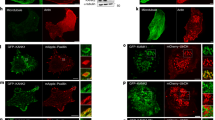
Abstract
Unconventional myosins are actin-based motors with a growing number of attributed functions1. Interestingly, it has been proposed that integrins are transported by unidentified myosins to facilitate cellular remodelling2. Here we present an interaction between the unconventional myosin-X (Myo10) FERM (band 4.1/ezrin/radixin/moesin) domain and an NPXY motif within β-integrin cytoplasmic domains. Importantly, knock-down of Myo10 by short interfering RNA impaired integrin function in cell adhesion, whereas overexpression of Myo10 stimulated the formation and elongation of filopodia in an integrin-dependent manner and relocalized integrins together with Myo10 to the tips of filopodia. This integrin relocalization and filopodia elongation did not occur with Myo10 mutants deficient in integrin binding or with a β1-integrin point mutant deficient in Myo10 binding. Taken together, these results indicate that Myo10-mediated relocalization of integrins might serve to form adhesive structures and thereby promote filopodial extension.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Berg, J.S., Powell, B.C. & Cheney, R.E. A millennial myosin census. Mol. Biol. Cell. 12, 780–794 (2001).
Vuori, K. & Ruoslahti, E. Connections count in cell migration. Nature Cell Biol. 1, E85–E87 (1999).
Grabham, P.W., Foley, M., Umeojiako, A. & Goldberg, D.J. Nerve growth factor stimulates coupling of β1 integrin to distinct transport mechanisms in the filopodia of growth cones. J. Cell Sci. 113 3003–3012 (2000).
Mermall, V., Post, P.L. & Mooseker, M.S. Unconventional myosins in cell movement, membrane traffic, and signal transduction. Science 279, 527–533 (1998).
Berg, J.S., Derfler, B.H., Pennisi, C.M., Corey, D.P. & Cheney, R.E. Myosin-X, a novel myosin with pleckstrin homology domains, associates with regions of dynamic actin. J. Cell Sci. 113, 3439–3451 (2000).
Berg, J.S. & Cheney, R.E. Myosin-X is an unconventional myosin that undergoes intrafilopodial motility. Nature Cell Biol. 4, 246–250 (2002).
Zhang, H., Li, Z., Viklund, E.K. & Strömblad, S. P21-activated kinase 4 interacts with integrin αvβ5 and regulates αvβ5-mediated cell migration. J. Cell Biol. 158, 1287–1297 (2002).
Mangeat, P., Roy, C. & Martin, M. ERM proteins in cell adhesion and membrane dynamics. Trends Cell Biol. 9, 187–192 (1999).
Ylänne, J. Conserved functions of the cytoplasmic domains of integrin β subunits. Frontiers Biosci. 3, 877–886 (1998).
Laflamme, S.E., Homan, S.M., Bodeau, A.L. & Mastrangelo, A.M. Integrin cytoplasmic domains as connectors to the cell's signal transduction apparatus. Matrix Biol. 16, 153–163 (1997).
Garcia-Alvarez, B. et al. Structural determinants of integrin recognition by talin. Mol. Cell. 11, 49–58 (2003).
Filardo, E.J., Brooks, P.C., Deming, S.L., Damsky, C. & Cheresh, D.A. Requirement of the NPXY motif in the integrin β3 subunit cytoplasmic tail for melanoma cell migration in vitro and in vivo. J. Cell Biol. 130 441–450 (1995).
Pearson, M.A., Reczek, D., Bretscher, A. & Karplus, P.A. Structure of the ERM protein moesin reveals the FERM domain fold masked by an extended actin binding tail domain. Cell 101, 259–270 (2000).
Calderwood, D.A. et al. The phosphotyrosine binding-like domain of talin activates integrins. J. Biol. Chem. 277, 21749–21758 (2002).
Calderwood, D.A. et al. Integrin β cytoplasmic domain interactions with phosphotyrosine-binding domains: a structural prototype for diversity in integrin signaling. Proc. Natl Acad. Sci. USA 100, 2272–2277 (2003).
Forman-Kay, J.D. & Pawson, T. Diversity in protein recognition by PTB domains. Curr. Opin. Struct. Biol. 9, 690–695 (1999).
Calderwood, D.A. et al. The talin head domain binds to integrin β subunit cytoplasmic tails and regulates integrin activation. J. Biol. Chem. 274, 28071–28074 (1999).
Steketee, M., Balazovich, K. & Tosney, K.W. Filopodial initiation and a novel filament-organizing center, the focal ring. Mol. Biol. Cell 12, 2378–2395 (2001).
Otey, C.A., Pavalko, F.M. & Burridge, K. An interaction between α-actinin and the β1 integrin subunit in vitro. J. Cell Biol. 111 721–729 (1990).
Horwitz, A., Duggan, K., Buck, C., Beckerle, M.C. & Burridge, K. Interaction of plasma membrane fibronectin receptor with talin—a transmembrane linkage. Nature 320, 531–533 (1986).
Pankov, R. et al. Specific β1integrin site selectively regulates Akt/protein kinase B signaling via local activation of protein phosphatase 2A. J. Biol. Chem. 278, 18671–1881 (2003).
Small, J.V., Stradal, T., Vignal, E. & Rottner, K. The lamellipodium: where motility begins. Trends Cell Biol. 12, 112–120 (2002).
Gerhardt, H. et al. VEGF guides angiogenic sprouting utilizing endothelial tip cell filopodia. J. Cell Biol. 161, 1163–1177 (2003).
Strömblad, S. & Cheresh, D.A. Cell adhesion and angiogenesis. Trends Cell Biol. 6, 462–468 (1996).
Soldi, R., Mitola, S., Strasly, M., Defilippi, P., Tarone, G. & Bussolino, F. Role of αvβ3 integrin in the activation of vascular endothelial growth factor receptor-2. EMBO J. 18, 882–892 (1999).
Tuxworth, R.I. et al. A role of myosin VII in dynamic cell adhesion. Curr. Biol. 11, 318–329 (2001).
Liu, X. et al. Mutations in the myosin VIIA gene cause non-syndromic recessive deafness. Nature Genet. 16, 188–190 (1997).
Wang, A. et al. Association of unconventional myosin MYO15 mutations with human nonsyndromic deafness DFNB3. Science 280, 1447–1451 (1998).
Littlewood-Evans, A. & Müller, U. Stereocilia defects in the sensory hair cells of the inner ear in mice deficient in integrin α8β1 . Nature Genet. 24, 424–428 (2000).
Jenkins, A.L. et al. Tyrosine phosphorylation of the β3 cytoplasmic domain mediates integrin–cytoskeletal interactions. J. Biol. Chem. 273, 13878–13885 (1998).
Acknowledgements
We thank E. Ruoslahti for providing the integrin β1 and β5 cDNA, K. M. Yamada and R. Pankov for providing GD25, GD25-β1-wt and GD25-β1-W775A cells, R. Agami for the pSuper vector, and A. Hofman, E.-K. Viklund and P. Lennartsson for technical assistance. We also thank the Center for Infectious Medicine for use of their confocal microscope supported by the Swedish Strategic Foundation. This study was supported by grants to S.S. from the Swedish Cancer Society and the Swedish Research Council, to H.Z. from the Swedish Society of Medicine and to R.E.C. from NIH (no. DC03299). H.Z. was supported by the Wenner-Gren Foundation in Sweden. S.S. holds a senior scientist position from the Swedish Research Council.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
Fig. 1, Fig. 2, Fig. 3 and Fig. 4 (PDF 482 kb)
Rights and permissions
About this article
Cite this article
Zhang, H., Berg, J., Li, Z. et al. Myosin-X provides a motor-based link between integrins and the cytoskeleton. Nat Cell Biol 6, 523–531 (2004). https://doi.org/10.1038/ncb1136
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1136
This article is cited by
-
Rosmarinic acid in combination with ginsenoside Rg1 suppresses colon cancer metastasis via co-inhition of COX-2 and PD1/PD-L1 signaling axis
Acta Pharmacologica Sinica (2024)
-
Macropinocytosis requires Gal-3 in a subset of patient-derived glioblastoma stem cells
Communications Biology (2021)
-
Platelet integrin αIIbβ3: signal transduction, regulation, and its therapeutic targeting
Journal of Hematology & Oncology (2019)
-
Phenotypic analysis of Myo10 knockout (Myo10tm2/tm2) mice lacking full-length (motorized) but not brain-specific headless myosin X
Scientific Reports (2019)
-
Myosin IIA and formin dependent mechanosensitivity of filopodia adhesion
Nature Communications (2019)