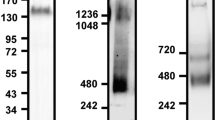
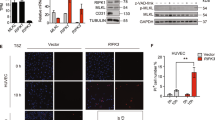
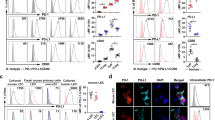
Abstract
During inflammation, leukocytes bind to the adhesion receptors ICAM-1 and VCAM-1 on the endothelial surface before undergoing transendothelial migration, also called diapedesis. ICAM-1 is also involved in transendothelial migration, independently of its role in adhesion, but the molecular basis of this function is poorly understood. Here we demonstrate that, following clustering, apical ICAM-1 translocated to caveolin-rich membrane domains close to the ends of actin stress fibres. In these F-actin-rich areas, ICAM-1 was internalized and transcytosed to the basal plasma membrane through caveolae. Human T-lymphocytes extended pseudopodia into endothelial cells in caveolin- and F-actin-enriched areas, induced local translocation of ICAM-1 and caveolin-1 to the endothelial basal membrane and transmigrated through transcellular passages formed by a ring of F-actin and caveolae. Reduction of caveolin-1 levels using RNA interference (RNAi) specifically decreased lymphocyte transcellular transmigration. We propose that the translocation of ICAM-1 to caveola- and F-actin-rich domains links the sequential steps of lymphocyte adhesion and transendothelial migration and facilitates lymphocyte migration through endothelial cells from capillaries into surrounding tissue.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Butcher, E. C. Leukocyte–endothelial cell recognition: three (or more) steps to specificity and diversity. Cell 67, 1033–1036 (1991).
Johnson-Leger, C., Aurrand-Lions, M. & Imhof, B. A. The parting of the endothelium: miracle, or simply a junctional affair? J. Cell Sci. 113, 921–933 (2000).
Burns, A. R. et al. Neutrophil transendothelial migration is independent of tight junctions and occurs preferentially at tricellular corners. J. Immunol. 159, 2893–2903 (1997).
Burns, A. R. et al. Analysis of tight junctions during neutrophil transendothelial migration. J. Cell Sci. 113, 45–57 (2000).
Oppenheimer-Marks, N. et al. Differential utilization of ICAM-1 and VCAM-1 during the adhesion and transendothelial migration of human T lymphocytes. J. Immunol. 147, 2913–2921 (1991).
Schenkel, A. R., Mamdouh, Z. & Muller, W. A. Locomotion of monocytes on endothelium is a critical step during extravasation. Nature Immunol. 5, 393–400 (2004).
Muller, W. A. Leukocyte–endothelial-cell interactions in leukocyte transmigration and the inflammatory response. Trends Immunol. 24, 327–334 (2003).
Feng, D. et al. Neutrophils emigrate from venules by a transendothelial cell pathway in response to FMLP. J. Exp. Med. 187, 903–915 (1998).
Carman, C. V. & Springer, T. A. A transmigratory cup in leukocyte diapedesis both through individual vascular endothelial cells and between them. J. Cell Biol. 167, 377–388 (2004).
Dvorak, A. M. & Feng, D. The vesiculo-vacuolar organelle (VVO). A new endothelial cell permeability organelle. J. Histochem. Cytochem. 49, 419–432 (2001).
Mundy, D. I. et al. Dual control of caveolar membrane traffic by microtubules and the actin cytoskeleton. J. Cell Sci. 115, 4327–4339 (2002).
Gratton, J. P., Bernatchez, P. & Sessa, W. C. Caveolae and caveolins in the cardiovascular system. Circ. Res. 94, 1408–1417 (2004).
Carman, C. V., Jun, C. D., Salas, A. & Springer, T. A. Endothelial cells proactively form microvilli-like membrane projections upon intercellular adhesion molecule 1 engagement of leukocyte LFA-1. J. Immunol. 171, 6135–6144 (2003).
Barreiro, O. et al. Dynamic interaction of VCAM-1 and ICAM-1 with moesin and ezrin in a novel endothelial docking structure for adherent leukocytes. J. Cell Biol. 157, 1233–1245 (2002).
Thompson, P. W., Randi, A. M. & Ridley, A. J. Intercellular adhesion molecule (ICAM)-1, but not ICAM-2, activates RhoA and stimulates c-fos and rhoA transcription in endothelial cells. J. Immunol. 169, 1007–1013 (2002).
Etienne-Manneville, S. et al. ICAM-1-coupled cytoskeletal rearrangements and transendothelial lymphocyte migration involve intracellular calcium signaling in brain endothelial cell lines. J. Immunol. 165, 3375–3383 (2000).
Kiemer, A. K. et al. Inhibition of p38 MAPK activation via induction of MKP-1: atrial natriuretic peptide reduces TNF-α-induced actin polymerization and endothelial permeability. Circ. Res. 90, 874–881 (2002).
Feng, D., Nagy, J. A., Dvorak, H. F. & Dvorak, A. M. Ultrastructural studies define soluble macromolecular, particulate, and cellular transendothelial cell pathways in venules, lymphatic vessels, and tumor-associated microvessels in man and animals. Microsc. Res. Tech. 57, 289–326 (2002).
Toomre, D. & Manstein, D. J. Lighting up the cell surface with evanescent wave microscopy. Trends Cell Biol. 11, 298–303 (2001).
Steyer, J. A. & Almers, W. A real-time view of life within 100 nm of the plasma membrane. Nature Rev. Mol. Cell Biol. 2, 268–275 (2001).
Vasile, E., Simionescu, M. & Simionescu, N. Visualization of the binding, endocytosis, and transcytosis of low-density lipoprotein in the arterial endothelium in situ. J. Cell Biol. 96, 1677–1689 (1983).
Pelkmans, L., Puntener, D. & Helenius, A. Local actin polymerization and dynamin recruitment in SV40-induced internalization of caveolae. Science 296, 535–539 (2002).
Kvietys, P. R. & Sandig, M. Neutrophil diapedesis: paracellular or transcellular? News Physiol. Sci. 16, 15–19 (2001).
Engelhardt, B. & Wolburg, H. Transendothelial migration of leukocytes: through the front door or around the side of the house? Eur. J. Immunol. 34, 2955–2963 (2004).
Makinen, T. et al. Isolated lymphatic endothelial cells transduce growth, survival and migratory signals via the VEGF-C/D receptor VEGFR-3. EMBO J. 20, 4762–4773 (2001).
Sotgia, F. et al. Intracellular retention of glycosylphosphatidyl inositol-linked proteins in caveolin-deficient cells. Mol. Cell Biol. 22, 3905–3926 (2002).
Yang, L. et al. ICAM-1 regulates neutrophil adhesion and transcellular migration of TNF-I activated vascular endothelium under flow. Blood 106, 584–592 (2005).
Wojciak-Stothard, B., Williams, L. & Ridley, A. J. Monocyte adhesion and spreading on human endothelial cells is dependent on Rho-regulated receptor clustering. J. Cell Biol. 145, 1293–1307 (1999).
Heiska, L. et al. Association of ezrin with intercellular adhesion molecule-1 and -2 (ICAM-1 and ICAM-2). Regulation by phosphatidylinositol 4, 5-bisphosphate. J. Biol. Chem. 273, 21893–21900 (1998).
Stahlhut, M. & van Deurs, B. Identification of filamin as a novel ligand for caveolin-1: evidence for the organization of caveolin-1-associated membrane domains by the actin cytoskeleton. Mol. Biol. Cell 11, 325–337 (2000).
Parton, R. G. Caveolae — from ultrastructure to molecular mechanisms. Nature Rev. Mol. Cell Biol. 4, 162–167 (2003).
Simionescu, M., Gafencu, A. & Antohe, F. Transcytosis of plasma macromolecules in endothelial cells: a cell biological survey. Microsc. Res. Tech. 57, 269–288 (2002).
Pelkmans, L. et al. Genome-wide analysis of human kinases in clathrin- and caveolae/raft-mediated endocytosis. Nature 436, 78–86 (2005).
Fielding, C. J. Caveolae and signaling. Curr. Opin. Lipidol. 12, 281–287 (2001).
Wary, K. K., Mariotti, A., Zurzolo, C. & Giancotti, F. G. A requirement for caveolin-1 and associated kinase Fyn in integrin signaling and anchorage-dependent cell growth. Cell 94, 625–634 (1998).
Tilghman, R. W. & Hoover, R. L. The Src-cortactin pathway is required for clustering of E-selectin and ICAM-1 in endothelial cells. FASEB J. 16, 1257–1259 (2002).
Michaely, P. A., Mineo, C., Ying, Y. S. & Anderson, R. G. Polarized distribution of endogenous Rac1 and RhoA at the cell surface. J. Biol. Chem. 274, 21430–21436 (1999).
Etienne, S. et al. ICAM-1 signaling pathways associated with Rho activation in microvascular brain endothelial cells. J. Immunol. 161, 5755–5761 (1998).
Millan, J. et al. Lipid rafts mediate biosynthetic transport to the T lymphocyte uropod subdomain and are necessary for uropod integrity and function. Blood 99, 978–984 (2002).
Toomre, D. et al. Fusion of constitutive membrane traffic with the cell surface observed by evanescent wave microscopy. J. Cell Biol. 149, 33–40 (2000).
Simionescu, N. & Simionescu, M. Galloylglucoses of low molecular weight as mordant in electron microscopy. I. Procedure, and evidence for mordanting effect. J. Cell Biol. 70, 608–621 (1976).
Beardsley, A. et al. Loss of caveolin-1 polarity impedes endothelial cell polarization and directional movement. J. Biol. Chem. 280, 3541–3547 (2005).
Acknowledgements
This work was supported by the Ludwig Institute for Cancer Research and European Community contracts QLG1-CT-99-01036 and FP6–502935. J. Millán was supported by a Marie Curie fellowship (no. HPMF-CT-2000-01061) and British Heart Foundation intermediate fellowship (no. FS/04/006). We are grateful to the named donors for the gifts of plasmids and antibodies listed in the methods section, to E. Cernuda Morollon for providing T-lymphoblasts, and to members of the Ridley laboratory for helpful discussions. We thank Olympus for generously providing instrumentation and support to the Yale CINEMA lab.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3, S4 and Methods (PDF 855 kb)
Supplementary Information
Supplementary Movie 1 (MOV 3002 kb)
Supplementary Information
Supplementary Movie 2 (MOV 1306 kb)
Supplementary Information
Supplementary Movie 3 (MOV 2638 kb)
Supplementary Information
Supplementary Movie 4 (MOV 3080 kb)
Rights and permissions
About this article
Cite this article
Millán, J., Hewlett, L., Glyn, M. et al. Lymphocyte transcellular migration occurs through recruitment of endothelial ICAM-1 to caveola- and F-actin-rich domains. Nat Cell Biol 8, 113–123 (2006). https://doi.org/10.1038/ncb1356
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1356
This article is cited by
-
An Intriguing Structural Modification in Neutrophil Migration Across Blood Vessels to Inflammatory Sites: Progress in the Core Mechanisms
Cell Biochemistry and Biophysics (2024)
-
Lymphatic trafficking of immune cells and insights for cancer metastasis
Clinical & Experimental Metastasis (2023)
-
Lack of adducin impairs the stability of endothelial adherens and tight junctions and may be required for cAMP-Rac1-mediated endothelial barrier stabilization
Scientific Reports (2022)
-
Plasmolipin regulates basolateral-to-apical transcytosis of ICAM-1 and leukocyte adhesion in polarized hepatic epithelial cells
Cellular and Molecular Life Sciences (2022)
-
Appearance of claudin-5+ leukocyte subtypes in the blood and CNS during progression of EAE
Journal of Neuroinflammation (2021)