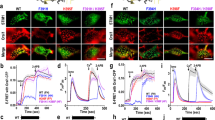
Abstract
Store-operated Ca2+ entry (SOCE) and Ca2+ release-activated Ca2+ currents (Icrac) are strongly suppressed during cell division, the only known physiological situation in which Ca2+ store depletion is uncoupled from the activation of Ca2+ influx. We found that the endoplasmic reticulum (ER) Ca2+ sensor STIM1 failed to rearrange into near-plasma membrane puncta in mitotic cells, a critical step in the SOCE-activation pathway. We also found that STIM1 from mitotic cells is recognized by the phospho-specific MPM-2 antibody, suggesting that STIM1 is phosphorylated during mitosis. Removal of ten MPM-2 recognition sites by truncation at amino acid 482 abolished MPM-2 recognition of mitotic STIM1, and significantly rescued STIM1 rearrangement and SOCE response in mitosis. We identified Ser 486 and Ser 668 as mitosis-specific phosphorylation sites, and STIM1 containing mutations of these sites to alanine also significantly rescued mitotic SOCE. Therefore, phosphorylation of STIM1 at Ser 486 and Ser 668, and possibly other sites, underlies suppression of SOCE during mitosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
12 January 2010
In this letter, statements regarding the suppression of SOCE during mitosis have been altered from those initially published, Two sentences have been altered, an addtional reference added and references in the Methods renumbered. This has been corrected in both the HTML and PDF versions of the article.
References
Berridge, M. J., Bootman, M. D. & Roderick, H. L. Calcium signalling: dynamics, homeostasis and remodelling. Nature Rev. Mol. Cell Biol. 4, 517–529 (2003).
Putney, J. W. A model for receptor-regulated calcium entry. Cell Calcium 7, 1–12 (1986).
Parekh, A. B. & Putney, J. W. Store-operated calcium channels. Physiol. Rev. 85, 757–810 (2005).
Ng, S. W., Di Capite, J., Singaravelu, K. & Parekh, A. B. Sustained activation of the tyrosine kinase Syk by antigen in mast cells requires local Ca2+ influx through Ca2+ release-activated Ca2+ channels. J. Biol. Chem. 283, 31348–31355 (2008).
Parekh, A. B. Ca2+ microdomains near plasma membrane Ca2+ channels: impact on cell function. J. Physiol. 586, 3043–3054 (2008).
Hoth, M. & Penner, R. Depletion of intracellular calcium stores activates a calcium current in mast cells. Nature 355, 353–355 (1992).
Roos, J. et al. STIM1, an essential and conserved component of store-operated Ca2+ channel function. J. Cell Biol. 169, 435–445 (2005).
Liou, J. et al. STIM is a Ca2+ sensor essential for Ca2+-store-depletion-triggered Ca2+ influx. Curr. Biol. 15, 1235–1241 (2005).
Feske, S. et al. A mutation in Orai1 causes immune deficiency by abrogating CRAC channel function. Nature 441, 179–185 (2006).
Vig, M. et al. CRACM1 is a plasma membrane protein essential for store-operated Ca2+ entry. Science 312, 1220–1223 (2006).
Zhang, S. L. et al. Genome-wide RNAi screen of Ca2+ influx identifies genes that regulate Ca2+ release-activated Ca2+ channel activity. Proc. Natl Acad. Sci. USA 103, 9357–9362 (2006).
Zhang, S. L. et al. STIM1 is a Ca2+ sensor that activates CRAC channels and migrates from the Ca2+ store to the plasma membrane. Nature 437, 902–905 (2005).
Wu, M. M., Buchanan, J., Luik, R. M. & Lewis, R. S. Ca2+ store depletion causes STIM1 to accumulate in ER regions closely associated with the plasma membrane. J. Cell Biol. 174, 803–813 (2006).
Lewis, R. S. The molecular choreography of a store-operated calcium channel. Nature 446, 284–287 (2007).
Putney, J. W. Jr New molecular players in capacitative Ca2+ entry. J. Cell Sci. 120, 1959–1965 (2007).
Preston, S. F., Sha'afi, R. I. & Berlin, R. D. Regulation of Ca2+ influx during mitosis: Ca2+ influx and depletion of intracellular Ca2+ stores are coupled in interphase but not mitosis. Cell Regul. 2, 915–925 (1991).
Preston, G. A., Barrett, J. C., Biermann, J. A. & Murphy, E. Effects of alterations in calcium homeostasis on apoptosis during neoplastic progression. Cancer Res. 57, 537–542 (1997).
Volpi, M. & Berlin, R. D. Intracellular elevations of free calcium induced by activation of histamine H1 receptors in interphase and mitotic HeLa cells: hormone signal transduction is altered during mitosis. J. Cell Biol. 107, 2533–2539 (1988).
Tani, D., Monteilh-Zoller, M. K., Fleig, A. & Penner, R. Cell cycle-dependent regulation of store-operated I(CRAC) and Mg2+-nucleotide-regulated MagNuM (TRPM7) currents. Cell Calcium 41, 249–260 (2007).
Hepler, P. K. The role of calcium in cell division. Cell Calcium 16, 322–330 (1994).
Whitaker, M. & Larman, M. G. Calcium and mitosis. Semin. Cell Dev. Biol. 12, 53–58 (2001).
Whitaker, M. Calcium microdomains and cell cycle control. Cell Calcium 40, 585–592 (2006).
Krek, W. & DeCaprio, J. A. Cell synchronization. Methods Enzymol. 254, 114–124 (1995).
Grigoriev, I. et al. STIM1 is a MT-plus-end-tracking protein involved in remodeling of the ER. Curr. Biol. 18, 177–182 (2008).
Smyth, J. T., DeHaven, W. I., Bird, G. S. & Putney, J. W. Jr Role of the microtubule cytoskeleton in the function of the store-operated Ca2+channel activator STIM1. J. Cell Sci. 120, 3762–3771 (2007).
Russa, A. D. et al. Microtubule remodeling mediates the inhibition of store-operated calcium entry (SOCE) during mitosis in COS-7 cells. Arch. Histol. Cytol. 71, 249–263 (2008).
Manji, S. S. et al. STIM1: a novel phosphoprotein located at the cell surface. Biochim. Biophys. Acta 1481, 147–155 (2000).
Williams, R. T. et al. Identification and characterization of the STIM (stromal interaction molecule) gene family: coding for a novel class of transmembrane proteins. Biochem. J. 357, 673–685 (2001).
Che, S., Weil, M. M., Nelman-Gonzalez, M., Ashorn, C. L. & Kuang, J. MPM-2 epitope sequence is not sufficient for recognition and phosphorylation by ME kinase-H. FEBS Lett. 413, 417–423 (1997).
Ding, M., Feng, Y. & Vandre, D. D. Partial characterization of the MPM-2 phosphoepitope. Exp. Cell Res. 231, 3–13 (1997).
Westendorf, J. M., Rao, P. N. & Gerace, L. Cloning of cDNAs for M-phase phosphoproteins recognized by the MPM2 monoclonal antibody and determination of the phosphorylated epitope. Proc. Natl Acad. Sci. USA 91, 714–718 (1994).
Yuan, J. P. et al. SOAR and the polybasic STIM1 domains gate and regulate Orai channels. Nature Cell Biol. (2009).
Park, C. Y. et al. STIM1 clusters and activates CRAC channels via direct binding of a cytosolic domain to Orai1. Cell 136, 876–890 (2009).
Muik, M. et al. A cytosolic homomerization and a modulatory domain within STIM1 C-terminus determine coupling to ORAI1 channels. J. Biol. Chem. 284, 8421–8426 (2009).
Kawasaki, T., Lange, I. & Feske, S. A minimal regulatory domain in the C terminus of STIM1 binds to and activates ORAI1 CRAC channels. Biochem. Biophys. Res. Commun. (2009).
Liou, J., Fivaz, M., Inoue, T. & Meyer, T. Live-cell imaging reveals sequential oligomerization and local plasma membrane targeting of stromal interaction molecule 1 after Ca2+ store depletion. Proc. Natl Acad. Sci. USA 104, 9301–9306 (2007).
Machaca, K. & Huan, S. Store-operated calcium entry inactivates at the germinal vesicle breakdown stage of Xenopus meiosis. J. Biol. Chem. 275, 38710–38715 (2000).
DeHaven, W. I., Smyth, J. T., Boyles, R. R. & Putney, J. W. Calcium inhibition and calcium potentiation of Orai1, Orai2, and Orai3 calcium release-activated calcium channels. J. Biol. Chem. 282, 17548–17556 (2007).
Smyth, J. T., DeHaven, W. I., Bird, G. S. & Putney, J. W. Jr Ca2+-store-dependent and -independent reversal of Stim1 localization and function. J. Cell Sci. 121, 762–772 (2008).
Choi, J. H., Williams, J., Cho, J., Falck, J. R. & Shears, S. B. Purification, sequencing, and molecular identification of a mammalian PP-InsP5 kinase that is activated when cells are exposed to hyperosmotic stress. J.Biol. Chem. 282, 30763–30775 (2007).
Acknowledgements
We thank J. Tucker and H. Rutledge for assistance with confocal microscopy and C. Bortner and M. Sifre for help with flow cytometry. C. Williams and D. Miller reviewed the manuscript and offered helpful suggestions. This work was supported by the Intramural Program, National Institute of Environmental Health Sciences, National Institutes of Health.
Author information
Authors and Affiliations
Contributions
J.T.S. and J.G.P. performed Ca2+ measurements; J.T.S. and W.I.D. performed electrophysiology; J.T.S. performed confocal microscopy; J.T.S. and M.F. performed biochemistry; J.T.S. and R.R.B. performed molecular biology; J.G.W. and K.L.J. performed mass spectrometry. All authors performed data analysis, and J.T.S., W.I.D., J.G.W. and J.W.P. performed project planning.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2041 kb)
Rights and permissions
About this article
Cite this article
Smyth, J., Petranka, J., Boyles, R. et al. Phosphorylation of STIM1 underlies suppression of store-operated calcium entry during mitosis. Nat Cell Biol 11, 1465–1472 (2009). https://doi.org/10.1038/ncb1995
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1995
This article is cited by
-
Neuronal Store-Operated Calcium Channels
Molecular Neurobiology (2023)
-
Phosphorylation of NMDA receptors by cyclin B/CDK1 modulates calcium dynamics and mitosis
Communications Biology (2020)
-
A novel, dynein-independent mechanism focuses the endoplasmic reticulum around spindle poles in dividing Drosophila spermatocytes
Scientific Reports (2019)
-
The STIM1-Orai1 pathway of store-operated Ca2+ entry controls the checkpoint in cell cycle G1/S transition
Scientific Reports (2016)