Abstract
The mixed lineage kinase domain-like protein (MLKL) has recently been identified as a key RIP3 (receptor interacting protein 3) downstream component of tumour necrosis factor (TNF)-induced necroptosis. MLKL is phosphorylated by RIP3 and is recruited to the necrosome through its interaction with RIP3. However, it is still unknown how MLKL mediates TNF-induced necroptosis. Here, we report that MLKL forms a homotrimer through its amino-terminal coiled-coil domain and locates to the cell plasma membrane during TNF-induced necroptosis. By generating different MLKL mutants, we demonstrated that the plasma membrane localization of trimerized MLKL is critical for mediating necroptosis. Importantly, we found that the membrane localization of MLKL is essential for Ca2+ influx, which is an early event of TNF-induced necroptosis. Furthermore, we identified that TRPM7 (transient receptor potential melastatin related 7) is a MLKL downstream target for the mediation of Ca2+ influx and TNF-induced necroptosis. Hence, our study reveals a crucial mechanism of MLKL-mediated TNF-induced necroptosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Change history
16 December 2013
In the version of this article originally published the 'Anti-FLAG' label at the top-right of the right panel in Fig. 1b should have read 'Anti-GFP'. This error has now been corrected in the online versions of the Article.
References
Chen, G. & Goeddel, D. V. TNF-R1 signaling: a beautiful pathway. Science 296, 1634–1635 (2002).
Ashkenazi, A. & Dixit, V. M. Apoptosis control by death and decoy receptors. Curr. Opin. Cell Biol. 11, 255–260 (1999).
Grivennikov, S. I., Greten, F. R. & Karin, M. Immunity, inflammation, and cancer. Cell 140, 883–899 (2010).
Vandenabeele, P., Galluzzi, L., Vanden Berghe, T. & Kroemer, G. Molecular mechanisms of necroptosis: an ordered cellular explosion. Nat. Rev. Mol. Cell Biol. 11, 700–714 (2010).
Christofferson, D. E. & Yuan, J. Necroptosis as an alternative form of programmed cell death. Curr. Opin. Cell Biol. 22, 263–268 (2010).
Green, D. R., Oberst, A., Dillon, C. P., Weinlich, R. & Salvesen, G.S. RIPK-dependent necrosis and its regulation by caspases: a mystery in five acts. Mol. Cell 44, 9–16 (2011).
Cho, Y. S. et al. Phosphorylation-driven assembly of the RIP1-RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell 137, 1112–1123 (2009).
He, S. et al. Receptor interacting protein kinase-3 determines cellular necrotic response to TNF-alpha. Cell 137, 1100–1111 (2009).
Zhang, D. W. et al. RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science 325, 332–336 (2009).
Sun, L. et al. Mixed lineage kinase domain-like protein mediates necrosis signaling downstream of RIP3 kinase. Cell 148, 213–227 (2012).
Zhao, J. et al. Mixed lineage kinase domain-like is a key receptor interacting protein 3 downstream component of TNF-induced necrosis. Proc. Natl Acad. Sci. USA 109, 5322–5327 (2012).
O’Donnell, M. A. et al. Caspase 8 inhibits programmed necrosis by processing CYLD. Nat. Cell Biol. 13, 1437–1442 (2011).
Narayan, N. et al. The NAD-dependent deacetylase SIRT2 is required for programmed necrosis. Nature 492, 199–204 (2012).
Mocarski, E. S., Upton, J. W. & Kaiser, W. J. Viral infection and the evolution of caspase 8-regulated apoptotic and necrotic death pathways. Nat. Rev. Immunol. 12, 79–88 (2012).
Welz, P. S. et al. FADD prevents RIP3-mediated epithelial cell necrosis and chronic intestinal inflammation. Nature 477, 330–334 (2011).
Zhang, H. et al. Functional complementation between FADD and RIP1 in embryos and lymphocytes. Nature 471, 373–376 (2011).
Oberst, A. et al. Catalytic activity of the caspase-8-FLIP(L) complex inhibits RIPK3-dependent necrosis. Nature 471, 363–367 (2011).
Lin, Y. et al. Tumor necrosis factor-induced nonapoptotic cell death requires receptor-interacting protein-mediated cellular reactive oxygen species accumulation. J. Biol. Chem. 279, 10822–10828 (2004).
Zong, W. X. & Thompson, C. B. Necrotic death as a cell fate. Genes Dev. 20, 1–15 (2006).
Wang, Z., Jiang, H., Chen, S., Du, F. & Wang, X. The mitochondrial phosphatase PGAM5 functions at the convergence point of multiple necrotic death pathways. Cell 148, 228–243 (2012).
Wolf, E., Kim, P. S. & Berger, B. MultiCoil: a program for predicting two- and three-stranded coiled coils. Protein Sci. 6, 1179–1189 (1997).
Degterev, A. et al. Chemical inhibitor of nonapoptotic cell death with therapeutic potential for ischemic brain injury. Nat. Chem. Biol. 1, 112–119 (2005).
Li, J. et al. The RIP1/RIP3 necrosome forms a functional amyloid signaling complex required for programmed necrosis. Cell 150, 339–350 (2012).
Oancea, E., Teruel, M. N., Quest, A. F. & Meyer, T. Green fluorescent protein (GFP)-tagged cysteine-rich domains from protein kinase C as fluorescentindicators for diacylglycerol signaling in living cells. J. Cell Biol. 140, 485–498 (1998).
Aarts, M. et al. A key role for TRPM7 channels in anoxic neuronal death. Cell 115, 863–877 (2003).
McNulty, S. & Fonfria, E. The role of TRPM channels in cell death. Pflugers Arch. 451, 235–242 (2005).
Bates-Withers, C., Sah, R. & Clapham, D.E. TRPM7, the Mg(2+) inhibited channel and kinase. Adv. Exp. Med. Biol. 704, 173–183 (2011).
Tian, L. et al. Imaging neural activity in worms, flies and mice with improved GCaMP calcium indicators. Nat. Methods 6, 875–881 (2009).
Murphy, J. M. et al. The pseudokinase MLKL mediates necroptosis via a molecular switch mechanism. Immunity 39, 443–453 (2013).
Sciacca, M. F. et al. Cations as switches of amyloid-mediatedmembrane disruption mechanisms: calcium and IAPP. Biophys. J. 104, 173–184 (2013).
Desai, B. N. et al. Cleavage of TRPM7 releases the kinase domain from the ion channel and regulates its participation in Fas-induced apoptosis. Dev. Cell 22, 1149–1162 (2012).
Newton, K., Sun, X. & Dixit, V. M. Kinase RIP3 is dispensable for normal NF-kappa Bs, signaling by the B-cell and T-cell receptors, tumor necrosis factor receptor 1, and Toll-like receptors 2 and 4. Mol. Cell Biol. 24, 1464–1469 (2004).
Wu, J. et al. Mlkl knockout mice demonstrate the indispensable role of Mlkl in necroptosis. Cell Res. 23, 994–1006 (2013).
Acknowledgements
We thank J. Han for MLKL-deficient MEF cells and MLKL antibody, and L. Looger for GCaMP3 plasmid. This research was supported by the Intramural Research Program of the Center for Cancer Research, National Cancer Institute, National Institutes of Health.
Author information
Authors and Affiliations
Contributions
Z.C. and Z-G.L. conceived the study. Z.C. and S.J. did most of the experimental work. J.Z. did some confocal imaging experiments and cell death assays using MEF cells and TRPM7 shRNA JurkatFADD−/− cells. L-g.W. and H-C.C. designed and performed whole-cell patch clamp recording. Y.W. helped with confocal microscopy. J.L. generated MEF cells from RIP3-deficient mice and performed analysis. Z.C., S.C. and Z-G.L. wrote the paper. Z-G.L. supervised the study. All authors discussed the results and implications and commented on the manuscript at all stages.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
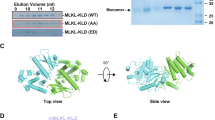
Supplementary Figure 1 Structure determination and assembly pattern prediction of coiled-coil domain, related to Fig. 1.
(a) Sequence alignment of the N-terminal coiled-coil domain of MLKL from vertebrate species. Two distinctive heptad repeat pattern (abcdefg)n characteristic of a coiled-coil architecture are indicated above the alignment. Coiled-coil residues occupying hydrophobic ‘a’ and ‘d’ position are denoted by yellow and red. These coiled-coil motifs are commonly shared among the MLKL orthologs in different vertebrate species. (b) Prediction of oligomerization states of coiled-coil domain of MLKL by Multicoil program (http://groups.csail.mit.edu/cb/multicoil/cgi-bin/multicoil.cgi). The score for potential coiled-coils with a dimeric probability is indicated by blue line and a trimeric probability is indicated by red line. The sum of these two probabilities is the total probability for forming a coiled-coil, which is indicated by black line.
Supplementary Figure 2 Quantification of plasma membrane translocation of DsRed-MLKL in response to TSZ treatment, related to Fig. 4d.
(a-c) MLKL shRNA HT29 cells transfected with DsRed tagged WT MLKL (a), CC1 (b) or CC2 (c) mutants and then treated with TSZ as indicated. A line intensity profile across the cell was obtained in a given image. Representative intensity profiles are shown in the right. Y-axis, Gray level; X-axis, Line distance. (d) The image shown in (a) was used for a representative example to show how to calculate a relative ratio of flurorescence intensity between plasma membrane adjacent area and cytosolic area. The ratio [R] was calculated by dividing the plasma membrane intensity [Ipm] by the average cytosolic fluorescence intensity [Icyt]. Scale bar, 5 μm.
Supplementary Figure 3 Subcellular localization of endogenous MLKL and RIP3 in HT29 cells.
Control shRNA (left panel) or MLKL shRNA (right panel) HT29 cells were treated with TSZ as indicated. Subcellular localization of (a) MLKL or (b) RIP3 was detected by confocal imaging of anti-MLKL antibody or anti-RIP3 antibody staining, respectively. Cell morphology was shown by F-actin probe phalloidin staining. DAPI was used for nuclear staining. White arrows indicate membrane localization. Scale bar, 10 μm.
Supplementary Figure 4 Cell surface protein isolation in MEF cells and J774A.1 cells.
MEF cells (a) or J774A.1 cells (b) were treated with TSZ as indicated. Total cellular lysate and the biotinylated, cell surface fraction were resolved on reducing gel and analyzed by immunoblot as indicated. Na+/K+ ATPase was used as a positive control for transmembrane/surface protein while Actin, COX IV and Calnexin were used as a positive control for cytosolic, mitochondrial and ER membrane proteins, respectively. Uncropped images of western blots are shown in Supplementary Fig. 8.
Supplementary Figure 5 MLKL-mediated Calcium influx is involved in plasma membrane rupture during necroptosis, related to Fig. 6.
(a) Flow cytometric analysis of intracellular Ca2+ of control shRNA, MLKL shRNA or RIP3 shRNA HT29 cells after treated with TSZ for 4 h with/out NSA or Nec-1 as indicated. Cells were harvested and the Fluo4 fluorescent cells were determined by FACS analysis using Fluo4. (b) HT29 cells were treated with TSZ either in normal DMEM or calcium free DMEM at different time points as indicated. The cell lysates were resolved on non-reducing gel and analyzed by immunoblot with anti-MLKL, anti RIP3, anti-RIP1 or anti-Actin antibodies. * indicates phosphorylated RIP3. (c) Representative image of live HT29 MLKL shRNA cells co-expressing DsRed-MLKL together with RIP3-EYFP treated with TSZ at time 0 and 4 hours in calcium free DMEM. Scale bar, 5 μm. Uncropped images of western blots are shown in Supplementary Fig. 8.
Supplementary Figure 6 Extracellular calcium is involved in necroptosis in JurkatFADD−/− and J774A.1 cells.
JurkatFADD−/− or J774A.1 cells were cultured with/out calcium and treated with DMSO control or TSZ as indicated. (a), (c) Cell survival was determined by PI staining. Results shown are averages ± s.e.m. from three independent experiments. *P<0.05, **P<0.01. Statistics source data for Supplementary Fig. 6 can be found in Supplementary Table 2. (b), (d) JurkatFADD−/− or J774A.1 cells expressing control shRNA, MLKL shRNA or cells cultured in calcium free medium were treated with TSZ as indicated. Cells were harvested and the Fluo4 fluorescent cells were determined by FACS analysis using Fluo4. (e), (f) Immunoblot analysis of JuarkatFADD−/− (e) or J774A.1 (f) clones expressing control-shRNA or MLKL-shRNA with anti-MLKL or anti-Actin antibodies. Uncropped images of western blots are shown in Supplementary Fig. 8.
Supplementary Figure 7 ROS and calcium are both required for necroptosis in U937 cells.
(a) U937 cells were either cultured in calcium free medium or cultured in calcium free medium and pre-treated with NAC (5 mM). Then, the cells were treated with TSZ. After 10 hours, cell survival was determined by PI staining. Results shown are averages of triplicates ± SEM. (b) U937 cells cultured in calcium free DMEM were treated with TSZ as indicated. Cells were harvested and the Fluo4 fluorescent cells were determined by FACS analysis using Fluo4. (c) Immunoblot analysis of U937 clones expressing control-shRNA or MLKL-shRNA with anti-MLKL or anti-Actin antibodies. * indicates non-specific band. **,P<0.01. Statistics source data for Supplementary Fig. 7 can be found in Supplementary Table 1. Uncropped images of western blots are shown in Supplementary Fig. 8.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1708 kb)
Supplementary Table 1
Supplementary Information (XLSX 27 kb)
Representative time-lapse GCaMP3 fluorescence imaging of control-shRNA HT29 cells treated with TSZ for 8 hours.
Left, GFP channel; Middle, differential interference contrast channel (DIC); Right, Merged two channels. (MOV 2256 kb)
Representative time-lapse GCaMP3 fluorescence imaging of TRPM7-sh#1RNA HT29 cells treated with TSZ for 8 hours.
Left, GFP channel; Middle, differential interference contrast channel (DIC); Right, Merged two channels. (MOV 3095 kb)
Representative time-lapse GCaMP3 fluorescence imaging of control-shRNA HT29 cells cultured in calcium free DMEM and treated with TSZ for 8 hours.
Left, GFP channel; Middle, differential interference contrast channel (DIC); Right, Merged two channels. (MOV 3031 kb)
Rights and permissions
About this article
Cite this article
Cai, Z., Jitkaew, S., Zhao, J. et al. Plasma membrane translocation of trimerized MLKL protein is required for TNF-induced necroptosis. Nat Cell Biol 16, 55–65 (2014). https://doi.org/10.1038/ncb2883
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2883
This article is cited by
-
Saracatinib inhibits necroptosis and ameliorates psoriatic inflammation by targeting MLKL
Cell Death & Disease (2024)
-
MLKL polymerization-induced lysosomal membrane permeabilization promotes necroptosis
Cell Death & Differentiation (2024)
-
Mediators of necroptosis: from cell death to metabolic regulation
EMBO Molecular Medicine (2024)
-
LUBAC-mediated M1 Ub regulates necroptosis by segregating the cellular distribution of active MLKL
Cell Death & Disease (2024)
-
The role of TRPV4 in programmed cell deaths
Molecular Biology Reports (2024)