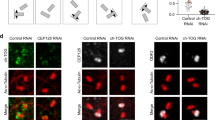
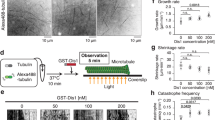
Abstract
The XMAP215/ch-TOG/Msps family of microtubule-associated proteins (MAPs) promote microtubule growth in vitro and are concentrated at centrosomes in vivo. We show here that Msps (mini-spindles protein) interacts with the centrosomal protein D-TACC, and that this interaction strongly influences microtubule behaviour in Drosophila embryos. If D-TACC levels are reduced, Msps does not concentrate at the centrosomes efficiently and the centrosomal microtubules appear to be destabilized. If D-TACC levels are increased, both D-TACC and Msps accumulate around the centrosomes/spindle poles, and the centrosomal microtubules appear to be stabilized. We show that the interaction between D-TACC and Msps is evolutionarily conserved. We propose that D-TACC and Msps normally cooperate to stabilize centrosomal microtubules by binding to their minus ends and binding to their plus ends as they grow out from the centrosome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Glover, D. M., Gonzalez, C. & Raff, J. W. The centrosome. Sci. Am. 268, 62–68 (1993).
Desai, A. & Mitchison, T. J. Microtubule polymerization dynamics. Annu. Rev. Cell Dev. Biol. 13, 83–117 (1997).
Kellogg, D. R., Moritz, M. & Alberts, B. M. The centrosome and cellular organization. Annu. Rev. Biochem. 63, 639–674 (1994).
Felix, M. A., Antony, C., Wright, M. & Maro, B. Centrosome assembly in vitro: role of γ-tubulin recruitment in Xenopus sperm aster formation. J. Cell Biol. 124, 19–31 (1994).
Joshi, H. C., Palacios, M. J., McNamara, L. & Cleveland, D. W. γ-tubulin is a centrosomal protein required for cell cycle-dependent microtubule nucleation. Nature 356, 80–83 (1992).
Oakley, B. R., Oakley, C. E., Yoon, Y. & Jung, M. K. γ-tubulin is a component of the spindle pole body that is essential for microtubule function in Aspergillus nidulans. Cell 61, 1289–1301 (1990).
Stearns, T. & Kirschner, M. In vitro reconstitution of centrosome assembly and function: the central role of γ-tubulin. Cell 76, 623–637 (1994).
Sunkel, C. E., Gomes, R., Sampaio, P., Perdigao, J. & Gonzalez, C. γ-tubulin is required for the structure and function of the microtubule organizing center in Drosophila neuroblasts. EMBO J. 14, 28–36 (1995).
Zheng, Y. X., Wong, M. L., Alberts, B. & Mitchison, T. Nucleation of microtubule assembly by a γ-tubulin-containing ring complex. Nature 378, 578–583 (1995).
Moritz, M., Braunfeld, M. B., Sedat, J. W., Alberts, B. & Agard, D. A. Microtubule nucleation by γ-tubulin-containing rings in the centrosome. Nature 378, 638–640 (1995).
Erickson, H. P. γ-tubulin nucleation: template or protofilament? Nature Cell Biol. 2, E93–E96 (2000).
Gunawardane, R. N., Lizarraga, S. B., Wiese, C., Wilde, A. & Zheng, Y. γ-tubulin complexes and their role in microtubule nucleation Curr. Top. Dev. Biol. 49, 55–73 (2000).
Compton, D. A. Focusing on spindle poles. J. Cell Sci. 111, 1477–1481 (1998).
Hyman, A. A. & Karsenti, E. Morphogenetic properties of microtubules and mitotic spindle assembly. Cell 84, 401–410 (1996).
Merdes, A. & Cleveland, D. W. Pathways of spindle pole formation: different mechanisms; conserved components. J. Cell Biol. 138, 953–956 (1997).
Cullen, C. F., Deak, P., Glover, D. M. & Ohkura, H. Mini spindles. A gene encoding a conserved microtubule-associated protein required for the integrity of the mitotic spindle in Drosophila. J. Cell Biol. 146, 1005–1018 (1999).
do Carmo Avides, M. & Glover, D. M. Abnormal spindle protein, Asp, and the integrity of mitotic centrosomal microtubule organizing centers. Science 283, 1733–1735 (1999).
Kellogg, D. R., Field, C. M. & Alberts, B. M. Identification of microtubule-associated proteins in the centrosome, spindle, and kinetochore of the early Drosophila embryo. J. Cell Biol. 109, 2977–2991 (1989).
Kellogg, D. R., Oegema, K., Raff, J., Schneider, K. & Alberts, B. M. CP60 a microtubule associated protein that is localized to the centrosome in a cell cycle specific manner. Mol. Biol. Cell 6, 1673–1684 (1995).
Oegema, K., Marshall, W. F., Sedat, J. W. & Alberts, B. M. Two proteins that cycle asynchronously between centrosomes and nuclear structures: Drosophila CP60 and CP190. J. Cell Sci. 110, 1573–1583 (1997).
Kidd, D. & Raff, J. W. LK6, a short lived protein kinase in Drosophila that can associate with microtubules and centrosomes. J. Cell Sci. 110, 209–219 (1997).
Gard, D. L. & Kirschner, M. W. A microtubule-associated protein from Xenopus eggs that specifically promotes assembly at the plus-end. J. Cell Biol. 105, 2203–2215 (1987).
Vasquez, R. J., Gard, D. L. & Cassimeris, L. XMAP from Xenopus eggs promotes rapid plus end assembly of microtubules and rapid microtubule polymer turnover. J. Cell Biol. 127, 985–993 (1994).
Tournebize, R. et al. Control of microtubule dynamics by the antagonistic activities of XMAP215 and XKCM1 in Xenopus egg extracts. Nature Cell Biol. 2, 13–19 (2000).
Spittle, C., Charrasse, S., Larroque, C. & Cassimeris, L. The interaction of TOGp with microtubules and tubulin. J. Biol. Chem. 275, 20748–20753 (2000).
Charrasse, S. et al. The TOGp protein is a new human microtubule-associated protein homologous to the Xenopus XMAP215. J. Cell Sci. 111, 1371–1383 (1998).
Matthews, L. R., Carter, P., Thierry-Mieg, D. & Kemphues, K. ZYG-9, a Caenorhabditis elegans protein required for microtubule organization and function, is a component of meiotic and mitotic spindle poles. J. Cell Biol. 141, 1159–1168 (1998).
Nabeshima, K. et al. p93dis1, which is required for sister chromatid separation, is a novel microtubule and spindle pole body-associating protein phosphorylated at the Cdc2 target sites. Genes Dev. 9, 1572–1585 (1995).
Wang, P. J. & Huffaker, T. C. Stu2p: a microtubule-binding protein that is an essential component of the yeast spindle pole body. J. Cell Biol. 139, 1271–1280 (1997).
Gergely, F., Kidd, D., Jeffers, K., Wakefield, J. G. & Raff, J. W. D-TACC: a novel centrosomal protein required for normal spindle function in the early Drosophila embryo. EMBO J. 19, 241–252 (2000).
Chen, H. M. et al. AZU-1: A candidate breast tumor suppressor and biomarker for tumor progression. Mol. Biol. Cell 11, 1357–1367 (2000).
Still, I. H., Hamilton, M., Vince, P., Wolfman, A. & Cowell, J. K. Cloning of TACC1, an embryonically expressed, potentially transforming coiled coil containing gene, from the 8p11 breast cancer amplicon. Oncogene 18, 4032–4038 (1999).
Still, I. H., Vince, P. & Cowell, J. K. The third member of the transforming acidic coiled coil-containing gene family, TACC3, maps in 4p16, close to translocation breakpoints in multiple myeloma, and is upregulated in various cancer cell lines. Genomics 58, 165–170 (1999).
Gergely, F. et al. The TACC domain identifies a new family of proteins that can interact with centrosomes and microtubules. Proc. Natl Acad. Sci. USA 97, 14352–12357 (2000).
Cullen, C. F. & Ohkura, H. Msps is localized to acentrosomal poles by Ncd and D-TACC to ensure the bipolarity of Drosophila meiotic spindles. Nature Cell Biol. 3, 637–642.
Laemmli, U. K. Cleavage of the structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685 (1970).
Shevchenko, A., Wilm, M., Vorm, O. & Mann, M. Mass spectrometric sequencing of proteins silver-stained polyacrylamide gels. Anal. Chem. 68, 850–858 (1996).
Raff, J. W., Kellogg, D. R. & Alberts, B. M. Drosophila γ-tubulin is part of a complex containing two previously identified centrosomal MAPs. J. Cell Biol. 121, 823–835 (1993).
Heuer, J. G., Li, K. & Kaufman, T. C. The Drosophila homeotic target gene centrosomin (cnn) encodes a novel centrosomal protein with leucine zippers and maps to a genomic region required for midgut morphogenesis. Development 121, 3861–3876 (1995).
Huang, J. & Raff, J. W. The disappearance of cyclin B at the end of mitosis is regulated spatially in Drosophila cells. EMBO J. 18, 2184–2195 (1999).
Rorth, P. Gal4 in the Drosophila female germline. Mech. Dev. 78, 113–118 (1998).
Towbin, H., Staehlin, T. & Gordon, J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedures and some applications. Proc. Natl Acad. Sci. USA 80, 4350–4354 (1979).
Acknowledgements
We thank H. Ohkura and F. Cullen for sharing results before publication and for supplying anti-Msps anti-serum and the mspsMJ15 allele. We thank B. Williams and M. Goldberg who provided us with their unpublished d-taccstella alleles. We also thank T. Hyman and D. Compton for providing us with anti-ch-TOG antibodies, and J. Kilmartin for advice about sample preparation for mass spectroscopy. We thank members of the laboratory for comments on the manuscript. This work was supported by a Wellcome Trust Senior Fellowship in Basic Biomedical Sciences (to J.W.R.), an MRC studentship (to M.J.L.), a Wellcome Trust Prize Studentship (to F.G.) and the Medical Research Council (S.Y.P.-C.).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Movie 1
The behaviour of a D-TACC–GFP fusion protein in a living embryo. Note how small dots of the fusion protein can be observed oscillating to and fro from the centrosomal region, as though binding to the plus ends of the centrosomal microtubules. (MOV 7992 kb)
Movie 2
The behaviour of a Msps–GFP fusion protein in a living embryo. Note how small dots of the fusion protein can be observed oscillating to and fro from the centrosomal region, as though binding to the plus ends of the centrosomal microtubules. (MOV 3252 kb)
Figure S1
The behaviour of D–TACC (top left panel), Msps (bottom left panel), microtubules (middle panel) and DNA (right panel) in embryos treated with taxol before fixation. (PDF 288 kb)
Rights and permissions
About this article
Cite this article
Lee, M., Gergely, F., Jeffers, K. et al. Msps/XMAP215 interacts with the centrosomal protein D-TACC to regulate microtubule behaviour. Nat Cell Biol 3, 643–649 (2001). https://doi.org/10.1038/35083033
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35083033
This article is cited by
-
A perinuclear microtubule-organizing centre controls nuclear positioning and basement membrane secretion
Nature Cell Biology (2020)
-
XMAP215 is a microtubule nucleation factor that functions synergistically with the γ-tubulin ring complex
Nature Cell Biology (2018)
-
Suppression of intestinal tumors by targeting the mitotic spindle of intestinal stem cells
Oncogene (2016)
-
Differentiating the roles of microtubule-associated proteins at meiotic kinetochores during chromosome segregation
Chromosoma (2016)
-
A small compound targeting TACC3 revealed its different spatiotemporal contributions for spindle assembly in cancer cells
Oncogene (2014)