Abstract
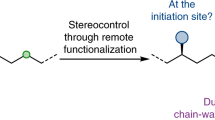
Exploiting the reactivity of one functional group within a molecule to generate a reaction at a different position is an ongoing challenge in organic synthesis. Effective remote functionalization protocols have the potential to provide access to almost any derivatives but are difficult to achieve. The difficulty is more pronounced for acyclic systems where flexible alkyl chains are present between the initiating functional group and the desired reactive centres. In this Review, we discuss the concept of remote functionalization of alkenes using metal complexes, leading to a selective reaction at a position distal to the initial double bond. We aim to show the vast opportunity provided by this growing field through selected and representative examples. Our aim is to demonstrate that using a double bond as a chemical handle, metal-assisted long-distance activation could be used as a powerful synthetic strategy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Breslow, R. Centenary lecture. Biomimetic chemistry. Chem. Soc. Rev. 1, 553–580 (1972).
Breslow, R. Biomimetic control of chemical selectivity. Acc. Chem. Res. 13, 170–177 (1980).
Schwarz, H. Remote functionalization of C-H and C-C bonds by “naked” transition metal ions (Cosi Fan Tutte). Acc. Chem. Res. 22, 282–287 (1989).
Ntambi, J. M. Regulation of stearoyl-CoA desaturase by polyunsaturated fatty acids and cholesterol. J. Lipid. Res. 40, 1549–1558 (1999).
Tang, R.-Y., Li, G. & Yu, J.-Q. Conformation-induced remote meta-C–H activation of amines. Nature 507, 215–220 (2014).
Wang, X.-C & et al. Ligand-enabled meta-C–H activation using a transient mediator. Nature 519, 334–338 (2015).
Dong, Z., Wang, J. & Dong, G. Simple amine-directed meta-selective C–H arylation via Pd/norbornene catalysis. J. Am. Chem. Soc. 137, 5887–5890 (2015).
Hermann, W. A. & Prinz, M. in Applied Homogeneous Catalysis with Organometallic Compounds 2nd edn, 1119–1124 (Wiley-VCH, 2002).
Vilches-Herrera, V., Domke, L. & Börner, A. Isomerization–hydroformylation tandem reactions. ACS Catal. 4, 1706–1724 (2014).
Mei, T.-S, Patel, H. H. & Sigman, M. S. Enantioselective construction of remote quaternary stereocenters. Nature 508, 340–344 (2014).
Crabtree, H. R. The Organometallic Chemistry of the Transition Metals 5th edn, 229–231 (Wiley-VCH, 2009).
Espinet, P. & Albéniz, A. C. in Current Methods in Inorganic Chemistry Vol. 3, 293–372 (Elsevier Science B. V., 2003).
Ittel, S. D., Johnson, L. K. & Brookhart, M. Late-metal catalysts for ethylene homo- and copolymerization. Chem. Rev. 100, 1169–1203 (2000).
Guan, Z. et al. Chain walking: a new strategy to control polymer topology. Science 283, 2059–2062 (1999).
Wilke, G. et al. Allyl-transition metal systems. Angew. Chem. Int. Ed. Engl. 5, 151–164 (1966).
Coffey, R. S. The isomerisation of octenes catalysed by phosphine complexes of iridium(III). Tetrahedron Lett. 6, 3809–38111 (1965).
Grotjahn, D. B. Heteroatoms moving protons: synthetic and mechanistic studies of bifunctional organometallic catalysis. Pure Appl. Chem. 82, 635–647 (2010).
Bair, J. S. et al. Linear-selective hydroarylation of unactivated terminal and internal olefins with trifluoromethyl-substituted arenes. J. Am. Chem. Soc. 136, 13098–13010 (2014).
Tao, J., Sun, F. & Fang, T. Mechanism of alkene isomerization by bifunctional ruthenium catalyst: A theoretical study. J. Organomet. Chem. 698, 1–6 (2012).
Sen, A. & Lai, T.-W. Catalytic isomerization of alkenes by palladium(II) compounds. An alternative mechanistic view. Inorg. Chem. 20, 4036–4038 (1981).
Gooßen, L. J., Ohlmann, D. M. & Dierker, M. Silver triflate-catalysed synthesis of γ-lactones from fatty acids. Green Chem. 12, 197–200 (2010).
Fan, J. et al. Palladium catalyzed isomerization of alkenes: a pronounced influence of an o-phenyl hydroxyl group. Org. Biomol. Chem. 7, 3168–31722 (2009).
Larionov, E., Li, H. & Mazet, C. Well-defined transition metal hydrides in catalytic isomerizations. Chem. Commun. 50, 9816–9826 (2014).
Cornils, B., Herrmann, W. A. & Rasch, M. Otto Roelen, pioneer in industrial homogeneous catalysis. Angew. Chem. Int. Ed. Engl. 33, 2144–2163 (1994).
Behr, A. et al. Highly selective tandem isomerization–hydroformylation reaction of trans-4-octene to n-nonanal with rhodium-BIPHEPHOS catalysis. J. Mol. Catal. A Chem. 206, 179–184 (2003).
Morrill, T. C. & D'Souza, C. A. Efficient hydride-assisted isomerization of alkenes via rhodium catalysis. Organometallics 22, 1626–1629 (2003).
Wu, L. et al. Ruthenium-catalyzed hydroformylation/reduction of olefins to alcohols: extending the scope to internal alkenes. J. Am. Chem. Soc. 135, 14306–14312 (2013).
Yuki, Y. et al. Tandem isomerization/hydroformylation/hydrogenation of internal alkenes to n-alcohols using Rh/Ru dual- or ternary-catalyst systems. J. Am. Chem. Soc. 135, 17393–17400 (2013).
Pugh, R. I., Drent, E. & Pringle, P. G. Tandem isomerisation–carbonylation catalysis: highly active palladium(II) catalysts for the selective methoxycarbonylation of internal alkenes to linear esters. Chem. Commun. 2001, 1476–1477 (2001).
Jimenez Rodriguez, C. et al. Highly selective formation of linear esters from terminal and internal alkenes catalysed by palladium complexes of bis-(di-tert-butylphosphinomethyl)benzene. Chem. Commun. 2004, 1720–1721 (2004).
Moballigh, A. et al. Highly selective hydroaminomethylation of internal alkenes to give linear amines. Chem. Eur. J. 12, 8979–8988 (2006).
Seayad, A. et al. Internal olefins to linear amines. Science 297, 1676–1678 (2002).
Vaultier, M. & Carboni, B. Comprehensive organometallic chemistry II (eds Abel, E. W., Stone, F. G. A. & Wilkinson, G.) (Oxford Press, 1995).
Burgess, K. & Ohlmeyer, M. J. Transition-metal-promoted hydroborations of alkenes, emerging methodology for organic transformations. Chem. Rev. 91, 1179–1191 (1991).
Pereira, S. & Srebnik, M. A study of hydroboration of alkenes and alkynes with pinacolborane catalyzed by transition metals. Tetrahedron Lett. 37, 3283–3286 (1996).
Lata, C. J. & Crudden, C. M. Dramatic effect of Lewis acids on the rhodium-catalyzed hydroboration of olefins. J. Am. Chem. Soc. 132, 131–137 (2010).
Evans, D. A., Fu, G. C. & Anderson, B. A. Mechanistic study of the rhodium(I)-catalyzed hydroboration reaction. J. Am. Chem. Soc. 114, 6679–6685 (1992).
Pereira, S. & Srebnik, M. Transition metal-catalyzed hydroboration of and CCl4 addition to alkenes. J. Am. Chem. Soc. 118, 909–910 (1996).
Edwards, D. R., Crudden, C. M. & Yam, K. One-pot carbon monoxide-free hydroformylation of internal olefins to terminal aldehydes. Adv. Synth. Catal. 347, 50–54 (2005).
Yamamoto, Y. et al. Iridium-catalyzed hydroboration of alkenes with pinacolborane. Tetrahedron 60, 10695–10700 (2004).
Obligacion, J. V. & Chirik, P. J. Highly selective bis(imino)pyridine iron-catalyzed alkene hydroboration. Org. Lett. 15, 2680–2683 (2013).
Obligacion, J. V. & Chirik, P. J. Bis(imino)pyridine cobalt catalyzed alkene isomerization-hydroboration: a strategy for remote hydrofunctionalization with terminal selectivity. J. Am. Chem. Soc. 135, 19107–19110 (2013).
Palmer, W. N. et al. High-activity cobalt catalysts for alkene hydroboration with electronically responsive terpyridine and α-diimine ligands. ACS Catal. 5, 622–626 (2015).
Ruddy, A. J. et al. (N-Phosphinoamidinate)cobalt-catalyzed hydroboration: alkene isomerization affords terminal selectivity. Chem. Eur. J. 20, 13918–13922 (2014).
Troegel, D. & Stohrer, J. Recent advances and actual challenges in late transition metal catalyzed hydrosilylation of olefins from an industrial point of view. Coord. Chem. Rev. 255, 1440–1459 (2011).
Marciniec, B. (ed.) Hydrosilylation. A Comprehensive Review on Recent Advances (Springer, 2009).
Saam, J. C. & Speier, J. L. The addition of silicon hydrides to olefinic double bonds. Part III. The addition to non-terminal olefins in the presence of chloroplatinic acid. J. Am. Chem. Soc. 80, 4104–4106 (1958).
Atienza, C. C. H. et al. Bis(imino)pyridine cobalt-catalyzed dehydrogenative silylation of alkenes: scope, mechanism, and origins of selective allylsilane formation. J. Am. Chem. Soc. 136, 12108–12118 (2014).
Lipshutz, B. H., Pfeiffer, S. S., Noson, K. & Tomioka, T. in Titanium and Zirconium in Organic Synthesis (ed. Marek, I.) Ch. 4, 110–148 (2002).
Marek, I. Chinkov, N. & Levin, A. A zirconium promenade – an efficient tool in organic synthesis. Synlett 2006, 501–514 (2006).
Annby, U. et al. Hydrozirconation isomerization-reactions of terminally functionalized olefins with zirconocene hydrides and general aspects. Acta Chem. Scand. 47, 425–433 (1993).
Gibson, T. & Tulich, L. Novel synthesis of long-chain primary alkyl compounds. J. Org. Chem. 46, 1821–1823 (1981).
Wipf, P. & Jahn, H. Synthetic applications of organochlorozirconocene complexes. Tetrahedron 52, 12853–12910 (1996).
Wipf, P. & Xu, W. Transmetalation reactions of organozirconocenes: a general, selective, and facile synthesis of ketones from acid chlorides. Synlett 1992, 718–721 (1992).
Wipf, P. & Smitrovich, J. H. Transmetalation reactions of alkylzirconocenes: copper-catalyzed conjugate addition to enones. J. Org. Chem. 56, 6494–6496 (1991).
Mola, L., Sidera, M. & Fletcher, S. P. Asymmetric remote C–H functionalization: use of internal olefins in tandem hydrometallation–isomerization–asymmetric conjugate addition sequences. Aust. J. Chem. 68, 401–403 (2015).
Uma, R., Crévisy, C. & Grée, R. Transposition of allylic alcohols into carbonyl compounds mediated by transition metal complexes. Chem. Rev. 103, 27–51 (2003).
Felfödi, K. & Bartók, M. Transformation of organic compounds in the presence of metal complexes. I. Transformations of unsaturated alcohols with metal complex catalysts. J. Organomet. Chem. 297, c37–c40 (1985).
Kisanga, P., Goj, L. A. & Widenhoefer, R. A. Cycloisomerization of functionalized 1, 5- and 1,6-dienes catalyzed by cationic palladium phenanthroline complexes. J. Org. Chem. 66, 635–637 (2001).
Goj, L. A. & Widenhoefer, R. A. Mechanistic studies of the cycloisomerization of dimethyl diallylmalonate catalyzed by a cationic palladium phenanthroline complex. J. Am. Chem. Soc. 123, 11133–11147 (2001).
Larionov, E. et al. Scope and mechanism in palladium-catalyzed isomerizations of highly substituted allylic, homoallylic and alkenyl alcohols. J. Am. Chem. Soc. 136, 16882–16894 (2014).
Kochi, T. et al. Chain-walking strategy for organic synthesis: catalytic cycloisomerization of 1,n-dienes. J. Am. Chem. Soc. 134, 16544–16547 (2012).
Yamamoto, Y. Transition-metal-catalyzed cycloisomerizations of α,ω-dienes. Chem. Rev. 112, 4736–4769 (2012).
Wakamatsu, H. et al. Isomerization reaction of olefin using RuClH(CO)(Ph3)3 . J. Org. Chem. 65, 3966–3970 (2000).
Doi, T. et al. RuHCl(CO)(PPh3)3-catalyzed chemoselective transfer-hydrogenation of enones leading to saturated ketones. Synlett 2006, 721–724 (2006).
Doi, T. et al. An unusual dimerization of primary unsaturated alcohols catalyzed by RuHCl(CO)(PPh3)3 . Chem. Commun. 2006, 1875–1877 (2006).
Fukuyama, T. et al. Ruthenium hydride catalyzed regioselective addition of aldehydes to enones to give 1,3-diketones. Angew. Chem. Int. Ed. 46, 5559–5561 (2007).
Molpolder, J. B. & Heck, R. F. A palladium-catalyzed arylation of allylic alcohols with aryl halides. J. Org. Chem. 41, 265–272 (1976).
Chalk, A. J. & Magennis, S. A. Palladium-catalyzed vinyl substitution reactions. II. Synthesis of aryl substituted allylic alcohols, aldehydes, and ketones from aryl halides and unsaturated alcohols. J. Org. Chem. 41, 1206–1209 (1976).
Tamaru, Y., Yamada, Y. & Yoshida, Z.-I. The palladium catalyzed thienylation of allylic alcohols with 2-bromothiophenes and their derivatives. Tetrahedron 35, 329–340 (1979).
Larock, R. C., Leung, W.-Y. & Stoltz-Dunn, S. Synthesis of aryl-substituted aldehydes and ketones via palladium-catalyzed coupling of aryl halides and non-allylic unsaturated alcohols. Tetrahedron Lett. 30, 6629–6632 (1989).
Larock, R. C. et al. Synthesis of aryl-substituted allylic amines via palladium-catalyzed coupling of aryl iodides, non-conjugated dienes, and amines. J. Org. Chem. 59, 8107–8114 (1994).
Larock, R. C., Lu, Y. & Bain, A. C. Palladium-catalyzed coupling of aryl iodides, nonconjugated dienes, and carbon nucleophiles by palladium migration. J. Org. Chem. 56, 4589–4590 (1991).
Wang, Y., Dong, X. & Larock, R. C. Synthesis of naturally occurring pyridine alkaloids via palladium-catalyzed coupling/migration chemistry. J. Org. Chem. 68, 3090–3098 (2003).
Werner, E. W. et al. Enantioselective Heck arylations of acyclic alkenyl alcohols using a redox-relay strategy. Science 338, 1455–1458 (2012).
Patel, H. H. & Sigman, M. S. Palladium-catalyzed enantioselective Heck alkenylation of acyclic alkenols using a redox-relay strategy. J. Am. Chem. Soc. 137, 3462–3465 (2015).
Mei, T.-S. et al. Enantioselective redox-relay oxidative Heck arylations of acyclic alkenyl alcohols using boronic acids. J. Am. Chem. Soc. 135, 6830–6833 (2013).
Dang, Y. et al. A computational mechanistic study of an unprecedented Heck-type relay reaction: insight into the origins of regio- and enantioselectivities. J. Am. Chem. Soc. 136, 986–988 (2014).
Hilton, M. J. et al. Investigating the nature of palladium chain-walking in the enantioselective redox-relay Heck reaction of alkenyl alcohols. J. Org. Chem. 79, 11841–11850 (2014).
Xu, L. et al. Mechanism, reactivity and selectivity in palladium-catalyzed redox-relay Heck arylation of alkenyl alcohols. J. Am. Chem. Soc. 136, 1960–1967 (2014).
Grotjahn, D. B. et al. Extensive isomerization of alkenes using a bifunctional catalyst: an alkene zipper. J. Am. Chem. Soc. 129, 9592–9593 (2007).
Erdogan, G. & Grotjahn, D. B. Mild and selective deuteration and isomerization of alkenes by a bifunctional catalyst and deuterium oxide. J. Am. Chem. Soc. 131, 10354–10355 (2009).
Chinkov, N., Levin, A. & Marek, I. Unsaturated fatty alcohol derivatives as a source of substituted allylzirconocene. Angew. Chem. Int. Ed. 45, 465–468 (2006).
Negishi, E.-I., Cederbaum, F. E. & Takahashi, T. Reaction of zirconocene dichloride with alkyllithiums or alkyl Grignard reagents as a convenient method for generating a “zirconocene” equivalent and its use in zirconium-promoted cyclization of alkenes, alkynes, dienes, enynes, and diynes. Tetrahedon Lett. 27, 2829–2832 (1986).
Chinkov, N., Majumdar, S. & Marek, I. Stereoselective preparation of dienyl zirconocene complexes via a tandem allylic C–H bond activation-elimination sequence. J. Am. Chem. Soc. 125, 13258–13264 (2003).
Miura, T. et al. Enantioselective synthesis of anti homoallylic alcohols from terminal alkynes and aldehydes based on concomitant use of cationic iridium complex and a chiral phosphoric acid. J. Am. Chem. Soc. 135, 11497–11500 (2013).
Ohlmann, D. M., Gooβen, L. J. & Dierker, M. Regioselective synthesis of β-aryl- and β-amino-substituted aliphatic esters by rhodium-catalyzed tandem double-bond migration/conjugate addition. Chem. Eur. J. 17, 9508–9519 (2011).
Rybtchinski, B. & Milstein, D. Metal insertion into C–C bonds in solution. Angew. Chem. Int. Ed. 38, 870–883 (1999).
Ruhland, K. Transition-metal-mediated cleavage and activation of C–C single bonds. Eur. J. Org. Chem. 2012, 2683–2706 (2012).
Marek, I. et al. Selective carbon–carbon bond cleavage for the stereoselective synthesis of acyclic systems. Angew. Chem. Int. Ed. 54, 414–429 (2015).
Porri, L. et al. Catalysts derived from ruthenium and iridium for the ring-opening polymerization of cycloolefins. Makromol. Chem. 176, 3121–3125 (1975).
France, M. B., Feldman, J. & Grubbs, R. H. An iridium-based catalyst system for metathesis/isomerization of acyclic olefins including methyl oleate. J. Chem. Soc. Chem. Commun. 1994, 1307–1308 (1994).
Consorti, C. S., Aydos, G. L. P. & Dupont, J. Tandem isomerization–metathesis catalytic processes of linear olefins in ionic liquid biphasic system. Chem. Commun. 46, 9058–9060 (2010).
Ohlmann, D. M. et al. Isomerizing olefin metathesis as a strategy to access defined distributions of unsaturated compounds from fatty acids. J. Am. Chem. Soc. 134, 13716–13729 (2012).
Dobereiner, G. E. et al. A one-pot tandem olefin isomerization/metathesis-coupling (ISOMET) reaction. ACS Catal. 4, 3069–3076 (2014).
Baader, S. et al. Synthesis of tsetse fly attractants from a cashew nut shell extract by isomerising metathesis. Green Chem. 16, 4885–4890 (2014).
Masarwa, A. et al. Merging allylic C–H activation and selective C–C bond activation. Nature 505, 199–203 (2014).
Vasseur, A. et al. Remote functionalization of hydrocarbons with reversibility enhanced stereocontrol. Chem. Sci. 6, 2770–2776 (2015).
Acknowledgements
This research was supported by the European Research Council under the Seventh Framework Program of the European Community (ERC grant agreement no 338912). I.M. holds the Sir Michael and Lady Sobell Academic Chair.
Author information
Authors and Affiliations
Contributions
A.V., J.B. and I.M. contributed to discussions and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Vasseur, A., Bruffaerts, J. & Marek, I. Remote functionalization through alkene isomerization. Nature Chem 8, 209–219 (2016). https://doi.org/10.1038/nchem.2445
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.2445
This article is cited by
-
Stereodivergent 1,3-difunctionalization of alkenes by charge relocation
Nature (2024)
-
Alkene 1,1-difunctionalizations via organometallic-radical relay
Nature Catalysis (2023)
-
Nickel-catalyzed direct stereoselective α-allylation of ketones with non-conjugated dienes
Nature Communications (2023)
-
Multi-site programmable functionalization of alkenes via controllable alkene isomerization
Nature Chemistry (2023)
-
Palladium-catalyzed regiodivergent hydrochlorocarbonylation of alkenes for formation of acid chlorides
Nature Communications (2023)