Abstract
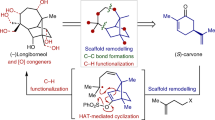
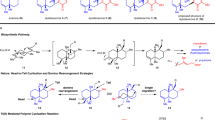
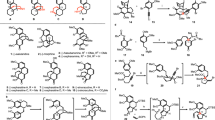
Polyprenylated polycyclic acylphloroglucines (PPAPs) are a family of natural products that possess a wide range of different important biological activities because of the relative position and configuration of four substituents that decorate one common central bicyclo[3.3.1]nonane-2,4,9-trione core. The rigid bicyclic framework with its lipophilic side chains and its hydrophilic trione moiety represents a nature-derived lead structure that arranges the substituents (R1 to R4) into a defined topographical orientation. As the substituents are responsible for the biological activities, the seven-step synthetic approach presented here sets the stage for an iterative introduction of R1 to R4 and thus generates structurally diverse trans-type B PPAPs. Four natural and one non-natural trans-type B PPAPs were prepared starting from acetylacetone with overall yields that ranged from 6 to 22%. The concept of separating framework construction from decorating transformations plus the minimization of protecting-group operations are the key issues for the realization of our synthetic approach.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ciochina, R. & Grossman, R. B. Polycyclic polyprenylated acylphloroglucinols. Chem. Rev. 106, 3963–3986 (2006).
McCandlish, L. E., Hanson, J. C. & Stout, G. H. The structures of two derivatives of bicyclo[3.3.1]nonane-2,4,9-trione. A natural product: clusianone, C33H42O4, and trimethylated catechinic acid, C18H20O6 . Acta Crystallogr. B 32, 1793–1801 (1976).
Ito, C. et al. Polyprenylated benzophenones from Garcinia assigu and their potential cancer chemopreventive activities. J. Nat. Prod. 66, 206–209 (2003).
Piccinelli, A. L. et al. Structural revision of clusianone and 7-epi-clusianone and anti-HIV activity of polyisoprenylated benzophenones. Tetrahedron 61, 8206–8211 (2005).
Santos, M. H., Nagem, N. L. & De Oliveira, T. T. Epiclusianone: a new natural product derivative of bicyclo[3.3.1]nonane-2,4,9-trione. Acta Crystallogr. C 54, 1990–1992 (1998).
Murata, R. M. et al. Antiproliferative effect of benzophenones and their influence on cathepsin activity. Phytother. Res. 24, 379–383 (2010).
Neves, J. S. et al. Antianaphylactic properties of 7-epiclusianone, a tetraprenylated benzophenone isolated from Garcinia brasiliensis. Planta Med. 73, 644–649 (2007).
Cruz, A. J., Lemos, V. S., dos Santos, M. H., Nagem, T. J. & Cortes, S. F. Vascular effects of 7-epiclusianone, a prenylated benzophenone from Rheedia gardneriana, on the rat aorta. Phytomedicine 13, 442–445 (2006).
Pereira, I. O. et al. Leishmanicidal activity of benzophenones and extracts from Garcinia brasiliensis Mart. fruits. Phytomedicine 17, 339–345 (2010).
Almeida, L. S. B. et al. Antimicrobial activity of Rheedia brasiliensis and 7-epiclusianone against Streptococcus mutans. Phytomedicine 15, 886–891 (2008).
Alves, T. M. et al. Biological activities of 7-epiclusianone. J. Nat. Prod. 62, 369–371 (1999).
Martins, F. T. et al. Natural polyprenylated benzophenones inhibiting cysteine and serine proteases. Eur. J. Med. Chem. 44, 1230–1239 (2009).
Hamed, W. et al. Oblongifolins A–D, polyprenylated benzoylphloroglucinol derivatives from Garcinia oblongifolia. J. Nat. Prod. 69, 774–777 (2006).
Tanaka, N. et al. Prenylated benzophenones and xanthones from Hypericum scabrum. J. Nat. Prod. 67, 1870–1875 (2004).
Winkelmann, K., Heilmann, J., Zerbe, O., Rali, T. & Sticher, O. New prenylated bi- and tricyclic phloroglucinol derivatives from Hypericum papuanum. J. Nat. Prod. 64, 701–706 (2001).
Kuramochi, A., Usuda, H., Yamatsugu, K., Kanai, M. & Shibasaki, M. Total synthesis of (±)-garsubellin A. J. Am. Chem. Soc. 127, 14200–14201 (2005).
Siegel, D. R. & Danishefsky, S. J. Total synthesis of garsubellin A. J. Am. Chem. Soc. 128, 1048–1049 (2006).
Rodeschini, V., Ahmad, N. M. & Simpkins, N. S. Synthesis of (+/–)-clusianone: high-yielding bridgehead and diketone substitutions by regioselective lithiation of enol ether derivatives of bicyclo[3.3.1]nonane-2,4,9-triones. Org. Lett. 8, 5283–5285 (2006).
Qi, J. & Porco, J. A. Rapid access to polyprenylated phloroglucinols via alkylative dearomatization–annulation: total synthesis of (+/–)-clusianone. J. Am. Chem. Soc. 129, 12682–12683 (2007).
Tsukano, C., Siegel, D. R. & Danishefsky, S. J. Differentiation of nonconventional ‘carbanions’ – the total synthesis of nemorosone and clusianone. Angew. Chem. Int. Ed. 46, 8840–8844 (2007).
Nuhant, P., David, M., Pouplin, T., Delpech, B. & Marazano, C. α,α′-Annulation of 2,6-prenyl-substituted cyclohexanone derivatives with malonyl chloride: application to a short synthesis of (±)-clusianone. Formation and rearrangement of a biogenetic-like intermediate. Org. Lett. 9, 287–289 (2007).
Shimizu, Y., Shi, S., Usuda, H., Kanai, M. & Shibasaki, M. Catalytic asymmetric total synthesis of ent-hyperforin. Angew. Chem. Int. Ed. 49, 1103–1106 (2010).
Simpkins, N. S., Taylor, J. D., Weller, M. D. & Hayes, C. J. Synthesis of nemorosone via a difficult bridgehead substitution reaction. Synlett 4, 639–643 (2010).
Garnsey, M. R., Lim, D., Yost, J. M. & Coltart, D. M. Development of a strategy for the asymmetric synthesis of polycyclic polyprenylated acylphloroglucinols via N-amino cyclic carbamate hydrazones: application to the total synthesis of (+)-clusianone. Org. Lett. 12, 5234–5237 (2010).
Zhang, Q., Mitasev, B., Qi, J. & Porco, J. A. Jr Total synthesis of plukenetione A. J. Am. Chem. Soc. 132, 14212–14215 (2010).
Fleming, I. & Lee, D. A synthesis of (±)-lavandulol using a silyl-to-hydroxy conversion in the presence of 1,1-disubstituted and trisubstituted double bonds. J. Chem. Soc. Perkin Trans. 1 17, 2701–2710 (1998).
Qi, J., Beeler, A. B., Zhang, Q. & Porco, J. A. Jr Catalytic enantioselective alkylative dearomatization–annulation: total synthesis and absolute configuration assignment of hyperibone K. J. Am. Chem. Soc. 132, 13642–13644 (2010).
Huckin, S. N. & Weiler, L. C-Acetylation of ketones. Can. J. Chem. 52, 1379–1380 (1974).
Amat, M., Llor, N., Checa, B., Molins, E. & Bosch, J. A synthetic approach to ervatamine-silicine alkaloids. Enantioselective total synthesis of (−)-16-episilicine. J. Org. Chem. 75, 178–189 (2010).
Plietker, B. A highly regioselective salt-free iron-catalyzed allylic alkylation. Angew. Chem. Int. Ed. 45, 1469–1473 (2006).
Plietker, B., Dieskau, A., Möws, K. & Jatsch, A. Ligand dependant mechanistic dichotomy in iron-catalyzed allylic substitutions – σ-allyl- vs. π-allyl mechanism. Angew. Chem. Int. Ed. 47, 198–201 (2008).
Holzwarth, M., Dieskau, A., Tabassam, M. & Plietker, B. Preformed π-allyl iron complexes as potent, well-defined catalysts for the allylic substitution. Angew. Chem. Int. Ed. 48, 7251–7255 (2009).
Acknowledgements
Dedicated to Barry M. Trost on the occasion of his 70th birthday. The authors thank the Deutsche Forschungsgemeinschaft, the Deutsche Krebshilfe e.V., the Landesgraduiertenstiftung Baden-Württemberg (PhD grant for N.B.) and the Studienstiftung des deutschen Volkes (PhD grant for K.M.) for financial support.
Author information
Authors and Affiliations
Contributions
N.B. prepared the natural products 1 – 5 . K.M. was involved in model studies towards the synthesis of O-methyl hyperibone and crystallized this compound (see Supplementary Information). B.P. designed the study, analysed the data and wrote the paper. All the authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 1036 kb)
Supplementary information
Crystallographic data for compound 21 (CIF 23 kb)
Rights and permissions
About this article
Cite this article
Biber, N., Möws, K. & Plietker, B. The total synthesis of hyperpapuanone, hyperibone L, epi-clusianone and oblongifolin A. Nature Chem 3, 938–942 (2011). https://doi.org/10.1038/nchem.1170
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchem.1170
This article is cited by
-
Anticancer Properties and Mechanism of Action of Oblongifolin C, Guttiferone K and Related Polyprenylated Acylphloroglucinols
Natural Products and Bioprospecting (2021)
-
Characteristic metabolites of Hypericum plants: their chemical structures and biological activities
Journal of Natural Medicines (2021)
-
Programmable meroterpene synthesis
Nature Communications (2020)
-
Aroyl and acyl cyanides as orthogonal protecting groups or as building blocks for the synthesis of heterocycles
Molecular Diversity (2019)