Abstract
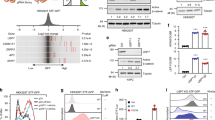
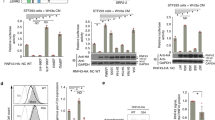
The von Hippel–Lindau protein pVHL suppresses renal tumorigenesis in part by promoting the degradation of hypoxia-inducible HIF-α transcription factors1; additional mechanisms have been proposed2. pVHL also stabilizes the plant homeodomain protein Jade-1, which is a candidate renal tumour suppressor that may correlate with renal cancer risk3,4,5. Here we show that Jade-1 binds the oncoprotein β-catenin in Wnt-responsive fashion. Moreover, Jade-1 destabilizes wild-type β-catenin but not a cancer-causing form of β-catenin. Whereas the well-established β-catenin E3 ubiquitin ligase component β-TrCP ubiquitylates only phosphorylated β-catenin6, Jade-1 ubiquitylates both phosphorylated and non-phosphorylated β-catenin and therefore regulates canonical Wnt signalling in both Wnt-off and Wnt-on phases. Thus, the different characteristics of β-TrCP and Jade-1 may ensure optimal Wnt pathway regulation. Furthermore, pVHL downregulates β-catenin in a Jade-1-dependent manner and inhibits Wnt signalling, supporting a role for Jade-1 and Wnt signalling in renal tumorigenesis. The pVHL tumour suppressor and the Wnt tumorigenesis pathway are therefore directly linked through Jade-1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Maxwell, P. H. et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 399, 271–275 (1999).
Cohen, H. T. & McGovern, F. J. Renal-cell carcinoma. N. Engl. J. Med. 353, 2477–2490 (2005).
Zhou, M. I. et al. The von Hippel–Lindau tumor suppressor stabilizes novel plant homeodomain protein Jade-1. J. Biol. Chem. 277, 39887–39898 (2002).
Zhou, M. I., Wang, H., Foy, R. L., Ross, J. J. & Cohen, H. T. Tumor suppressor von Hippel–Lindau (VHL) stabilization of Jade-1 protein occurs through plant homeodomains and is VHL mutation dependent. Cancer Res. 64, 1278–1286 (2004).
Zhou, M. I. et al. Jade-1, a candidate renal tumor suppressor that promotes apoptosis. Proc. Natl Acad. Sci. USA 102, 11035–11040 (2005).
Winston, J. T. et al. The SCFβ–TRCP–ubiquitin ligase complex associates specifically with phosphorylated destruction motifs in IκBα and β-catenin and stimulates IκBα ubiquitination in vitro. Genes Dev. 13, 270–283 (1999).
Nusse, R. Wnt signaling in disease and in development. Cell Res. 15, 28–32 (2005).
Panchenko, M. V., Zhou, M. I. & Cohen, H. T. von Hippel–Lindau partner Jade-1 is a transcriptional co-activator associated with histone acetyltransferase activity. J. Biol. Chem. 279, 56032–56041 (2004).
Latres, E., Chiaur, D. S. & Pagano, M. The human F box protein β-Trcp associates with the Cul1/Skp1 complex and regulates the stability of β-catenin. Oncogene 18, 849–854 (1999).
Schulz, I. Permeabilizing cells: some methods and applications for the study of intracellular processes. Methods Enzymol. 192, 280–300 (1990).
Aberle, H., Bauer, A., Stappert, J., Kispert, A. & Kemler, R. β-Catenin is a target for the ubiquitin–proteasome pathway. EMBO J. 16, 3797–3804 (1997).
Coscoy, L., Sanchez, D. J. & Ganem, D. A novel class of herpesvirus-encoded membrane-bound E3 ubiquitin ligases regulates endocytosis of proteins involved in immune recognition. J. Cell Biol. 155, 1265–1273 (2001).
Lu, Z., Xu, S., Joazeiro, C., Cobb, M. H. & Hunter, T. The PHD domain of MEKK1 acts as an E3 ubiquitin ligase and mediates ubiquitination and degradation of ERK1/2. Mol. Cell 9, 945–956 (2002).
Belaidouni, N. et al. Overexpression of human β TrCP1 deleted of its F box induces tumorigenesis in transgenic mice. Oncogene 24, 2271–2276 (2005).
McCrea, P. D., Brieher, W. M. & Gumbiner, B. M. Induction of a secondary body axis in Xenopus by antibodies to β-catenin. J. Cell Biol. 123, 477–484 (1993).
Jho, E. H. et al. Wnt/β-catenin/Tcf signaling induces the transcription of Axin2, a negative regulator of the signaling pathway. Mol. Cell. Biol. 22, 1172–1183 (2002).
Iliopoulos, O., Ohh, M. & Kaelin, W. G. Jr. pVHL19 is a biologically active product of the von Hippel–Lindau gene arising from internal translation initiation. Proc. Natl Acad. Sci. USA 95, 11661–11666 (1998).
Hovanes, K. et al. β-Catenin-sensitive isoforms of lymphoid enhancer factor-1 are selectively expressed in colon cancer. Nature Genet. 28, 53–57 (2001).
Larabell, C. A. et al. Establishment of the dorso-ventral axis in Xenopus embryos is presaged by early asymmetries in β-catenin that are modulated by the Wnt signaling pathway. J. Cell Biol. 136, 1123–1136 (1997).
Liu, J. et al. Siah-1 mediates a novel β-catenin degradation pathway linking p53 to the adenomatous polyposis coli protein. Mol. Cell 7, 927–936 (2001).
Nastasi, T. et al. Ozz-E3, a muscle-specific ubiquitin ligase, regulates β-catenin degradation during myogenesis. Dev. Cell 6, 269–282 (2004).
Hart, M. et al. The F-box protein β-TrCP associates with phosphorylated β-catenin and regulates its activity in the cell. Curr. Biol. 9, 207–210 (1999).
Kitagawa, M. et al. An F-box protein, FWD1, mediates ubiquitin-dependent proteolysis of β-catenin. EMBO J. 18, 2401–2410 (1999).
Sansom, O. J., Griffiths, D. F., Reed, K. R., Winton, D. J. & Clarke, A. R. Apc deficiency predisposes to renal carcinoma in the mouse. Oncogene 24, 8205–8210 (2005).
Qian, C. N. et al. Cystic renal neoplasia following conditional inactivation of Apc in mouse renal tubular epithelium. J. Biol. Chem. 280, 3938–3945 (2005).
Saadi-Kheddouci, S. et al. Early development of polycystic kidney disease in transgenic mice expressing an activated mutant of the β-catenin gene. Oncogene 20, 5972–5981 (2001).
Battagli, C. et al. Promoter hypermethylation of tumor suppressor genes in urine from kidney cancer patients. Cancer Res. 63, 8695–8699 (2003).
Peruzzi, B., Athauda, G. & Bottaro, D. P. The von Hippel–Lindau tumor suppressor gene product represses oncogenic β-catenin signaling in renal carcinoma cells. Proc. Natl Acad. Sci. USA 103, 14531–14536 (2006).
Simons, M. et al. Inversin, the gene product mutated in nephronophthisis type II, functions as a molecular switch between Wnt signaling pathways. Nature Genet. 37, 537–543 (2005).
Nieuwkoop, J. & Faber, J. Normal Table of Xenopus laevis (North-Holland, Amsterdam, 1967).
Dominguez, I. et al. Protein kinase CK2 is required for dorsal axis formation in Xenopus embryos. Dev. Biol. 274, 110–124 (2004).
Kao, K. R. & Elinson, R. P. The entire mesodermal mantle behaves as Spemann's organizer in dorsoanterior enhanced Xenopus laevis embryos. Dev. Biol. 127, 64–77 (1988).
Acknowledgements
We thank Z.-X. Xiao (Boston University) for insightful suggestions and careful review of the manuscript; K. Symes, M. Malikova and E. Smith (all of Boston University) for Xenopus laevis embryos; R. Kemler (Max Planck Institute for Immunobiology, Germany) for providing the β-catenin S33A construct; W. Birchmeier (Max Delbruck Center for Molecular Medicine, Germany) for β-catenin C and N terminus deletion constructs; and R. Benarous (Institute Pasteur, France) for wild-type and DN β-TrCP in pcDNA3.1 Myc/His vector. This work was supported by fellowship grants from the National Kidney Foundation and Polycystic Kidney Disease Foundation (to V.C.C.) and by National Institutes of Health (NIH) Training Grant T32 DK07053 (for V.C.C. and R.L.F.); by American Heart Association grant SDG 0535485T and American Cancer Society grant IRG-72-001-32-IRG (to M.V.P.); by a pilot research grant from the Department of Medicine at Boston University School of Medicine and a Karin Grunebaum Junior Faculty Cancer Research Award (to I.D.); and by NIH grants R01 CA71796 (to D.C.S.) and R01 CA79830 and R01 DK67569 (to H.T.C.). Part of this work was presented at the American Society of Nephrology annual meeting in San Diego, California, USA, in November 2006, and at the American Society of Nephrology annual meeting in San Francisco, California, USA, in November 2007.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2887 kb)
Rights and permissions
About this article
Cite this article
Chitalia, V., Foy, R., Bachschmid, M. et al. Jade-1 inhibits Wnt signalling by ubiquitylating β-catenin and mediates Wnt pathway inhibition by pVHL. Nat Cell Biol 10, 1208–1216 (2008). https://doi.org/10.1038/ncb1781
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1781
This article is cited by
-
New insights in ubiquitin-dependent Wnt receptor regulation in tumorigenesis
In Vitro Cellular & Developmental Biology - Animal (2024)
-
RENGE infers gene regulatory networks using time-series single-cell RNA-seq data with CRISPR perturbations
Communications Biology (2023)
-
Identification of a novel Calpain-2-SRC feed-back loop as necessity for β-Catenin accumulation and signaling activation in hepatocellular carcinoma
Oncogene (2022)
-
Genome-wide association study and functional validation implicates JADE1 in tauopathy
Acta Neuropathologica (2022)
-
WNK regulates Wnt signalling and β-Catenin levels by interfering with the interaction between β-Catenin and GID
Communications Biology (2020)