Abstract
We have previously demonstrated that Stat3 regulates lysosomal-mediated programmed cell death (LM-PCD) during mouse mammary gland involution in vivo. However, the mechanism that controls the release of lysosomal cathepsins to initiate cell death in this context has not been elucidated. We show here that Stat3 regulates the formation of large lysosomal vacuoles that contain triglyceride. Furthermore, we demonstrate that milk fat globules (MFGs) are toxic to epithelial cells and that, when applied to purified lysosomes, the MFG hydrolysate oleic acid potently induces lysosomal leakiness. Additionally, uptake of secreted MFGs coated in butyrophilin 1A1 is diminished in Stat3-ablated mammary glands and loss of the phagocytosis bridging molecule MFG-E8 results in reduced leakage of cathepsins in vivo. We propose that Stat3 regulates LM-PCD in mouse mammary gland by switching cellular function from secretion to uptake of MFGs. Thereafter, perturbation of lysosomal vesicle membranes by high levels of free fatty acids results in controlled leakage of cathepsins culminating in cell death.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Luzio, J. P., Pryor, P. R. & Bright, N. A. Lysosomes: fusion and function. Nat. Rev. Mol. Cell Biol. 8, 622–632 (2007).
Foghsgaard, L. et al. Cathepsin B acts as a dominant execution protease in tumor cell apoptosis induced by tumor necrosis factor. J. Cell Biol. 153, 999–1010 (2001).
Kroemer, G. & Jaattela, M. Lysosomes and autophagy in cell death control. Nat. Rev. Cancer 5, 886–897 (2005).
Cox, T. M. & Cachón-González, M. B. The cellular pathology of lysosomal diseases. J. Pathol. 226, 241–254 (2012).
Manzoni, C. & Lewis, P. A. Dysfunction of the autophagy/lysosomal degradation pathway is a shared feature of the genetic synucleinopathies. FASEB J. 27, 3424–3429 (2013).
Kirkegaard, T. & Jaattela, M. Lysosomal involvement in cell death and cancer. Biochim. Biophys. Acta 1793, 746–754 (2009).
Kallunki, T., Olsen, O. D. & Jaattela, M. Cancer-associated lysosomal changes: friends or foes? Oncogene 32, 1995–2004 (2012).
Vasiljeva, O. et al. Reduced tumour cell proliferation and delayed development of high-grade mammary carcinomas in cathepsin B-deficient mice. Oncogene 27, 4191–4199 (2008).
Kreuzaler, P. & Watson, C. J. Killing a cancer: what are the alternatives? Nat. Rev. Cancer 12, 411–424 (2012).
Watson, C. J. & Kreuzaler, P. A. Remodeling mechanisms of the mammary gland during involution. Int. J. Dev. Biol. 55, 757–762 (2011).
Watson, C. J. Involution: apoptosis and tissue remodelling that convert the mammary gland from milk factory to a quiescent organ. Breast Cancer Res. 8, 203 (2006).
Watson, C. J. & Khaled, W. T. Mammary development in the embryo and adult: a journey of morphogenesis and commitment. Development 135, 995–1003 (2008).
Jeong, J. et al. A test of current models for the mechanism of milk-lipid droplet secretion. Traffic 14, 974–986 (2013).
Lund, L. R. et al. Two distinct phases of apoptosis in mammary gland involution: proteinase-independent and -dependent pathways. Development 122, 181–193 (1996).
Chapman, R. S. et al. Suppression of epithelial apoptosis and delayed mammary gland involution in mice with a conditional knockout of Stat3. Genes Dev. 13, 2604–2616 (1999).
Humphreys, R. C. et al. Deletion of Stat3 blocks mammary gland involution and extends functional competence of the secretory epithelium in the absence of lactogenic stimuli. Endocrinology 143, 3641–3650 (2002).
Kreuzaler, P. A. et al. Stat3 controls lysosomal-mediated cell death in vivo. Nat. Cell Biol. 13, 303–309 (2011).
Kerr, J. F. & Searle, J. Deletion of cells by apoptosis during castration-induced involution of the rat prostate. Virchows Arch. B Cell Pathol. 13, 87–102 (1973).
Lockshin, R. A. & Zakeri, Z. Caspase-independent cell death? Oncogene 23, 2766–2773 (2004).
Luke, C. J. & Silverman, G. A. Necrotic cell death: harnessing the Dark side of the Force in mammary gland involution. Nat. Cell Biol. 13, 197–199 (2011).
Aits, S. & Jäättelä, M. Lysosomal cell death at a glance. J. Cell Sci. 126, 1905–1912 (2013).
Galluzzi, L., Bravo-San Pedro, J. M. & Kroemer, G. Organelle-specific initiation of cell death. Nat. Cell Biol. 16, 728–736 (2014).
Kågedal, K. et al. Lysosomal membrane permeabilization during apoptosis — involvement of Bax? Int. J. Exp. Pathol. 86, 309–321 (2005).
Ono, K., Kim, S. O. & Han, J. Susceptibility of lysosomes to rupture is a determinant for plasma membrane disruption in tumor necrosis factor alpha-induced cell death. Mol. Cell. Biol. 23, 665–676 (2003).
Zaragoza, R. et al. Nitration of cathepsin D enhances its proteolytic activity during mammary gland remodelling after lactation. Biochem. J. 419, 279–288 (2009).
Margaryan, N. V. et al. New insights into cathepsin D in mammary tissue development and remodeling. Cancer Biol. Ther. 10, 457–466 (2010).
Helminen, H. J. & Ericsson, J. L. E. Effects of enforced milk stasis on mammary gland epithelium, with special reference to changes in lysosomes and lysosomal enzymes. Exp. Cell Res. 68, 411–427 (1971).
Gomes, L. C., Benedetto, G. D. & Scorrano, L. During autophagy mitochondria elongate, are spared from degradation and sustain cell viability. Nat. Cell Biol. 13, 589–598 (2011).
Klionsky, D. J. et al. Guidelines for the use and interpretation of assays for monitoring autophagy. Autophagy 8, 445–544 (2012).
Mather, I. & Keenan, T. Origin and secretion of milk lipids. J. Mammary Gland Biol. Neoplasia 3, 259–273 (1998).
Smith, S., Watts, R. & Dils, R. Quantitative gas–liquid chromatographic analysis of rodent milk triglycerides. J. Lipid Res. 9, 52–57 (1968).
Listenberger, L. L. et al. Triglyceride accumulation protects against fatty acid-induced lipotoxicity. Proc. Natl Acad. Sci. USA 100, 3077–3082 (2003).
Yuzefovych, L., Wilson, G. & Rachek, L. Different effects of oleate vs. palmitate on mitochondrial function, apoptosis, and insulin signaling in L6 skeletal muscle cells: Role of oxidative stress. Am. J. Physiol. Endocrinol. Metab. 299, E1096–E1105 (2010).
Huai, J. et al. TNF-induced lysosomal membrane permeability (LMP) is downstream of MOMP and triggered by caspase-mediated p75 cleavage and ROS formation. J. Cell Sci. 126, 4015–4025 (2013).
Jespersen, H., Andersen, J. H., Ditzel, H. J. & Mouritsen, O. G. Lipids, curvature stress, and the action of lipid prodrugs: free fatty acids and lysolipid enhancement of drug transport across liposomal membranes. Biochimie 94, 2–10 (2012).
Rammer, P. et al. BAMLET activates a lysosomal cell death program in cancer cells. Mol. Cancer Ther. 9, 24–32 (2010).
Fontana, A., Spolaore, B. & Polverino de Laureto, P. The biological activities of protein/oleic acid complexes reside in the fatty acid. Biochim. Biophys. Acta 1834, 1125–1143 (2013).
Ogg, S. L., Weldon, A. K., Dobbie, L., Smith, A. J. H. & Mather, I. H. Expression of butyrophilin (Btn1a1) in lactating mammary gland is essential for the regulated secretion of milk-lipid droplets. Proc. Natl Acad. Sci. USA 101, 10084–10089 (2004).
Clarkson, R., Wayland, M., Lee, J., Freeman, T. & Watson, C. Gene expression profiling of mammary gland development reveals putative roles for death receptors and immune mediators in post-lactational regression. Breast Cancer Res. 6, R92–R109 (2004).
Singh, R. et al. Autophagy regulates lipid metabolism. Nature 458, 1131–1135 (2009).
Monks, J., Smith-Steinhart, C., Kruk, E. R., Fadok, V. A. & Henson, P. M. Epithelial cells remove apoptotic epithelial cells during post-lactation involution of the mouse mammary gland. Biol. Reprod. 78, 586–594 (2008).
Hughes, K., Wickenden, J. A., Allen, J. E. & Watson, C. J. Conditional deletion of Stat3 in mammary epithelium impairs the acute phase response and modulates immune cell numbers during post-lactational regression. J. Pathol. 227, 106–117 (2012).
Merkel, M., Tilkorn, A-C., Greten, H. & Ameis, D. Lysosomal Acid Lipase Vol. 109, 95–107 (Methods Mol. Biol., 1999).
Atabai, K. et al. Mfge8 is critical for mammary gland remodeling during involution. Mol. Biol. Cell 16, 5528–5537 (2005).
Hanayama, R. & Nagata, S. Impaired involution of mammary glands in the absence of milk fat globule EGF factor 8. Proc. Natl Acad. Sci. USA 102, 16886–16891 (2005).
Lockshin, R. A. & Williams, C. M. Programmed cell death, Cytolytic enzymes in relation to the breakdown of the intersegmental muscles of silkmoths. J. Insect Physiol. 11, 831–844 (1965).
Hara-Nishimura, I. & Hatsugai, N. The role of vacuole in plant cell death. Cell Death Differ. 18, 1298–1304 (2011).
vanDoorn, W. G. et al. Morphological classification of plant cell deaths. Cell Death Differ. 18, 1241–1246 (2011).
Cornillon, S. et al. Programmed cell death in Dictyostelium. J. Cell Sci. 107, 2691–2704 (1994).
Maltese, W. A. & Overmeyer, J. H. Methuosis: nonapoptotic cell death associated with vacuolization of macropinosome and endosome compartments. Am. J. Pathol. 184, 1630–1642 (2014).
Kitambi, S. S. et al. Vulnerability of glioblastoma cells to catastrophic vacuolization and death induced by a small molecule. Cell 157, 313–328 (2014).
Unger, R. H., Scherer, P. E. & Holland, W. L. Dichotomous roles of leptin and adiponectin as enforcers against lipotoxicity during feast and famine. Mol. Biol. Cell 24, 3011–3015 (2013).
Bournazou, E. & Bromberg, J. Targeting the tumour microenvironment: JAK-STAT3 signaling. JAK-STAT 2, e23828 (2013).
Alonzi, T. et al. Essential role of STAT3 in the control of the acute-phase response as revealed by inducible gene activation in the liver. Mol. Cell. Biol. 21, 1621–1632 (2001).
Selbert, S. & Bentley, D. Efficient BLG-Cre mediated gene deletion in the mammary gland. Transgenic Res. 7, 387–398 (1998).
Roth, W. et al. Cathepsin L deficiency as molecular defect of furless: hyperproliferation of keratinocytes and pertubation of hair follicle cycling. FASEB J. 14, 2075–2086 (2000).
Silvestre, J-S. et al. Lactadherin promotes VEGF-dependent neovascularization. Nat. Med. 11, 499–506 (2005).
Reichmann, E., Ball, R., Groner, B. & Friis, R. R. New mammary epithelial and fibroblastic cell clones in coculture form structures competent to differentiate functionally. J. Cell Biol. 108, 1127–1138 (1989).
Jahreiss, L., Renna, M., Bittman, R., Arthur, G. & Rubinsztein, D. C. 1-O-Hexadecyl-2-O-methyl-3-O-(2′-acetamido-2′-deoxy-beta-D-glucopyranosyl)-sn-glycerol (Gln) induces cell death with more autophagosomes which is autophagy-independent. Autophagy 5, 835–846 (2009).
Becken, U., Jeschke, A., Veltman, K. & Hass, A. Cell-free fusion of bacteria-containing phagosomes with endocytic compartments. Proc. Natl Acad. Sci. USA 107, 20726–20731 (2010).
Acknowledgements
We thank H. Skelton for assistance with histology, A. Gilmore (University of Manchester, UK) for the Bax–GFP construct, I. Mather (University of Maryland, USA) for the anti-BTN antibody and useful advice, and D. Neal, S. Felisbino and S. Hawkins (CRUK Cambridge Institute, University of Cambridge, UK) for providing mouse prostate tissue and advice. We thank also T. Reinheckel for providing the cathepsin L KO mice. In addition, we thank A. Tolkovsky and Z. Zakeri for helpful discussions. This work was supported by a grant from the Medical Research Council programme grant no. MR/J001023/1 (T.J.S. and B.L-L.) and a Cancer Research UK Cambridge Cancer Centre PhD studentship (H.K.R.).
Author information
Authors and Affiliations
Contributions
T.J.S. and B.L-L. carried out most of the experiments, H.K.R. contributed the cathepsin L−/− tissue samples, A.R-M. provided the prostate samples, J.S. carried out the TEM and immunogold analysis and assisted in data interpretation. T.J.S., B.L-L. and C.J.W. designed the work, analysed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
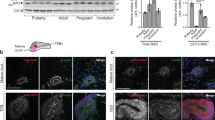
Supplementary Figure 1 Cathepsin D and LAMP2 staining in the involuting mammary gland.
(a) Staining for cathepsin D (red) in the lactating and involuting gland. Three animals were assessed per condition. (b) The pro-form of cathepsin D was detected at approximately 46 kDa and higher levels were present at 24 h involution compared to lactating mammary glands. There was no difference between control (C) and Stat3 knockout (KO). Lanes represent independent biological samples. (c) LAMP2 staining (red) is detected lining large vacuolar structures in the control 24 h involuting mammary gland but not in the lactating mammary gland. This becomes more apparent at 48 h and 72 h. In the Stat3 KO mammary gland, LAMP2-positive vacuoles are only seen from 48 h onwards. One animal per condition was analysed. (d) Confocal images displaying immunostaining for cathepsin D (red) is shown in grey-scale and merged with staining for triglyceride (lipidtox, green). Arrowheads show lipid droplets inside lysosomal vesicles. Three animals were used. Nuclei are visualized by Hoechst (blue). Scale bars = 20 μm.
Supplementary Figure 2
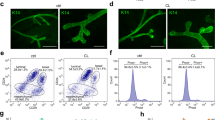
(a) Milk induces lysosomal lipid accumulation. Confocal images show Lysotracker red staining overlapping with that for triglyceride (lipidtox, green) (colocalization shown by arrowheads). Four representative images from two independent experiments displayed. Nuclei are stained with Hoechst (blue). Scale bars first three rows = 1 μm, 4th row = 2 μm. (b) Free fatty acids induce cell death. Staining for triglyceride (lipidtox, green) in EpH4 cells treated overnight with 1 mM oleic acid (OA) and palmitic acid (PA) showing lipid accumulation in EpH4 cells. Nuclei are stained with Hoechst (blue); Scale bars = 10 μm. Brightfield images showing cytotoxicity in fatty acid treated EpH4 cells; Scale bars = 100 μm. (c) Fatty acid induced cell death was assessed by trypan blue positivity. Means ± s.e.m. from n = three independent experiments with 2–3 technical replicates performed per experiment shown (∗P < 0.05, one-way ANOVA, Dunnett’s Multiple Comparison post-test). For raw values, see the corresponding worksheet in Supplementary Table 3.
Supplementary Figure 3
(a) Optimisation of digitonin cytosol extraction assay. EpH4 cells were extracted with increasing concentrations of digitonin and cathepsin activity assayed over time with the synthetic substrate Z-Phe-Arg-AMC. Total activity was measured by extraction with 0.1% TritonX-100. A digitonin concentration of 25 μg ml−1 was selected for cytosol extraction assays. All data is plotted, optimisation performed on one occasion. (b) Fatty acids induce deacidification of the lysosomal compartment. A population of low Lysotracker Red staining (region R8) is induced with 1 mM OA or PA, indicative of de-acidification of lysosomes. Cells treated with 1 mM PA also display a population with higher lysotracker red staining. Quantification of n = four independent experiments as described in (b). Means ± s.e.m. are shown, associated statistics source data can be found in the corresponding worksheet in Supplementary Table 3. (c) OA and PA (500 μM) treated cells showing populations of Lysotracker Red fluorescence (R13), with low levels indicative of de-acidification of lysosomes (n = 1 (PA) and 2 (OA) independent experiments raw values can be found in the corresponding worksheet in Supplementary Table 3).
Supplementary Figure 4 Fatty acid treatment does not result in Bax translocation to lysosomes.
(a) EpH4 cells were transfected with GFP-Bax (green) and treated with ethanol, 500 μm oleic acid (OA), palmitic acid (PA) overnight prior to fixation and LAMP2 immunostaining (red). Cells were treated with 30 ng ml−1 TNFα and 10 μg ml−1 cycloheximide for 6.5 h in serum free conditions as a control. No obvious lysosomal co-localization was observed under these conditions. Two representative examples from all conditions displayed, experiment performed once. (b) GFP-Bax transfected EpH4 cells treated with TNFα and cycloheximide or 1 μM staurosporine as indicated for 6.5 h were fixed and immunostained for AIF (red) and show mitochondrial localization of Bax under these conditions. Nuclei are stained with Hoechst (blue). Scale bars = 10 μm.
Supplementary information
Supplementary Information
Supplementary Information (PDF 898 kb)
Supplementary Table 1
Supplementary Information (XLSX 9 kb)
Supplementary Table 2
Supplementary Information (XLSX 9 kb)
Supplementary Table 3
Supplementary Information (XLSX 225 kb)
Rights and permissions
About this article
Cite this article
Sargeant, T., Lloyd-Lewis, B., Resemann, H. et al. Stat3 controls cell death during mammary gland involution by regulating uptake of milk fat globules and lysosomal membrane permeabilization. Nat Cell Biol 16, 1057–1068 (2014). https://doi.org/10.1038/ncb3043
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb3043
This article is cited by
-
Using Organoids to Tap Mammary Gland Diversity for Novel Insight
Journal of Mammary Gland Biology and Neoplasia (2024)
-
Intracellular calcium links milk stasis to lysosome-dependent cell death during early mammary gland involution
Cellular and Molecular Life Sciences (2024)
-
Hormonal regulation of mammary gland development and lactation
Nature Reviews Endocrinology (2023)
-
Fur removal promotes an earlier expression of involution-related genes in mammary gland of lactating mice
Journal of Comparative Physiology B (2023)
-
Phosphorylation of STAT3 at Tyr705 contributes to TFEB-mediated autophagy-lysosomal pathway dysfunction and leads to ischemic injury in rats
Cellular and Molecular Life Sciences (2023)