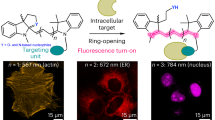
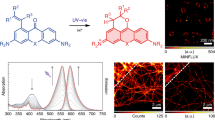
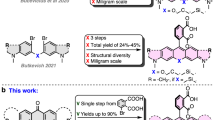
Abstract
Single-molecule fluorescence techniques have emerged as a powerful approach to understanding complex biological systems. However, a challenge researchers still face is the limited photostability of nearly all organic fluorophores, including the cyanine and Alexa dyes. We report a new, monovalent probe that emits in the far-red region of the visible spectrum with properties desirable for single-molecule optical imaging. This probe is based on a ring-fused boron-dipyrromethene (BODIPY) core that is conjugated to a polyglycerol dendrimer (PGD). The dendrimer makes the hydrophobic fluorophore water-soluble. This probe exhibits excellent brightness, with an emission maximum of 705 nm. We have observed strikingly long and stable emission from individual PGD–BODIPY probes, even in the absence of anti-fading agents such as Trolox, a combined oxidizing–reducing agent often used in single-molecule studies for improving the photostability of common imaging probes. These interesting properties greatly simplify use of the fluorophore.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Giepmans, B. N. G., Adams, S. R., Ellisman, M. H. & Tsien, R. Y. The fluorescent toolbox for assessing protein location and function. Science 312, 217–224 (2006).
Lavis, L. D. & Raines, R. T. Bright ideas for chemical biology. ACS Chem. Biol. 3, 142–155 (2008).
Kobayashi, H., Ogawa, M., Alford, R., Choyke, P. L. & Urano, Y. New strategies for fluorescent probe design in medical diagnostic imaging. Chem. Rev. 110, 2620–2640 (2010).
Cordes, T., Vogelsang, J. & Tinnefeld, P. On the mechanism of trolox as antiblinking and antibleaching reagent. J. Am. Chem. Soc. 131, 5018–5019 (2009).
Berezin, M. Y. & Achilefu, S. Fluorescence lifetime measurements and biological imaging. Chem. Rev. 110, 2641–2684 (2010).
Mujumdar, R. B., Ernst, L. A., Mujumdar, S. R., Lewis, C. J. & Waggoner, A. S. Cyanine dye labeling reagents: sulfoindocyanine succinimidyl esters. Bioconj. Chem. 4, 105–111 (1993).
Funatsu, T., Harada, Y., Tokunaga, M., Saito, K. & Yanagida, T. Imaging of single fluorescent molecules and individual ATP turnovers by single myosin molecules in aqueous solution. Nature 374, 555–559 (1995).
Heilemann, M., Margeat, E., Kasper, R., Sauer, M. & Tinnefeld, P. Carbocyanine dyes as efficient reversible single-molecule optical switch. J. Am. Chem. Soc. 127, 3801–3806 (2005).
Roy, R., Hohng, S. & Ha, T. A practical guide to single-molecule FRET. Nature Methods 5, 507–516 (2008).
Weiss, S. Fluorescence spectroscopy of single biomolecules. Science 283, 1676–1683 (1999).
Lu, H. P., Xun, L. & Xie, X. S. Single-molecule enzymatic dynamics. Science 282, 1877–1882 (1998).
Moerner, W. E. & Fromm, D. P. Methods of single-molecule fluorescence spectroscopy and microscopy. Rev. Sci. Instrum. 74, 3597–3619 (2003).
Ha, T. Single-molecule fluorescence resonance energy transfer. Methods 25, 78–86 (2001).
Ha, T. & Tinnefeld, P. Photophysics of fluorescent probes for single-molecule biophysics and super-resolution imaging. Annu. Rev. Phys. Chem. 63, 595–617 (2012).
Giloh, H. & Sedat, J. W. Fluorescence microscopy: reduced photobleaching of rhodamine and fluorescein protein conjugates by n-propyl gallate. Science 217, 1252–1255 (1982).
Lichtman, J. W. & Conchello, J-A. Fluorescence microscopy. Nature Methods 2, 910–919 (2005).
Rasnik, I., McKinney, S. A. & Ha, T. Nonblinking and long-lasting single-molecule fluorescence imaging. Nature Methods 3, 891–893 (2006).
Vogelsang, J. et al. A reducing and oxidizing system minimizes photobleaching and blinking of fluorescent dyes. Angew. Chem. Int. Ed. 47, 5465–5469 (2008).
Dave, R., Terry, D. S., Munro, J. B. & Blanchard, S. C. Mitigating unwanted photophysical processes for improved single-molecule fluorescence imaging. Biophys. J. 96, 2371–2381 (2009).
Altman, R. B. et al. Cyanine fluorophore derivatives with enhanced photostability. Nature Methods 9, 68–71 (2012).
Loudet, A. & Burgess, K. BODIPY dyes and their derivatives: syntheses and spectroscopic properties. Chem. Rev. 107, 4891–4932 (2007).
Ulrich, G., Ziessel, R. & Harriman, A. The chemistry of fluorescent bodipy dyes: versatility unsurpassed. Angew. Chem. Int. Ed. 47, 1184–1201 (2008).
Benniston, A. C. & Copley, G. Lighting the way ahead with boron dipyrromethene (Bodipy) dyes. Phys. Chem. Chem. Phys. 11, 4124–4131 (2009).
Le Gall, A. et al. Improved photon yield from a green dye with a reducing and oxidizing system. ChemPhysChem 12, 1657–1660 (2011).
Johnson, I. & Spence, M. T. Z. The Handbook: A Guide to Fluorescent Probes and Labeling Technologies 11th edn (Molecular Probes, 2010).
Hinkeldey, B., Schmitt, A., & Jung, G. Comparative photostability studies of BODIPY and fluorescein dyes by using fluorescence correlation spectroscopy. ChemPhysChem 9, 2019–2027 (2008).
Chen, J., Burghart, A., Derecskei-Kovacs, A. & Burgess, K. 4,4-Difluoro-4-bora-3a,4a-diaza-s-indacene (BODIPY) dyes modified for extended conjugation and restricted bond rotations. J. Org. Chem. 65, 2900–2906 (2000).
Zhao, W. & Carreira, E. M. Conformationally restricted aza-bodipy: a highly fluorescent, stable, near-infrared-absorbing dye. Angew. Chem. Int. Ed. 44, 1677–1679 (2005).
Ulrich, G., Goeb, S., De Nicola, A., Retailleau, P. & Ziessel, R. Synthesis of bisisoindolomethene dyes bearing anisole or ethylthiophene residues for red and near-IR fluorescence. Synlett 2007, 1517–1520 (2007).
Atilgan, S., Ozdemir, T. & Akkaya, E. U. A sensitive and selective ratiometric near IR fluorescent probe for zinc ions based on the distyryl–bodipy fluorophore. Org. Lett. 10, 4065–4067 (2008).
Zeng, L. et al. Anthracene-fused BODIPYs as near-infrared dyes with high photostability. Org. Lett. 13, 6026–6029 (2011).
Umezawa, K., Nakamura, Y., Makino, H., Citterio, D. & Suzuki, K. Bright, color-tunable fluorescent dyes in the visible–near-infrared region. J. Am. Chem. Soc. 130, 1550–1551 (2008).
Umezawa, K., Matsui, A., Nakamura, Y., Citterio, D. & Suzuki, K. Bright, color-tunable fluorescent dyes in the Vis/NIR region: establishment of new ‘tailor-made’ multicolor fluorophores based on borondipyrromethene. Chem. Eur. J. 15, 1096–1106 (2008).
Atilgan, S., Ekmekci, Z., Dogan, A. L., Guc, D. & Akkaya, E. U. Water soluble distyryl-boradiazaindacenes as efficient photosensitizers for photodynamic therapy. Chem. Commun. 4398–4400 (2006).
Li, L., Han, J., Nguyen, B. & Burgess, K. Syntheses and spectral properties of functionalized, water-soluble BODIPY derivatives. J. Org. Chem. 73, 1963–1970 (2008).
Komatsu, T. et al. Development of 2,6-carboxy-substituted boron dipyrromethene (BODIPY) as a novel scaffold of ratiometric fluorescent probes for live cell imaging. Chem. Commun. 7015–7017 (2009).
Niu, S. L. et al. Water-soluble BODIPY derivatives. Org. Lett. 11, 2049–2052 (2009).
Bura, T. & Ziessel, R. Water-soluble phosphonate-substituted BODIPY derivatives with tunable emission channels. Org. Lett. 13, 3072–3075 (2011).
Yang, S. K. et al. Monovalent, clickable, uncharged, water-soluble perylenediimide-cored dendrimers for target-specific fluorescent biolabeling. J. Am. Chem. Soc. 133, 9964–9967 (2011).
Heek, T. et al. Highly fluorescent water-soluble polyglycerol-dendronized perylene bisimide dyes. Chem. Commun. 46, 1884–1886 (2010).
Schlick, K. H., Morgan, J. R., Weiel, J. J., Kelsey, M. S. & Cloninger, M. J. Clusters of ligands on dendrimer surfaces. Bioorg. Med. Chem. Lett. 21, 5078–5083 (2011).
Elmer, S. L., Man, S. & Zimmerman, S. C. Synthesis of polyglycerol, porphyrin-cored dendrimers using click chemistry. Eur. J. Org. Chem. 2008, 3845–3851 (2008).
Kolb, H. C., Finn, M. G. & Sharpless, K. B. Click chemistry: diverse chemical function from a few good reactions. Angew. Chem. Int. Ed. 40, 2004–2021 (2001).
Zill, A. T. et al. Clickable polyglycerol hyperbranched polymers and their application to gold nanoparticles and acid-labile nanocarriers. Chem. Commun. 47, 1279–1281 (2011).
Shi, X., Lim, J. & Ha, T. Acidification of the oxygen scavenging system in single-molecule fluorescence studies: in situ sensing with a ratiometric dual-emission probe. Anal. Chem. 82, 6132–6138 (2010).
Shi, X. et al. Quantitative fluorescence labeling of aldehyde-tagged proteins for single-molecule imaging. Nature Methods 9, 499–503 (2012).
Acknowledgements
The authors acknowledge the National Institutes of Health (GM087448 to S.C.Z., GM065367 and AI083025 to T.H.) and the National Science Foundation (PHY-0822613) for financial support.
Author information
Authors and Affiliations
Contributions
S.K.Y. and S.C.Z. conceived the study. S.K.Y. designed and synthesized the polyglycerol-dendronized fluorophores. X.S. performed the single-molecule experiments. X.S. and S.P. performed the microtubule labelling experiments. S.K.Y., X.S., T.H. and S.C.Z. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 2226 kb)
Supplementary Movie
Supplementary Movie (MOV 4396 kb)
Rights and permissions
About this article
Cite this article
Yang, S., Shi, X., Park, S. et al. A dendritic single-molecule fluorescent probe that is monovalent, photostable and minimally blinking. Nature Chem 5, 692–697 (2013). https://doi.org/10.1038/nchem.1706
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1706
This article is cited by
-
Synthesis, oligonucleotide incorporation and fluorescence properties in DNA of a bicyclic thymine analogue
Scientific Reports (2018)
-
Collaborative routes to clarifying the murky waters of aqueous supramolecular chemistry
Nature Chemistry (2018)
-
Photostable and photoswitching fluorescent dyes for super-resolution imaging
JBIC Journal of Biological Inorganic Chemistry (2017)
-
A simple and versatile design concept for fluorophore derivatives with intramolecular photostabilization
Nature Communications (2016)
-
Probes: paths to photostability
Nature Methods (2015)