Abstract
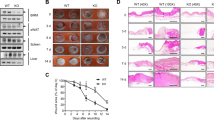
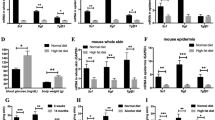
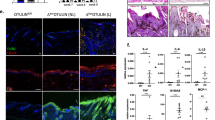
The generation of animals lacking SMAD proteins, which transduce signals from transforming growth factor-β (TGF-β), has made it possible to explore the contribution of the SMAD proteins to TGF-β activity in vivo. Here we report that, in contrast to predictions made on the basis of the ability of exogenous TGF-β to improve wound healing, Smad3-null (Smad3ex8/ex8) mice paradoxically show accelerated cutaneous wound healing compared with wild-type mice, characterized by an increased rate of re-epithelialization and significantly reduced local infiltration of monocytes. Smad3ex8/ex8 keratinocytes show altered patterns of growth and migration, and Smad3ex8/ex8 monocytes exhibit a selectively blunted chemotactic response to TGF-β. These data are, to our knowledge, the first to implicate Smad3 in specific pathways of tissue repair and in the modulation of keratinocyte and monocyte function in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Massague, J. TGF-beta signal transduction. Annu. Rev. Biochem. 67, 753–791 (1998).
Derynck, R., Zhang, Y. & Feng, X. H. Smads: transcriptional activators of TGF-beta responses. Cell 95, 737–740 (1998).
Roberts, A. B. Transforming growth factor-β: activity and efficacy in animal models of wound healing. Wound Repair Regen. 3, 408–418 (1995).
O"Kane, S. & Ferguson, M. W. J. Transforming growth factor beta s and wound healing. Int. J. Biochem. Cell Biol. 29, 63–78 (1997).
Yang, X. et al. Targeted disruption of SMAD3 results in impaired mucosal immunity and diminished T cell responsiveness to TGF-beta. EMBO J. 18, 1280–1291 (1999).
Datto, M. B. et al. Targeted disruption of Smad3 reveals an essential role in transforming growth factor beta-mediated signal transduction. Mol. Cell Biol. 19, 2495–2504 (1999).
Zhu, Y., Richardson, J. A., Parada, L. F., & Graff, J. M. Smad3 mutant mice develop metastatic colorectal cancer. Cell 18, 703–714 (1998).
Weinstein, M., Yang, X., Li, C., Xu, X., & Deng, C. Failure of extraembryonic membrane formation and mesoderm induction in embryos lacking the tumor suppressor Smad2. Proc. Natl Acad. Sci. USA 95, 9378–9383 (1998).
Ashcroft, G. S. et al. Estrogen accelerates cutaneous wound healing associated with an increase in TGF-beta1 levels. Nature Med. 3, 1209–1215 (1997).
Gross, J. et al. On the mechanism of skin wound “contraction”: a granulation tissue “knockout” with a normal phenotype. Proc. Natl Acad. Sci. USA 92, 5982–5986 (1995).
Wahl, S. M. et al. Transforming growth factor type beta induces monocyte chemotaxis and growth factor production. Proc. Natl Acad. Sci. USA 84, 5788–5792 (1987).
Leibovich, S. J. & Ross, R. The role of the macrophage in wound repair. A study with hydrocortisone and antimacrophage serum. Am. J. Pathol. 78, 71–100 (1975).
McCartney-Francis, N., & Wahl, S. M. Transforming growth factor beta: a matter of life and death. J. Leuk. Biol. 55, 401–409 (1994).
Pierce, G. F. et al. Transforming growth factor beta reverses the glucocorticoid-induced wound-healing deficit in rats: possible regulation in macrophages by platelet-derived growth factor. Proc. Natl Acad. Sci. USA 86, 2229–2233 (1989).
Vindevoghel, L. et al. SMAD3/4-dependent transcriptional activation of the human type VII collagen gene (COL7A1) promoter by transforming growth factor beta. Proc. Natl Acad. Sci. USA 95, 14769–14774 (1998).
Chen, S. J. et al. Stimulation of type I collagen transcription in human skin fibroblasts by TGF-beta: involvement of Smad3. J. Invest. Dermatol. 112, 49–57 (1999).
Hocevar, B. A., Brown, T. L. & Howe, P. H. TGF-beta induces fibronectin synthesis through a c-Jun N-terminal kinase-dependent, Smad4-independent pathway. EMBO J. 18, 1345–1356 (1999).
Wiseman, D. M., Polverini, P. J., Kamp, D. W. & Leibovich, S. J. Transforming growth factor-beta (TGF beta) is chemotactic for human monocytes and induces their expression of angiogenic activity. Biochem. Biophys. Res. Commun. 157, 793–800 (1988).
Wahl, S. M., Allen, J. B., Weeks, B. S., Wong, H. L. & Klotman, P. E. Transforming growth factor beta enhances integrin expression and type IV collagenase secretion in human monocytes. Proc. Natl Acad. Sci. USA 90, 4577–4581 (1993).
Zambruno, G. et al. Transforming growth factor-beta 1 modulates beta 1 and beta 5 integrin receptors and induces the de novo expression of the alpha v beta 6 heterodimer in normal human keratinocytes: implications for wound healing. J. Cell Biol. 129, 853–865 (1995).
Mustoe, T. A., Pierce, G. F., Morishima, C. & Deuel, T. F. Growth factor-induced acceleration of tissue repair through direct and inductive activities in a rabbit dermal ulcer model. J. Clin. Invest. 87, 694–703 (1991).
Hebda, P. A. Stimulatory effects of transforming growth factor-beta and epidermal growth factor on epidermal cell outgrowth from porcine skin explant cultures. J. Invest. Dermatol. 91, 440–445 (1988).
Yanagisawa, K. et al. Induction of apoptosis by Smad3 and down-regulation of Smad3 expression in response to TGF-beta in human normal lung epithelial cells. Oncogene 17, 1743–1747 (1998).
Dennler, S., Huet, S. & Gauthier, J. M. A short amino-acid sequence in MH1 domain is responsible for functional differences between Smad2 and Smad3. Oncogene 18, 1643–1648 (1999).
Ulloa, L., Doody, J. & Massague, J. Inhibition of transforming growth factor-beta/SMAD signalling by the interferon-gamma/STAT pathway. Nature 397, 710–713 (1999).
Yanagisawa, J. et al. Convergence of transforming growth factor-beta and vitamin D signaling pathways on SMAD transcriptional coactivators. Science 283, 1317–1321 (1999).
Kurokawa, M. et al. The oncoprotein Evi-1 represses TGF-beta signalling by inhibiting Smad3. Nature 2, 92–96 (1998).
de Caestecker, M. P. et al. Smad2 transduces common signals from receptor serine-threonine and tyrosine kinases. Genes Dev. 12, 587–592 (1998).
Kretzschmar, M. et al. A mechanism of repression of TGFbeta/Smad signaling by oncogenic Ras. Genes Dev. 1, 804–816 (1999).
Liu, X. et al. Transforming growth factor beta-induced phosphorylation of Smad3 is required for growth inhibition and transcriptional induction in epithelial cells. Proc. Natl Acad. Sci. USA 94, 10669–10674 (1997).
Feldman, G. et al. STAT5A-deficient mice demonstrate a defect in granulocyte-macrophage colony-stimulating factor-induced proliferation and gene expression. Blood 90, 1768–1776 (1997).
Dlugosz, A. A., Glick, A. B., Tennenbaum, T., Weinberg, W. C. & Yuspa, S. H. Isolation and utilization of epidermal keratinocytes for oncogene research. Methods Enzymol. 254, 3–20 (1995).
Danielpour, D. et al. Immunodetection and quantitation of the two forms of transforming growth factor-beta (TGF-beta 1 and TGF-beta 2) secreted by cells in culture. J. Cell Physiol. 138, 79–86 (1989).
Acknowledgements
G.S.A. was supported by a Clinician Scientist Fellowship from the Wellcome Trust. We thank L. Hansen for help with the keratinocyte assays.
Correspondence and requests for materials should be addressed to A.B.R..
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ashcroft, G., Yang, X., Glick, A. et al. Mice lacking Smad3 show accelerated wound healing and an impaired local inflammatory response. Nat Cell Biol 1, 260–266 (1999). https://doi.org/10.1038/12971
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/12971
This article is cited by
-
Antiviral drugs prolong survival in murine recessive dystrophic epidermolysis bullosa
EMBO Molecular Medicine (2024)
-
Transcriptomic and in vivo approaches introduced human iPSC-derived microvesicles for skin rejuvenation
Scientific Reports (2023)
-
3D collagen migration patterns reveal a SMAD3-dependent and TGF-β1-independent mechanism of recruitment for tumour-associated fibroblasts in lung adenocarcinoma
British Journal of Cancer (2023)
-
Smad3 regulates smooth muscle cell fate and mediates adverse remodeling and calcification of the atherosclerotic plaque
Nature Cardiovascular Research (2022)
-
The complexity of TGFβ/activin signaling in regeneration
Journal of Cell Communication and Signaling (2021)