Abstract
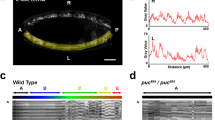
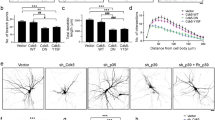
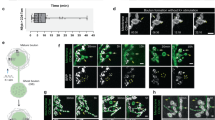
Dendrites allow neurons to integrate sensory or synaptic inputs, and the spatial disposition and local density of branches within the dendritic arbor limit the number and type of inputs1,2. Drosophila melanogaster dendritic arborization (da) neurons provide a model system to study the genetic programs underlying such geometry in vivo. Here we report that mutations of motor-protein genes, including a dynein subunit gene (dlic) and kinesin heavy chain (khc), caused not only downsizing of the overall arbor, but also a marked shift of branching activity to the proximal area within the arbor. This phenotype was suppressed when dominant-negative Rab5 was expressed in the mutant neurons, which deposited early endosomes in the cell body. We also showed that 1) in dendritic branches of the wild-type neurons, Rab5-containing early endosomes were dynamically transported and 2) when Rab5 function alone was abrogated, terminal branches were almost totally deleted. These results reveal an important link between microtubule motors and endosomes in dendrite morphogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Jan, Y. N. & Jan, L. Y. The control of dendrite development. Neuron 40, 229–242 (2003).
London, M. & Hausser, M. Dendritic computation. Annu. Rev. Neurosci. 28, 503–532 (2005).
Grueber, W. B., Jan, L. Y. & Jan, Y. N. Tiling of the Drosophila epidermis by multidendritic sensory neurons. Development 129, 2867–2878 (2002).
Lee, T. & Luo, L. Mosaic analysis with a repressible cell marker for studies of gene function in neuronal morphogenesis. Neuron 22, 451–461 (1999).
Goldstein, L. S. & Gunawardena, S. Flying through the Drosophila cytoskeletal genome. J. Cell Biol. 150, 63–68 (2000).
Vale, R. D. The molecular motor toolbox for intracellular transport. Cell 112, 467–480 (2003).
Mesngon, M. T. et al. Regulation of cytoplasmic dynein ATPase by Lis1. J. Neurosci. 26, 2132–2139 (2006).
Bowman, A. B. et al. Drosophila roadblock and Chlamydomonas LC7: a conserved family of dynein-associated proteins involved in axonal transport, flagellar motility, and mitosis. J. Cell Biol. 146, 165–180 (1999).
Liu, Z., Steward, R. & Luo, L. Drosophila Lis1 is required for neuroblast proliferation, dendritic elaboration and axonal transport. Nature Cell Biol. 2, 776–783 (2000).
Rolls, M. M. et al. Polarity and intracellular compartmentalization of Drosophila neurons. Neural Develop. 2, 7 (2007).
Stowers, R. S., Megeath, L. J., Gorska-Andrzejak, J., Meinertzhagen, I. A. & Schwarz, T. L. Axonal transport of mitochondria to synapses depends on milton, a novel Drosophila protein. Neuron 36, 1063–1077 (2002).
Zerial, M. & McBride, H. Rab proteins as membrane organizers. Nature Rev. Mol. Cell Biol. 2, 107–117 (2001).
Welte, M. A. Bidirectional transport along microtubules. Curr. Biol. 14, 525–537 (2004).
Driskell, O. J., Mironov, A., Allan, V. J. & Woodman, P. G. Dynein is required for receptor sorting and the morphogenesis of early endosomes. Nature Cell Biol. 9, 113–120 (2007).
Wucherpfennig, T., Wilsch-Brauninger, M. & Gonzalez-Gaitan, M. Role of Drosophila Rab5 during endosomal trafficking at the synapse and evoked neurotransmitter release. J. Cell Biol. 161, 609–624 (2003).
Shin, H. W. et al. An enzymatic cascade of Rab5 effectors regulates phosphoinositide turnover in the endocytic pathway. J. Cell Biol. 170, 607–618 (2005).
Entchev, E. V., Schwabedissen, A. & Gonzalez-Gaitan, M. Gradient formation of the TGF-β homolog Dpp. Cell 103, 981–991 (2000).
Shimizu, H., Kawamura, S. & Ozaki, K. An essential role of Rab5 in uniformity of synaptic vesicle size. J. Cell Sci. 116, 3583–3590 (2003).
Hirokawa, N. & Takemura, R. Molecular motors and mechanisms of directional transport in neurons. Nature Rev. Neurosci. 6, 201–214 (2005).
Goldstein, L. S. Do disorders of movement cause movement disorders and dementia? Neuron 40, 415–425 (2003).
He, Y. et al. Role of cytoplasmic dynein in the axonal transport of microtubules and neurofilaments. J. Cell Biol. 168, 697–703 (2005).
Miki, H., Setou, M., Kaneshiro, K. & Hirokawa, N. All kinesin superfamily protein, KIF, genes in mouse and human. Proc. Natl Acad. Sci. USA 98, 7004–7011 (2001).
Brendza, R. P., Serbus, L. R., Saxton, W. M. & Duffy, J. B. Posterior localization of dynein and dorsal-ventral axis formation depend on kinesin in Drosophila oocytes. Curr. Biol. 12, 1541–1545 (2002).
Lenz, J. H., Schuchardt, I., Straube, A. & Steinberg, G. A dynein loading zone for retrograde endosome motility at microtubule plus-ends. EMBO J. 25, 2275–2286 (2006).
Ye, B. et al. Growing dendrites and axons differ in their reliance on the secretory pathway. Cell 130, 717–729 (2007).
Miaczynska, M., Pelkmans, L. & Zerial, M. Not just a sink: endosomes in control of signal transduction. Curr. Opin. Cell Biol. 16, 400–406 (2004).
Sweeney, N. T., Brenman, J. E., Jan, Y. N. & Gao, F. B. The coiled-coil protein shrub controls neuronal morphogenesis in Drosophila. Curr. Biol. 16, 1006–1011 (2006).
Gruenberg, J. & Stenmark, H. The biogenesis of multivesicular endosomes. Nature Rev. Mol. Cell Biol. 5, 317–323 (2004).
Pfister, K. K. et al. Genetic analysis of the cytoplasmic dynein subunit families. PLoS Genet. 2, e1 (2006).
Sugimura, K. et al. Distinct developmental modes and lesion-induced reactions of dendrites of two classes of Drosophila sensory neurons. J. Neurosci. 23, 3752–3760 (2003).
Brechbiel, J. L. & Gavis, E. R. Spatial regulation of nanos is required for its function in dendrite morphogenesis. Curr. Biol. 18, 745–750 (2008).
Wu, M. N. et al. Syntaxin 1A interacts with multiple exocytic proteins to regulate neurotransmitter release in vivo. Neuron 23, 593–605 (1999).
Funayama, R., Saito, M., Tanobe, H. & Ishikawa, F. Loss of linker histone H1 in cellular senescence. J. Cell Biol. 175, 869–880 (2006).
Acknowledgements
The reagents were provided by the Developmental Studies Hybridoma Bank at the University of Iowa, the Bloomington Stock Center, the Drosophila Genetic Resource Center at Kyoto Institute of Technology, J. Knoblich, D. Bilder, L. Luo, Y. N. Jan, H. Shimizu and W. Saxton. We thank Yuh-Nung Jan for communicating data before publication. We are grateful to K. Sugimura, K. Kousaka, T. Kiyomitsu, C. Obuse, M. Sone, Y. Okada, E. Gavis and M. Ohno for their technical advice and encouragement; T. Harumoto and S. Yonehara for allowing us to use their equipment; Y. Miyake and M. Futamata for their technical assistance. This work was supported by grants from the programs Grants-in-Aid for Scientific Research on Priority Areas-Molecular Brain Science (17024025 to T.U.) and for Cancer Research (to F.I. and M.S.) of the MEXT of Japan, by a Wellcome Trust Senior Research Fellowship (to H.O.), and by a NARSAD Young Investigator Award (to M.M.R.). D.S is a recipient of a Fellowship of the Japan Society for the Promotion of Science for Junior Scientists.
Author information
Authors and Affiliations
Contributions
D.Satoh carried out most of the experiments; D.Sato, T.T., M.S. and F.I. assisted with some experiments; H.O. and M.M.R. supplied reagents. D.Satoh and T.U. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4, S5, S6, S7, S8 and Supplementary Methods (PDF 2647 kb)
Supplementary Information
Supplementary Movie 1 (MOV 5837 kb)
Supplementary Information
Supplementary Movie 2 (MOV 5369 kb)
Supplementary Information
Supplementary Movie 3 (MOV 8705 kb)
Supplementary Information
Supplementary Movie 4 (MOV 7524 kb)
Supplementary Information
Supplementary Movie 5 (MOV 2073 kb)
Supplementary Information
Supplementary Movie 6 (MOV 4420 kb)
Supplementary Information
Supplementary Movie 7 (MOV 4420 kb)
Rights and permissions
About this article
Cite this article
Satoh, D., Sato, D., Tsuyama, T. et al. Spatial control of branching within dendritic arbors by dynein-dependent transport of Rab5-endosomes. Nat Cell Biol 10, 1164–1171 (2008). https://doi.org/10.1038/ncb1776
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1776
This article is cited by
-
The Exocyst Component Exo70 Modulates Dendrite Arbor Formation, Synapse Density, and Spine Maturation in Primary Hippocampal Neurons
Molecular Neurobiology (2019)
-
Competition between microtubule-associated proteins directs motor transport
Nature Communications (2018)
-
Regulation of long-distance transport of mitochondria along microtubules
Cellular and Molecular Life Sciences (2018)
-
N-acetyl-D-glucosamine kinase interacts with dynein light-chain roadblock type 1 at Golgi outposts in neuronal dendritic branch points
Experimental & Molecular Medicine (2015)
-
Drosophila Strip serves as a platform for early endosome organization during axon elongation
Nature Communications (2014)