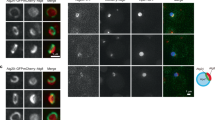
Abstract
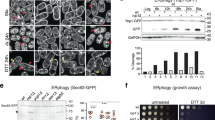
Autophagy is a bulk degradation process in eukaryotic cells and has fundamental roles in cellular homeostasis.The origin and source of autophagosomal membranes are long-standing questions in the field. Using electron microscopy, we show that, in mammalian culture cells, the endoplasmic reticulum (ER) associates with early autophagic structures called isolation membranes (IMs). Overexpression of an Atg4B mutant, which causes defects in autophagosome formation, induces the accumulation of ER–IM complexes. Electron tomography revealed that the ER–IM complex appears as a subdomain of the ER that formed a cradle encircling the IM, and showed that both ER and isolation membranes are interconnected.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Mizushima, N. Autophagy: process and function. Genes Dev. 21, 2861–2873 (2007).
Suzuki, K. & Ohsumi, Y. Molecular machinery of autophagosome formation in yeast, Saccharomyces cerevisiae. FEBS Lett. 581, 2156–2161 (2007).
Yoshimori, T. & Noda, T. Toward unraveling membrane biogenesis in mammalian autophagy. Curr. Opin. Cell Biol. 20, 401–407 (2008).
Juhasz, G. & Neufeld, T. P. Autophagy: a forty-year search for a missing membrane source. PLoS Biol. 4, e36 (2006).
Dunn, W.A. Jr Autophagy and related mechanisms of lysosome-mediated protein degradation. Trends Cell Biol. 4, 139–143 (1994).
Furuno, K. et al. Immunocytochemical study of the surrounding envelope of autophagic vacuoles in cultured rat hepatocytes. Exp. Cell Res. 189, 261–268 (1990).
Yamamoto, A., Masaki, R., Fukui, Y. & Tashiro, Y. Absence of cytochrome P-450 and presence of autolysosomal membrane antigens on the isolation membranes and autophagosomal membranes in rat hepatocytes. J. Histochem. Cytochem. 38, 1571–1581 (1990).
Yamamoto, A., Masaki, R. & Tashiro, Y. Characterization of the isolation membranes and the limiting membranes of autophagosomes in rat hepatocytes by lectin cytochemistry. J. Histochem. Cytochem. 38, 573–580 (1990).
Kovacs, A. L., Palfia, Z., Rez, G., Vellai, T. & Kovacs, J. Sequestration revisited: integrating traditional electron microscopy, de novo assembly and new results. Autophagy 3, 655–662 (2007).
Axe, E. L. et al. Autophagosome formation from membrane compartments enriched in phosphatidylinositol 3-phosphate and dynamically connected to the endoplasmic reticulum. J. Cell Biol. 182, 685–701 (2008).
Walker, S., Chandra, P., Manifava, M., Axe, E. & Ktistakis, N. T. Making autophagosomes: localized synthesis of phosphatidylinositol 3-phosphate holds the clue. Autophagy 4, 1093–1096 (2008).
Fujita, N. et al. An Atg4B mutant hampers the lipidation of LC3 paralogues and causes defects in autophagosome closure. Mol. Biol. Cell 19, 4651–4659 (2008).
Fengsrud, M. et al. Ultrastructural and immunocytochemical characterization of autophagic vacuoles in isolated hepatocytes: effects of vinblastine and asparagine on vacuole distributions. Exp. Cell Res. 221, 504–519 (1995).
Mizushima, N. et al. Dissection of autophagosome formation using Apg5-deficient mouse embryonic stem cells. J. Cell Biol. 152, 657–668 (2001).
Mizushima, N. et al. Mouse Apg16L, a novel WD-repeat protein, targets to the autophagic isolation membrane with the Apg12–Apg5 conjugate. J. Cell Sci. 116, 1679–1688 (2003).
Saitoh, T. et al. Loss of the autophagy protein Atg16L1 enhances endotoxin-induced IL-1β production. Nature 456, 264–268 (2008).
McIntosh, R., Nicastro, D. & Mastronarde, D. New views of cells in 3D: an introduction to electron tomography. Trends Cell Biol. 15, 43–51 (2005).
Bannykh, S. I., Rowe, T. & Balch, W. E. The organization of endoplasmic reticulum export complexes. J. Cell Biol. 135, 19–35 (1996).
Saitoh, T. et al. TWEAK induces NF-kappaB2 p100 processing and long lasting NF-kappaB activation. J. Biol. Chem. 278, 36005–36012 (2003).
Mastronarde, D. N. Dual-axis tomography: an approach with alignment methods that preserve resolution. J. Struct. Biol. 120, 343–352 (1997).
Kremer, J. R., Mastronarde, D. N. & McIntosh, J. R. Computer visualization of three-dimensional image data using IMOD. J. Struct. Biol. 116, 71–76 (1996).
Wakana, Y. et al. Bap31 is an itinerant protein that moves between the peripheral endoplasmic reticulum (ER) and a juxtanuclear compartment related to ER-associated Degradation. Mol. Biol. Cell 19, 1825–1836 (2008).
Acknowledgements
We thank H. Nishioka and N. Kajimura for technical assistance with electron tomography, K. Nishino for assistance with setting up a computer and for useful comments on figure presentations, H. Omori for technical assistance with immunoelectron microscopy and K. Matsunaga and N. Taguchi for technical assistance with plasmid construction. This study was supported by grants from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
Author information
Authors and Affiliations
Contributions
M.H.N., N.F., T.N., T.Y. and A.Y. designed experiments. M.H.N. performed conventional electron microscopy and electron tomography, N.F. performed cell biological and immuofluorescence experiments and A.Y. performed immunoelectron microscopy. M.H.N., T.Y. and A.Y. wrote the manuscript. T.N. and A.Y. contributed to conceptual discussions. A.Y. and T.Y. supervised the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 3010 kb)
Supplementary Movie 1
Supplementary Movie 1 (MOV 5714 kb)
Supplementary Movie 2
Supplementary Movie 2 (MOV 1100 kb)
Supplementary Movie 3
Supplementary Movie 3 (MOV 2845 kb)
Supplementary Movie 4
Supplementary Movie 4 (MOV 2014 kb)
Supplementary Movie 5
Supplementary Movie 5 (MOV 1487 kb)
Supplementary Movie 6
Supplementary Movie 6 (MOV 1758 kb)
Rights and permissions
About this article
Cite this article
Hayashi-Nishino, M., Fujita, N., Noda, T. et al. A subdomain of the endoplasmic reticulum forms a cradle for autophagosome formation. Nat Cell Biol 11, 1433–1437 (2009). https://doi.org/10.1038/ncb1991
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1991
This article is cited by
-
Roles of Neuronal Protein Kinase Cε on Endoplasmic Reticulum Stress and Autophagic Formation in Diabetic Neuropathy
Molecular Neurobiology (2024)
-
Beyond energy provider: multifunction of lipid droplets in embryonic development
Biological Research (2023)
-
The ABL-MYC axis controls WIPI1-enhanced autophagy in lifespan extension
Communications Biology (2023)
-
Cancer cells adapt FAM134B/BiP mediated ER-phagy to survive hypoxic stress
Cell Death & Disease (2022)
-
Recycling of autophagosomal components from autolysosomes by the recycler complex
Nature Cell Biology (2022)