Abstract
Genome maintenance defects cause complex disease phenotypes characterized by developmental failure, cancer susceptibility and premature ageing. It remains poorly understood how DNA damage responses function during organismal development and maintain tissue functionality when DNA damage accumulates with ageing. Here we show that the FOXO transcription factor DAF-16 is activated in response to DNA damage during development, whereas the DNA damage responsiveness of DAF-16 declines with ageing. We find that in contrast to its established role in mediating starvation arrest, DAF-16 alleviates DNA-damage-induced developmental arrest and even in the absence of DNA repair promotes developmental growth and enhances somatic tissue functionality. We demonstrate that the GATA transcription factor EGL-27 co-regulates DAF-16 target genes in response to DNA damage and together with DAF-16 promotes developmental growth. We propose that EGL-27/GATA activity specifies DAF-16-mediated DNA damage responses to enable developmental progression and to prolong tissue functioning when DNA damage persists.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Loeb, L. A. & Harris, C. C. Advances in chemical carcinogenesis: a historical review and prospective. Cancer Res. 68, 6863–6872 (2008).
Schumacher, B., Garinis, G. A. & Hoeijmakers, J. H. J. Age to survive: DNA damage and aging. Trends Genet. 24, 77–85 (2008).
Cleaver, J. E., Lam, E. T. & Revet, I. Disorders of nucleotide excision repair: the genetic and molecular basis of heterogeneity. Nat. Rev. Genet. 10, 756–768 (2009).
Wolters, S. & Schumacher, B. Genome maintenance and transcription integrity in aging and disease. Front. Genet. 4, 19 (2013).
Bartek, J. & Lukas, J. DNA damage checkpoints: from initiation to recovery or adaptation. Curr. Opin. Cell Biol. 19, 238–245 (2007).
Sulston, J. E. & Horvitz, H. R. Post-embryonic cell lineages of the nematode, Caenorhabditis elegans. Dev. Biol. 56, 110–156 (1977).
Gartner, A., Milstein, S., Ahmed, S., Hodgkin, J. & Hengartner, M. O. A conserved checkpoint pathway mediates DNA damage-induced apoptosis and cell cycle arrest in C. elegans. Mol. Cell 5, 435–443 (2000).
Stergiou, L., Doukoumetzidis, K., Sendoel, A. & Hengartner, M. O. The nucleotide excision repair pathway is required for UV-C-induced apoptosis in Caenorhabditis elegans. Cell Death Differ. 14, 1129–1138 (2007).
Lans, H. et al. Involvement of global genome repair, transcription coupled repair, and chromatin remodeling in UV DNA damage response changes during development. PLoS Genet. 6, e1000941 (2010).
Guarente, L. & Kenyon, C. Genetic pathways that regulate ageing in model organisms. Nature 408, 255–262 (2000).
Chen, N. et al. WormBase: a comprehensive data resource for Caenorhabditis biology and genomics. Nucleic Acids Res. 33, D383–D389 (2005).
Murakami, S. & Johnson, T. E. A genetic pathway conferring life extension and resistance to UV stress in Caenorhabditis elegans. Genetics 143, 1207–1218 (1996).
Baugh, L. R. & Sternberg, P. W. DAF-16/FOXO regulates transcription of cki-1/Cip/Kip and repression of lin-4 during C. elegans L1 arrest. Curr. Biol. 16, 780–785 (2006).
Ogg, S. et al. The Fork head transcription factor DAF-16 transduces insulin-like metabolic and longevity signals in C. elegans. Nature 389, 994–999 (1997).
Libina, N., Berman, J. R. & Kenyon, C. Tissue-specific activities of C. elegans DAF-16 in the regulation of lifespan. Cell 115, 489–502 (2003).
Henderson, S. T. & Johnson, T. E. daf-16 integrates developmental and environmental inputs to mediate aging in the nematode Caenorhabditis elegans. Curr. Biol. 11, 1975–1980 (2001).
Riou, L. et al. Differential repair of the two major UV-induced photolesions in trichothiodystrophy fibroblasts. Cancer Res. 64, 889–894 (2004).
Kenyon, C., Chang, J., Gensch, E., Rudner, A. & Tabtiang, R. A. C. elegans mutant that lives twice as long as wild type. Nature 366, 461–464 (1993).
Gottlieb, S. & Ruvkun, G. daf-2, daf-16 and daf-23: genetically interacting genes controlling Dauer formation in Caenorhabditis elegans. Genetics 137, 107–120 (1994).
Morris, J. Z., Tissenbaum, H. A. & Ruvkun, G. A phosphatidylinositol-3-OH kinase family member regulating longevity and diapause in Caenorhabditis elegans. Nature 382, 536–539 (1996).
Tullet, J. M. A. et al. Direct inhibition of the longevity-promoting factor SKN-1 by insulin-like signaling in C. elegans. Cell 132, 1025–1038 (2008).
An, J. H. & Blackwell, T. K. SKN-1 links C. elegans mesendodermal specification to a conserved oxidative stress response. Genes Dev. 17, 1882–1893 (2003).
Boyd, W. A. et al. Nucleotide excision repair genes are expressed at low levels and are not detectably inducible in Caenorhabditis elegans somatic tissues, but their function is required for normal adult life after UVC exposure. Mutat. Res. 683, 57–67 (2010).
Baugh, L. R., Demodena, J. & Sternberg, P. W. RNA Pol II accumulates at promoters of growth genes during developmental arrest. Science 324, 92–94 (2009).
Thomas-Chollier, M. et al. RSAT: regulatory sequence analysis tools. Nucleic Acids Res. 36, W119–27 (2008).
Murphy, C. T. et al. Genes that act downstream of DAF-16 to influence the lifespan of Caenorhabditis elegans. Nature 424, 277–283 (2003).
Paek, J. et al. Mitochondrial SKN-1/Nrf mediates a conserved starvation response. Cell Metab. 16, 526–537 (2012).
Gupta, S., Stamatoyannopoulos, J. A., Bailey, T. L. & Noble, W. S. Quantifying similarity between motifs. Genome Biol. 8, R24 (2007).
Curran, S. P., Wu, X., Riedel, C. G. & Ruvkun, G. A soma-to-germline transformation in long-lived Caenorhabditis elegans mutants. Nature 459, 1079–1084 (2009).
Budovskaya, Y. V. et al. An elt-3/elt-5/elt-6 GATA transcription circuit guides aging in C. elegans. Cell 134, 291–303 (2008).
Zhang, P., Judy, M., Lee, S-J. & Kenyon, C. Direct and indirect gene regulation by a life-extending FOXO protein in C. elegans: roles for GATA factors and lipid gene regulators. Cell Metab. 17, 85–100 (2013).
Xu, X. & Kim, S. K. The GATA transcription factor egl-27 delays aging by promoting stress resistance in Caenorhabditis elegans. PLoS Genet. 8, e1003108 (2012).
Charlet-Berguerand, N. et al. RNA polymerase II bypass of oxidative DNA damage is regulated by transcription elongation factors. EMBO J. 25, 5481–5491 (2006).
Barriere, A. & Felix, M. A. Temporal dynamics and linkage disequilibrium in natural Caenorhabditis elegans populations. Genetics 176, 999–1011 (2007).
Hader, D. P. & Lebert, M. in Environmental UV Radiation: Impact on Ecosystems and Human Health and Predictive Models (eds Ghetti, F., Checcucci, G. & Bornman, J. F.) 95–108 Vol. 57 (Springer, 2006).
Mostoslavsky, R. et al. Genomic instability and aging-like phenotype in the absence of mammalian SIRT6. Cell 124, 315–329 (2006).
Niedernhofer, L. J. et al. A new progeroid syndrome reveals that genotoxic stress suppresses the somatotroph axis. Nature 444, 1038–1043 (2006).
Van der Pluijm, I. et al. Impaired genome maintenance suppresses the growth hormone–insulin-like growth factor 1 axis in mice with Cockayne syndrome. PLoS Biol. 5, e2 (2006).
Garinis, G. A. et al. Persistent transcription-blocking DNA lesions trigger somatic growth attenuation associated with longevity. Nat. Cell Biol. 11, 604–615 (2009).
Hsu, A. L., Murphy, C. T. & Kenyon, C. Regulation of aging and age-related disease by DAF-16 and heat-shock factor. Science 300, 1142–1145 (2003).
Johnson, T. E. et al. Longevity genes in the nematode Caenorhabditis elegans also mediate increased resistance to stress and prevent disease. J. Inherit. Metab. Dis. 25, 197–206 (2002).
Sykes, S. M. et al. AKT/FOXO signaling enforces reversible differentiation blockade in myeloid leukemias. Cell 146, 697–708 (2011).
Solari, F., Bateman, A. & Ahringer, J. The Caenorhabditis elegans genes egl-27 and egr-1 are similar to MTA1, a member of a chromatin regulatory complex, and are redundantly required for embryonic patterning. Development 126, 2483–2494 (1999).
Ch’ng, Q. & Kenyon, C. egl-27 generates anteroposterior patterns of cell fusion in C. elegans by regulating Hox gene expression and Hox protein function. Development 126, 3303–3312 (1999).
Herman, M. A. et al. EGL-27 is similar to a metastasis-associated factor and controls cell polarity and cell migration in C. elegans. Development 126, 1055–1064 (1999).
Kumar, R., Wang, R. A. & Bagheri-Yarmand, R. Emerging roles of MTA family members in human cancers. Semin. Oncol. 30, 30–37 (2003).
Nicolson, G. L. et al. Tumor metastasis-associated human MTA1 gene and its MTA1 protein product: role in epithelial cancer cell invasion, proliferation and nuclear regulation. Clin. Exp. Metastasis 20, 19–24 (2003).
Li, D. Q., Ohshiro, K., Khan, M. N. & Kumar, R. Requirement of MTA1 in ATR-mediated DNA damage checkpoint function. J. Biol. Chem. 285, 19802–19812 (2010).
Luijsterburg, M. S. et al. Stochastic and reversible assembly of a multiprotein DNA repair complex ensures accurate target site recognition and efficient repair. J. Cell Biol. 189, 445–463 (2010).
Kirkwood, T. B. Understanding the odd science of aging. Cell 120, 437–447 (2005).
De Magalhaes, J. P. & Church, G. M. Genomes optimize reproduction: aging as a consequence of the developmental program. Physiology 20, 252–259 (2005).
Kamath, R. S. et al. Systematic functional analysis of the Caenorhabditis elegans genome using RNAi. Nature 421, 231–237 (2003).
Reboul, J. et al. C. elegans ORFeome version 1.1: experimental verification of the genome annotation and resource for proteome-scale protein expression. Nat. Genet. 34, 35–41 (2003).
Brenner, S. The genetics of Caenorhabditis elegans. Genetics 77, 71–94 (1974).
Pfaffl, M. W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 29, e45 (2001).
Furuyama, T., Nakazawa, T., Nakano, I. & Mori, N. Identification of the differential distribution patterns of mRNAs and consensus binding sequences for mouse DAF-16 homologues. Biochem. J. 349, 629–634 (2000).
Sakurai, H. & Enoki, Y. Novel aspects of heat shock factors: DNA recognition, chromatin modulation and gene expression. FEBS J. 277, 4140–4149 (2010).
Sinclair, J. & Hamza, I. A novel heme-responsive element mediates transcriptional regulation in Caenorhabditis elegans. J. Biol. Chem. 285, 39536–39543 (2010).
Habbig, S. et al. NPHP4, a cilia-associated protein, negatively regulates the Hippo pathway. J. Cell Biol. 193, 633–642 (2011).
Acknowledgements
We thank S. Keller and K. Riehl for excellent technical assistance, D. Magner and A. Steckelberg for contributions earlier in the project, C. Jüngst and A. Schauss of the CECAD Imaging Facility, and J. Altmüller, I. Bässmann and C. Becker of the CCG. Some strains were kindly provided by the MITANI Lab through the National Bio-Resource Project of the MEXT, Japan and CGC (funded by the NIH Office of Research Infrastructure Programs (P40 OD010440)). V.B. received an IGSDHD, and M.A.E. an EMBO long-term fellowship. B.Schermer was supported by the Deutsche Forschungsgemeinschaft (SFB832 and SCHE 1562/2). B.Schumacher acknowledges financial support from the Deutsche Forschungsgemeinschaft (CECAD, SFB 829, SFB 670 and KFO 286), the European Research Council (ERC Starting grant 260383), Marie Curie (FP7 ITN CodeAge 316354, aDDRess 316390, MARRIAGE 316964 and ERG 239330), the German-Israeli Foundation (GIF, 2213-1935.13/2008 and 1104-68.11/2010), the Deutsche Krebshilfe (109453) and the Bundesministerium für Forschung und Bildung (Sybacol FKZ0315893A-B).
Author information
Authors and Affiliations
Contributions
M.M.M., L.C-R., V.B., M.A.E., R-U.M., A.B.W., S.G. and J.I.S. performed experiments and analysed data; L.C-R., P.F. and B.Schumacher conducted bioinformatics analysis; T.B. and B.Schermer contributed materials; B.Schumacher wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1
(A) L1 larvae were treated with UV and mock treated and developmental stages assessed at indicated time points afterwards (average of n = 8 independent experiments is shown, >26 individuals analysed per experiment; error bars = SD). (B) NER mutants csb-1, xpc-1, xpa-1 and xpc-1;csb-1 double mutant were treated as L1 larvae and developmental stages assessed 48 h post treatment (average of n = 3 independent experiments per strain and dose is shown, >525 individuals analysed per experiment; error bars = SD). (C) Anaphase bridges are present in mitotically dividing germ cells 16 hs after 60 mJ/cm2 UV irradiation in xpc-1(tm3886) mutants (lower panel), but not in wt (upper panel). Young adult worms were treated 24 h post L4 (DAPI staining, scale bar corresponds to 5um, representative picture shown, experiment repeated 3 times). (D) UV response in wt (N2) worms is positively correlated with low dose UV response in NER deficient xpa-1 mutants and with starvation response. Fed wild type worms were treated with 60 mJ/cm2 UVB, xpa-1 mutants with 10 mJ/cm2 and compared to respective non-treated controls. Significantly differentially expressed genes were analysed by Spearman rank correlation (D) between UV treated wt and xpa-1 mutants (left bar) and UV treated and starved wt worms (right bar). (E) shows xy blot of fold changes of UV treated wt and xpa-1 mutant worms. (F) Functional clustering revealed significantly overrepresented biological processes of the interaction network (Figure 1B). (G) Subcellular DAF-16::GFP localization upon UV-irradiation. L3 larvae of wt, xpa-1 and csb-1 mutants carrying the zIs356 daf-16::GFP transgene were irradiated with 150 mJ/cm2 and kept at 20 °C (average of n = 4 independent experiments; 10 individuals analysed per experiment; error bars = SEM).
Supplementary Figure 2
(A) DAF-16::GFP cellular localization upon low dose UV treatment in adult worms. Wt and xpa-1 mutant adult worms during the first, third, fifth, seventh and tenth day of adulthood were exposed to 60 mJ/cm2 and incubated at 20 °C. The subcellular localization of the GFP signal was scored after 3, 5, 7, 10 and 24 hours of the UVB irradiation (average of n = 3 independent experiments pair strain and time point; 10 individuals analysed per experiment; error bars = SEM). (B, C) Initial induction of CPD lesions upon UV-treatment is unaltered throughout adulthood. DAF-16::GFP transgenic animals in wt (B) or xpa-1 (C) background were exposed to UV or left untreated. Immediately after treatment DNA was extracted and in serial dilutions probed with anti-CPD antibodies (representative results shown; experiment repeated 3 times) (D) Additional daf-2 alleles show reduced developmental arrest and enhanced resistance to UV-induced damage (average of n = 3 independent experiments per strain and dose is shown, > 1900 individuals analysed per experiment; error bars = SD, two-tailed T-Test compared to wt, ∗ = p < 0.05, ∗∗ = p < 0.01, ∗∗∗ = p < 0.001). (E) daf-16 alleles were UV treated at L1 stage and larval stages were analysed 48 h post treatment (average of n = 3 independent experiments per strain and dose is shown, > 771 individuals error bars = SD, ∗ = p < 0.05, ∗∗ = p < 0.01, ∗∗∗ = p < 0.001, two-tailed T-Test compared to wt). (F) Adult lifespan at 20 °C of wt, daf-2, daf-2;daf-16 that were treated with UV at the L1 stage. Equal numbers of synchronized L1 larvae were treated with 80 mJ/cm2 UVB or mock treated. (regression of n > 163 individuals per strain and dose, see Suppl. Table 12 for exact n values). N2 median survival: 0 mJ/cm2 16d, 80 mJ/cm2 13d p < 0.0001; daf-2 median survival: 0 mJ/cm2 29d, 80 mJ/cm2 24.5d p = 0.58; daf-2;daf-16 median survival: 0 mJ/cm2 15d, 80 mJ/cm2 13d p < 0.01 (Log-rank Test).
Supplementary Figure 3
(A) L1 larvae were synchronized and non-GFP (homozygous for skn-1) worms were selected using a Union Biometrica biosorter. Wt control was also run through the biosorter. Worms were afterwards exposed to UV and larval stages assessed 48 h post treatment (average of n = 3 independent experiments per strain and dose is shown, > 220 individuals analysed per experiment; ∗ = p < 0.05, two-tailed T-Test compared to wt). (B) L1 larvae were synchronized and non-GFP skn-1 mutant (homozygous for skn-1) worms were selected using a Union Biometrica biosorter. Controls were also run through the biosorter. Worms were afterwards exposed to UV and larval stages assessed 48 h post treatment (average of n = 3 independent experiments per strain and dose is shown, >441 individuals analysed per experiment; ∗ = p < 0.05, two-tailed T-Test, in pairwise comparisons of strains with and without the skn-1(zu135) background). (C) Expression of SKN-1::GFP is unaffected on UV treatment. SKN-1::GFP is constitutively expressed in ASI neurons (arrows). Upon NaAz treatment, SKN-1::GFP expression is induced in intestinal nuclei (arrowheads). L3 larvae were treated with 100 mJ/cm2 UV and kept at 20 °C; images were taken 4 h after UV treatment or after 5 min of 50 mM NaAz exposure. The left panels show untreated worms, the middle panels UV-treated worms, and the right panels NaAz treated worms. A) shows SKN-1::GFP transgenic animals, Is007[skn-1b/c::GFP;rol-6], and B) SKN-1::GFP;xpa-1 transgenic mutant animals, BJS226[skn-1b/c::GFP;rol-6];xpa-1(ok698). Pictures were taken using a confocal microscope equipped with a white light laser (Leica TCS SP8). The sample was excited with a wavelength of 488 nm and two different emission signals were collected simultaneously between 490- 512 nm (GFP + autofluorescence, green) and between 570 - 652 nm (autofluorescence only, red). The overlay of the two channels clearly separates the signal from the GFP staining (green) and the autofluorescence signal (orange). Representative images shown (experiment repeated at least 5 times in the laboratory).
Supplementary Figure 4
(A) daf-2(e1370) maintains motility of irradiated xpa-1 worms. L4 larvae were UV or mock treated and single adult worms were left on OP50 seeded plate for 1 hour on day 9 after the irradiation. Representative pictures of tracks caused by motion of the worm on the plate are shown (experiment repeated at least 3 times in the laboratory). (B) IIS attenuation leads to enhanced longevity even in the presence of unrepaired DNA lesions. Median lifespan at 20 °C of xpa-1(ok698) young adult worms UVB irradiated or mock treated 24 hs after L4: 0 mJ/cm2: 19 days; 2.5 mJ/cm2: 17 days; 5 mJ/cm2: 14.5 days and xpa-1(ok698),daf-2(e1370) L4 worms: 0 mJ/cm2: 33.5 days; 2.5 mJ/cm2: 28 days; 5 mJ/cm2: 25 days. Comparison of the survival curves by the Log-rank (Mantel-Cox) test shows that the xpa-1 curve is always significantly different (∗∗∗∗ p < 0.0001) than the corresponding xpa-1, daf-2 curve at all doses. The p-values of the differences of the survival curve of xpa-1 at 2.5 mJ/cm2 is (∗∗∗ p < 0.0007) and at 5 mJ/cm2 (∗∗∗∗ p < 0.0001) compared to the xpa-1 0 mJ/cm2 curve; for xpa, daf-2 the p-values of the differences of the survival curve of xpa-1 at 2.5 mJ/cm2 is (∗ p < 0.0318) and at 5 mJ/cm2 (∗∗∗∗ p < 0.0001) compared to the xpa-1, daf-2 0 mJ/cm2 curve. (regression of n = 90 individuals per strain and dose). (C) Reduced lifespan in xpa-1 mutants on UV treatment. Median lifespan at 20 °C of wt L4 worms (21 days) is not significantly different (p-value 0.4538) than the one of xpa-1 worms (20 days). After treatment of L4 worms median life span is significantly reduced: after 2.5 mJ/cm2 xpa-1 worms (16 days), wt (19 days) [p = 0.0001]; after 10 mJ/cm2 xpa-1 worms (16 days), wt (21 days) [p < 0.0001]; after 25 mJ/cm2 xpa-1 worms (12 days), wt (23 days) [p < 0.0001]; (Comparison of the survival curves by the Log-rank (Mantel-Cox) test; regression of n > 74 individuals in all experiments). (D) Synchronous L1 larvae were treated similarly as in the other developmental arrest assay experiments, but after irradiation they were kept for 30 hs at 20 °C and afterwards at 25 °C for 18 hs until the larval stages were scored (average of n = 3 independent experiments per strain and dose is shown, > 818 animals per dose and strain; error bars = SD, two-tailed T-Test.) > 100 untreated glp-4 worms were transferred to a new plate and kept at 25 °C for additional 3 days and no embryos were laid.
Supplementary Figure 5
Functional clustering of genes that were up- or downregulated upon UV treatment (p < 0.01, FC > ±1.5) or starvation (p < 0.01, FC > ±2) reveals significantly overrepresented biological processes.
Supplementary Figure 6
(A) L1 larvae were synchronized and non-GFP (homozygous for skn-1) worms were selected using a Union Biometrica biosorter. Wt control was also run through the biosorter. Synchronized L1 larvae were either fed for four hours or left under starvation and exposed to UV treatment. Upon UV treatment larvae were provided with food and developmental stages were assessed 48 h post UV irradiation (average of n = 3 independent experiments is shown, > 295 individuals analysed per experiment; error bars = SD, ∗∗∗ = p < 0.001, two-tailed T-Test comparing pre-starved to pre-fed of the same strain). (B) Biological processes that were significantly (p < 0.01) overrepresented among genes that were significantly induced (FC > 1.5; p < 0.01) upon UV in daf-2 mutants and were more strongly induced upon UV in daf-2 vs. daf-2;daf-16, daf-2 vs. wt, and wt vs. daf-16. (C) GATA mutants were UV treated at L1 stage and larval stages were analysed 48 h post treatment (average of n = 3 independent experiments per strain and dose is shown, > 1006 individuals analysed per experiment; error bars = SD). (D) Genetic map based on sequencing results of egl-27(n170) and egl-27(ok1670) mutants superimposed on wormbase.org screenshot.
Supplementary Figure 7
(A) Oxidative stress induced by paraquat affects development in both daf-16 and egl-27 mutants The daf-16, egl-27 double mutant showed a synthetic developmental arrest phenotype in the presence of paraquat. Synchronized L1 worms of N2(wt), daf-16(mu86), egl-27(ok1670) and daf-16(mu86);egl-27(ok1670) were put on plates containing 0.1 mM or 5 mM paraquat or on control plates and the developmental stages assessed 48 h post treatment (average of n = 3 independent experiments per strain and dose; > 30 individuals analysed per experiment; error bars = SD, ∗ = p < 0.05, ∗∗ = p < 0.01, ∗∗∗ = p < 0.001, two-tailed T-Test). (B) daf-16 but not egl-27 mutants are significantly less resistant to heat shock at 35 °C. L4 larvae of N2(wt), daf-16(mu86) and egl-27(ok1670) were kept at 35 °C for the indicated time and were scored for survival (average of n = 2 independent experiments for each strain and time point; 30 individuals analysed per experiment; error bars = SD;∗ = p < 0.05, ∗∗ = p < 0.01, ∗∗∗ = p < 0.001, two-tailed T-Test compared to wt). (C) egl-27 mutation leads to developmental arrest in pre-dauer larval stages at 25C in the daf-2 mutational background, while daf-16 mutants are unable to form dauer larvae but proceed with development. Synchronized L1 larvae of daf-2(e1370), daf-2(e1370), daf-16(mu86) and daf-2(e1370), egl-27(ok1670) were incubated at 25C for 48 hours and were scored for developmental stage (average of n = 6 independent experiments per strain; > 30 individuals analysed per experiment; error bars = SD;∗ = p < 0.05, ∗∗ = p < 0.01, ∗∗∗ = p < 0.001, two-tailed T-Test compared to wt). (D) Differential interference contrast (DIC) images of DAF-16::GFP and EGL-27::GFP, of Figure 7A. L1 synchronized larvae were fed and UV treated with 150 mJ/cm2 and kept at 20 °C, images were taken 6 hs after. Upper panels shows examples of DAF-16::GFP worms, left panel mock treated and right panel a completely nuclear DAF-16::GFP UV-irradiated worm. Lower panels show examples of EGL-27::GFP worms, left panel mock treated and right panel UV-irradiated.
Supplementary Figure 8
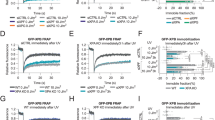
(A, B) Images of the whole immunoblots underlying the images in Figure 7B left panel. (A) IPs stained both anti-V5 and anti-Flag (2° antibody light chain-specific). (B) Lysates stained both anti-V5 and anti-Flag, Left panel short exposure, right panel long exposure. * additional band that is likely to be a DAF-16.V5 degradation product as it is detected by the V5 antibody in lanes containing DAF-16.V5 in some but not all experiments. This band co-precipitates with EGL-27FL.Flag as well. Note that this band does not correspond to a possible staining of the heavy chain (which generally runs above the 50 kD marker)- since a light chain-specific 2° antibody was used. (C-E) Images of the whole immunoblots underlying the images in Fig. 7B, right panel. (C) IPs stained anti-V5 (secondary antibody non-light chain specific). (D) IPs stained anti-Flag (after anti-V5, secondary antibody non-light chain specific). (E) Lysates stained anti-V5. (F) Domain structure of EGL-27d.
Supplementary information
Supplementary Information
Supplementary Information (PDF 991 kb)
Supplementary Table 1
Supplementary Information (XLSX 49 kb)
Supplementary Table 2
Supplementary Information (XLSX 163 kb)
Supplementary Table 3
Supplementary Information (XLSX 206 kb)
Supplementary Table 4
Supplementary Information (XLSX 426 kb)
Supplementary Table 5
Supplementary Information (XLSX 72 kb)
Supplementary Table 6
Supplementary Information (XLSX 22 kb)
Supplementary Table 7
Supplementary Information (XLSX 17 kb)
Supplementary Table 8
Supplementary Information (XLSX 31 kb)
Supplementary Table 9
Supplementary Information (XLSX 127 kb)
Supplementary Table 10
Supplementary Information (XLSX 15 kb)
Supplementary Table 11
Supplementary Information (XLSX 37 kb)
Supplementary Table 12
Supplementary Information (XLSX 11 kb)
Supplementary Table 13
Supplementary Information (XLSX 10 kb)
Rights and permissions
About this article
Cite this article
Mueller, M., Castells-Roca, L., Babu, V. et al. DAF-16/FOXO and EGL-27/GATA promote developmental growth in response to persistent somatic DNA damage. Nat Cell Biol 16, 1168–1179 (2014). https://doi.org/10.1038/ncb3071
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3071
This article is cited by
-
Ribosome biogenesis disruption mediated chromatin structure changes revealed by SRAtac, a customizable end to end analysis pipeline for ATAC-seq
BMC Genomics (2023)
-
The central role of DNA damage in the ageing process
Nature (2021)
-
GATA3 recruits UTX for gene transcriptional activation to suppress metastasis of breast cancer
Cell Death & Disease (2019)
-
DAF-16/FOXO and HLH-30/TFEB function as combinatorial transcription factors to promote stress resistance and longevity
Nature Communications (2018)
-
RNA surveillance via nonsense-mediated mRNA decay is crucial for longevity in daf-2/insulin/IGF-1 mutant C. elegans
Nature Communications (2017)