Abstract
During cell division, spindle microtubules attach to chromosomes through kinetochores, protein complexes on the chromosome1. The central question is how microtubules find kinetochores. According to the pioneering idea termed search-and-capture, numerous microtubules grow from a centrosome in all directions and by chance capture kinetochores2,3,4. The efficiency of search-and-capture can be improved by a bias in microtubule growth towards the kinetochores5,6, by nucleation of microtubules at the kinetochores7,8,9 and at spindle microtubules10,11, by kinetochore movement9, or by a combination of these processes12,13,14. Here we show in fission yeast that kinetochores are captured by microtubules pivoting around the spindle pole, instead of growing towards the kinetochores. This pivoting motion of microtubules is random and independent of ATP-driven motor activity. By introducing a theoretical model, we show that the measured random movement of microtubules and kinetochores is sufficient to explain the process of kinetochore capture. Our theory predicts that the speed of capture depends mainly on how fast microtubules pivot, which was confirmed experimentally by speeding up and slowing down microtubule pivoting. Thus, pivoting motion allows microtubules to explore space laterally, as they search for targets such as kinetochores.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cheeseman, I. M. & Desai, A. Molecular architecture of the kinetochore-microtubule interface. Nat. Rev. Mol. Cell Biol. 9, 33–46 (2008).
Mitchison, T. J. & Kirschner, M. W. Properties of the kinetochore in vitro. II. Microtubule capture and ATP-dependent translocation. J. Cell Biol. 101, 766–777 (1985).
Hill, T. L. Theoretical problems related to the attachment of microtubules to kinetochores. Proc. Natl Acad. Sci. USA 82, 4404–4408 (1985).
Holy, T. E. & Leibler, S. Dynamic instability of microtubules as an efficient way to search in space. Proc. Natl Acad. Sci. USA 91, 5682–5685 (1994).
Carazo-Salas, R. E. et al. Generation of GTP-bound Ran by RCC1 is required for chromatin-induced mitotic spindle formation. Nature 400, 178–181 (1999).
Wollman, R. et al. Efficient chromosome capture requires a bias in the ‘search-and-capture’ process during mitotic-spindle assembly. Curr. Biol. 15, 828–832 (2005).
Witt, P. L., Ris, H. & Borisy, G. G. Origin of kinetochore microtubules in Chinese hamster ovary cells. Chromosoma 81, 483–505 (1980).
Kitamura, E. et al. Kinetochores generate microtubules with distal plus ends: their roles and limited lifetime in mitosis. Dev. Cell 18, 248–259 (2010).
Paul, R. et al. Computer simulations predict that chromosome movements and rotations accelerate mitotic spindle assembly without compromising accuracy. Proc. Natl Acad. Sci. USA 106, 15708–15713 (2009).
Burbank, K. S., Groen, A. C., Perlman, Z. E., Fisher, D. S. & Mitchison, T. J. A new method reveals microtubule minus ends throughout the meiotic spindle. J. Cell Biol. 175, 369–375 (2006).
Mahoney, N. M., Goshima, G., Douglass, A. D. & Vale, R. D. Making microtubules and mitotic spindles in cells without functional centrosomes. Curr. Biol. 16, 564–569 (2006).
Mogilner, A. & Craig, E. Towards a quantitative understanding of mitotic spindle assembly and mechanics. J. Cell Sci. 123, 3435–3445 (2010).
O’Connell, C. B. & Khodjakov, A. L. Cooperative mechanisms of mitotic spindle formation. J. Cell Sci. 120, 1717–1722 (2007).
Duncan, T. & Wakefield, J. G. 50 ways to build a spindle: the complexity of microtubule generation during mitosis. Chromosome Res. 19, 321–333 (2011).
Funabiki, H., Hagan, I., Uzawa, S. & Yanagida, M. Cell cycle-dependent specific positioning and clustering of centromeres and telomeres in fission yeast. J. Cell Biol. 121, 961–976 (1993).
Sagolla, M. J., Uzawa, S. & Cande, W. Z. Individual microtubule dynamics contribute to the function of mitotic and cytoplasmic arrays in fission yeast. J. Cell Sci. 116, 4891–4903 (2003).
Zimmerman, S., Daga, R. R. & Chang, F. Intra-nuclear microtubules and a mitotic spindle orientation checkpoint. Nat. Cell Biol. 6, 1245–1246 (2004).
Gachet, Y. et al. Sister kinetochore recapture in fission yeast occurs by two distinct mechanisms, both requiring Dam1 and Klp2. Mol. Biol. Cell 19, 1646–1662 (2008).
Grishchuk, E. L. & McIntosh, J. R. Microtubule depolymerization can drive poleward chromosome motion in fission yeast. EMBO J. 25, 4888–4896 (2006).
Tanaka, K. et al. Molecular mechanisms of kinetochore capture by spindle microtubules. Nature 434, 987–994 (2005).
Beinhauer, J. D., Hagan, I. M., Hegemann, J. H. & Fleig, U. Mal3, the fission yeast homologue of the human APC-interacting protein EB-1 is required for microtubule integrity and the maintenance of cell form. J. Cell Biol. 139, 717–728 (1997).
Busch, K. E. & Brunner, D. The microtubule plus end-tracking proteins mal3p and tip1p cooperate for cell-end targeting of interphase microtubules. Curr. Biol. 14, 548–559 (2004).
Berg, H. C. Random Walks in Biology (Princeton Univ. Press, 1993).
Masuda, H., Hirano, T., Yanagida, M. & Cande, W. Z. In vitro reactivation ofspindle elongation in fission yeast nuc2 mutant cells. J. Cell Biol. 110, 417–425 (1990).
Lee, G. M. Characterization of mitotic motors by their relative sensitivity to AMP-PNP. J. Cell Sci. 94, 425–441 (1989).
Broersma, S. Rotational diffusion constant of a cylindrical particle. J. Chem. Phys. 32, 1626–1631 (1960).
Hunt, A. J., Gittes, F. & Howard, J. The force exerted by a single kinesin molecule against a viscous load. Biophys. J. 67, 766–781 (1994).
Tirado, M. M. & de la Torre, J. G. Translational friction coefficients of rigid, symmetric top macromolecules. Application to circular cylinders. J. Chem. Phys. 71, 2581–2587 (1979).
Drummond, D. R. & Cross, R. A. Dynamics of interphase microtubules in Schizosaccharomyces pombe. Curr. Biol. 10, 766–775 (2000).
Vogel, S. K., Raabe, I., Dereli, A., Maghelli, N. & Tolic-Norrelykke, I. Interphase microtubules determine the initial alignment of the mitotic spindle. Curr. Biol. 17, 438–444 (2007).
Gehlen, L. R. et al. Nuclear geometry and rapid mitosis ensure asymmetric episome segregation in yeast. Curr. Biol. 21, 25–33 (2011).
Shav-Tal, Y. et al. Dynamics of single mRNPs in nuclei of living cells. Science 304, 1797–1800 (2004).
Gopalakrishnan, M. & Govindan, B. S. A first-passage-time theory for search and capture of chromosomes by microtubules in mitosis. Bull. Math. Biol. 73, 2483–2506 (2011).
Ding, R., McDonald, K. L. & McIntosh, J. R. Three-dimensional reconstruction and analysis of mitotic spindles from the yeast, Schizosaccharomyces pombe. J. Cell Biol. 120, 141–151 (1993).
Neumann, F. R. & Nurse, P. Nuclear size control in fission yeast. J. Cell Biol. 179, 593–600 (2007).
Bahler, J. et al. Heterologous modules for efficient and versatile PCR-based gene targeting in Schizosaccharomyces pombe. Yeast 14, 943–951 (1998).
Penkett, C. J., Birtle, Z. E. & Bahler, J. Simplified primer design for PCR-based gene targeting and microarray primer database: two web tools for fission yeast. Yeast 23, 921–928 (2006).
Forsburg, S. L. & Rhind, N. Basic methods for fission yeast. Yeast 23, 173–183 (2006).
Russell, P. & Nurse, P. cdc25+ functions as an inducer in the mitotic control of fission yeast. Cell 45, 145–153 (1986).
Acknowledgements
We thank K. Sawin, A. Haese, Y. Caldarelli, E. Guarino, S. Kearsey and the Yeast Genetic Resource Center for strains and plasmids; B. Schroth-Diez from the Light Microscopy Facility of MPI-CBG for help with microscopy; I. Šarić for the drawings; W. Zachariae, S. Grill, J. Howard, D. Cimini, J. Gregan, M. Žanić, E. Paluch, N. Maghelli, M. Coelho and V. Ananthanarayanan for discussions and advice; the German Research Foundation (DFG) and the Human Frontier Science Program (HFSP) for financial support. M.R.C. was supported by a Marie Curie Intra-European Fellowship and D.R-J. by a Humboldt Research Fellowship for Postdoctoral Researchers.
Author information
Authors and Affiliations
Contributions
I.K. carried out all experiments and data analysis, A.N. performed simulations, P.D., M.R.C. and A.H.K. carried out AMP-PNP and FRAP experiments, D.R-J. analysed the data shown in Supplementary Fig. S3b, A.K. developed the tracking software, B.L. and N.P. developed the theory, and I.M.T-N. and N.P. designed the project and wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 527 kb)
Supplementary Note
Supplementary Information (PDF 560 kb)
Supplementary Table 1
Supplementary Information (XLSX 9 kb)
Supplementary Table 2
Supplementary Information (XLSX 34 kb)
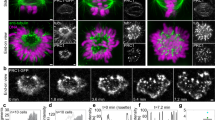
Capture of a lost kinetochore by the tip of a polar microtubule (end-on attachment).
Live cell microscopy of an S. pombe mitotic cell, expressing tubulin labeled with GFP (green), and the kinetochore protein Ndc80p labeled with tdTomato (magenta); strain AH01 (Supplementary Table S1). Images were acquired at 2 s intervals. The video is displayed at 15 fps. Time of recovery after cold stress is indicated in minutes:seconds. Scale bar represents 1 μm. The movie corresponds to Fig. 1c. (AVI 1446 kb)
Capture of a lost kinetochore by the lateral side of a polar microtubule (lateral attachment).
Live cell microscopy of an S. pombe mitotic cell, expressing tubulin labeled with GFP (green), and the kinetochore protein Ndc80p labeled with tdTomato (magenta); strain AH01 (Supplementary Table S1). Images were acquired at 2 s intervals. The video is displayed at 15 fps. Time of recovery after cold treatment is indicated in minutes:seconds. Scale bar represents 1 μm. The movie corresponds to Fig. 1d. (AVI 1086 kb)
Pivoting of polar microtubules around the SPB in a cell expressing Mal3-GFP.
Live cell microscopy of an S. pombe mitotic cell, expressing Mal3-GFP and Sid4 (SPB marker) labeled with GFP; strain YC001 (Supplementary Table S1). Note that Mal3-GFP visualizes the movement of the microtubule tip, which allows us to observe the pivoting of a growing microtubule. Mal3p is not present at the end of shrinking microtubules. Images were acquired at 250 ms intervals. The green line marks the position used to make the kymograph shown in Supplementary Fig. S3a. The video is displayed at 15 fps. Time is indicated in seconds. Scale bar represents 1 μm. (AVI 1005 kb)
Pivoting of polar microtubules in a cell treated with AMP-PNP.
Live cell microscopy of an S. pombe mitotic cell treated with 50 mM AMP-PNP (strain KI061, Supplementary Table S1). Images were acquired at 2.2 s intervals. The video is displayed at 15 fps. Time from the beginning of AMP-PNP treatment is indicated in minutes:seconds. Scale bar represents 1 μm. The movie corresponds to Fig. 2a. (AVI 3733 kb)
Rights and permissions
About this article
Cite this article
Kalinina, I., Nandi, A., Delivani, P. et al. Pivoting of microtubules around the spindle pole accelerates kinetochore capture. Nat Cell Biol 15, 82–87 (2013). https://doi.org/10.1038/ncb2640
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2640
This article is cited by
-
Mechanical Torque Promotes Bipolarity of the Mitotic Spindle Through Multi-centrosomal Clustering
Bulletin of Mathematical Biology (2022)
-
Pivoting of microtubules driven by minus-end-directed motors leads to spindle assembly
BMC Biology (2019)
-
Spatiotemporal control of spindle disassembly in fission yeast
Cellular and Molecular Life Sciences (2019)
-
Mitotic spindle: kinetochore fibers hold on tight to interpolar bundles
European Biophysics Journal (2018)
-
Mitotic spindle assembly in animal cells: a fine balancing act
Nature Reviews Molecular Cell Biology (2017)