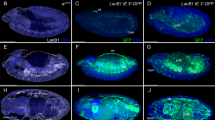
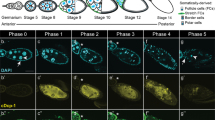
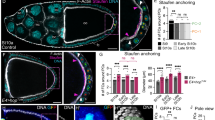
Abstract
Nuclear lamins are intermediate filaments that compose the nuclear lamina — the filamentous meshwork underlying the inner nuclear membrane — and are required for nuclear assembly, organization and maintenance. Here we present evidence that a nuclear lamin is also required for cytoplasmic organization in two highly polarized cell types. Zygotic loss-of-function mutations in the Drosophila gene encoding the principal lamin (Dm0) disrupt the directed outgrowth of cytoplasmic extensions from terminal cells of the tracheal system. Germline mutant clones disrupt dorsal–ventral polarity of the oocyte. In mutant oocytes, transcripts of the dorsal determinant Gurken, a transforming growth factor-α homologue, fail to localize properly around the anterodorsal surface of the oocyte nucleus; their ventral spread results in dorsalized eggs that resemble those of the classical dorsalizing mutations squid and fs(1)K10. The requirement of a nuclear lamin for cytoplasmic as well as nuclear organization has important implications for both the cellular functions of lamins and the pathogenesis of human diseases caused by lamin mutations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Samakovlis, C. et al. Development 122, 1395–1407 (1996).
Guillemin, K. et al. Development 122, 1353–1362 (1996).
Gruenbaum, Y. et al. J Cell Biol 106, 585–596 (1988).
Brand, A. H. & Perrimon, N. Development 118, 401–415 (1993).
Chou, T. B. & Perrimon, N. Genetics 144, 1673–1679 (1996).
Cohen, M., Lee, K. K., Wilson, K. L. & Gruenbaum, Y. Trends Biochem. Sci. 26, 41–47 (2001).
Lenz, B. B. et al. J. Cell Biol. 137, 1001–1016 (1997).
Liu, J. et al. Mol. Biol. Cell 11, 3937–3947 (2000).
Sullivan, T. et al. J. Cell Biol. 147, 913–920 (1999).
Wilson, K. L., Zastrow, M. S. & Lee, K. K. Cell 104, 647–650 (2001).
Nilson, L. A. & Schupbach, T. Curr. Top. Dev. Biol. 44, 203–243 (1999).
Roth, S., Neuman, S. F., Barcelo, G. & Schupbach, T. Cell 81, 967–978 (1995).
Roth, S. & Schupbach, T. Development 120, 2245–2257 (1994).
Peri, F., Bokel, C. & Roth, S. Mech. Dev. 81, 75–88 (1999).
Serano, T. L., Karlin-McGinness, M. & Cohen, R. S. Mech. Dev. 51, 183–192 (1995).
Marshall, W. F. & Sedat, J. W. Results Probl. Cell Differ. 25, 283–301 (1999).
Saunders, C. & Cohen, R. S. Mol. Cell 3, 43–54 (1999).
Norvell, A., Kelley, R. L., Wehr, K. & Schupbach, T. Genes Dev. 13, 864–876 (1999).
Maniotis, A. J., Chen, C. S. & Ingber, D. E. Proc. Natl Acad. Sci. USA 94, 849–854 (1997).
Fey, E. G., Wan, K. M. & Penman, S. J. Cell Biol. 98, 1973–1984 (1984).
Fatkin, D. et al. N. Engl. J. Med. 341, 1715–1724 (1999).
Bonne, G. et al. Ann. Neurol. 48, 170–180 (2000).
Wilson, K. L. Trends Cell Biol. 10, 125–129 (2000).
Fulton, A. B. J. Cell Biochem. 52, 148–152 (1993).
Mitsui, T. et al. J. Neuropathol. Exp. Neurol. 56, 94–101 (1997).
Toniolo, D. & Minetti, C. Curr. Opin. Genet. Dev. 9, 275–282 (1999).
Spradling, A. C. et al. Proc. Natl Acad. Sci. USA 92, 10824–10830 (1995).
Szidonya, J. & Reuter, G. Genet. Res. 51, 197–208 (1988).
Verheyen, E. & Cooley, L. in Drosophila melanogaster: Practical Uses in Cell and Molecular Biology (eds Goldstein, L. S. B. & Fyrberg, E. A.) 545–561 (Academic, New York, 1994).
Kopczynski, C. C., Davis, G. W. & Goodman, C. S. Science 271, 1867–1870 (1996).
Perrimon, N., Noll, E., McCall, K. & Brand, A. Dev. Genet. 12, 238–252 (1991).
Osman, M., Paz, M., Landesman, Y., Fainsod, A. & Gruenbaum, Y. Genomics 8, 217–224 (1990).
McGrail, M. & Hays, T. S. Development 124, 2409–2419 (1997).
Acknowledgements
We thank P. Fisher, T. Schubach and N. Perrimon for antibodies and fly strains; and I. Davis, Y. Gruenbaum, J. Sedat, K. Wilson, and members of the Krasnow lab for helpful discussions. This work was supported by a grant from the NIH. M.A.K. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Guillemin, K., Williams, T. & Krasnow, M. A nuclear lamin is required for cytoplasmic organization and egg polarity in Drosophila. Nat Cell Biol 3, 848–851 (2001). https://doi.org/10.1038/ncb0901-848
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb0901-848
This article is cited by
-
Laminopathies: what can humans learn from fruit flies
Cellular & Molecular Biology Letters (2018)
-
Non-farnesylated B-type lamin can tether chromatin inside the nucleus and its chromatin interaction requires the Ig-fold region
Chromosoma (2017)
-
The lamin Dm0 allele Ari3 acts as an enhancer of position effect variegation of the w m4 allele in Drosophila
Genetica (2007)
-
Lamin B1 duplications cause autosomal dominant leukodystrophy
Nature Genetics (2006)
-
Laminopathies: Multiple disorders arising from defects in nuclear architecture
Journal of Biosciences (2006)