Tetrahedron Lett. 30, 2263–2264 (1989).
Dean, S. M., Greenberg, W. A. & Wong, C. H. Recent advances in aldolase-catalyzed asymmetric synthesis. Adv. Synth. Catal. 349, 1308–1320 (2007).
Sugai, T., Shen, G. J., Ichikawa, Y. & Wong, C. H. Synthesis of 3-deoxy-D-manno-2-octulosonic acid (KDO) and its analogs based on KDO aldolase-catalyzed reactions. J. Am. Chem. Soc. 115, 413–421 (1993).
Williams, G. J., Woodhall, T., Farnsworth, L. M., Nelson, A. & Berry, A. Creation of a pair of stereochemically complementary biocatalysts. J. Am. Chem. Soc. 128, 16238–16247 (2006).
Fong, S., Machajewski, T. D., Mak, C. C. & Wong, C. H. Directed evolution of D-2-keto-3-deoxy-6-phosphogluconate aldolase to new variants for the efficient synthesis of D- and l-sugars. Chem. Biol. 7, 873–883 (2000).
Masamune, S., Choy, W., Petersen, J. S. and Sita, L. R. Double asymmetric synthesis and a new strategy for stereochemical control in organic synthesis. Angew. Chem. Int. Ed. 24, 1–76 (1985).
Weitz, D. J. & Bednarski, M. D. Synthesis of acyclic sugar aldehydes by ozonolysis of oximes. J. Org. Chem. 54, 4957–4959 (1989).
Inoue, S. & Kitajima, K. KDN (deaminated neuraminic acid): Dreamful past and exciting future of the newest member of the sialic acid family. Glycoconjugate J. 23, 277–290 (2006).
Ose, T. et al. Insight into a natural Diels-Alder reaction from the structure of macrophomate synthase. Nature 422, 185–189 (2003).
Denmark, S. E. & Henke, B. R. Investigations on transition-state geometry in the aldol condensation. J. Am. Chem. Soc. 113, 2177–2194 (1991).
Zimmerman, H. E. & Traxler, M. D. The stereochemistry of the Ivanov and Reformatsky reactions. I. J. Am. Chem. Soc. 79, 1920–1923 (1957).
Jäckel, C., Kast, P. & Hilvert, D. Protein design by directed evolution. Ann. Rev. Biophys. 37, 153–173 (2008).
Mugford, P. F., Wagner, U. G., Jiang, Y., Faber, K. & Kazlauskas, R. J. Enantiocomplementary enzymes: Classification, molecular basis for their enantiopreference, and prospects for mirror-image biotransformations. Angew. Chem. Int. Ed. 47, 8782–8793 (2008).
Acknowledgements
This work was supported by the ETH Zürich, a Marie Curie International Incoming Fellowship within the 7th European Community Framework Programme to D.G.G. (IIF-AEOM) and a PhD fellowship from the Studienstiftung des Deutschen Volkes to P.S.
Author information
Authors and Affiliations
Contributions
D.G.G., P.S., P.H.S. and D.H. designed research. D.G.G, P.S. and A.A. performed the experiments. D.G.G., P.S., P.H.S and D.H. analysed data. D.G.G., P.S., P.H.S. and D.H. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 3202 kb)
Rights and permissions
About this article
Cite this article
Gillingham, D., Stallforth, P., Adibekian, A. et al. Chemoenzymatic synthesis of differentially protected 3-deoxysugars. Nature Chem 2, 102–105 (2010). https://doi.org/10.1038/nchem.504
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.504
This article is cited by
-
A novel enzyme-catalyzed synthesis of N-substituted pyrrole derivatives
Molecular Diversity (2013)
-
A Novel Enzymatic Synthesis of Quinoline Derivatives
Catalysis Letters (2012)
-
Sweet flexibility
Nature Chemistry (2010)