Abstract
Influenza A viruses are important worldwide pathogens in humans and different animal species. The functions of most of the ten different viral proteins of this negative-strand RNA virus have been well elucidated1. However, little is known about the virus-induced intracellular signalling events that support viral replication. The Raf/MEK/ERK cascade is the prototype of mitogen-activated protein (MAP) kinase cascades and has an important role in cell growth, differentiation and survival2,3,4,5. Investigation of the function of this pathway has been facilitated by the identification of specific inhibitors such as U0126, which blocks the cascade at the level of MAPK/ERK kinase (MEK)6,7,8. Here we show that infection of cells with influenza A virus leads to biphasic activation of the Raf/MEK/ERK cascade. Inhibition of Raf signalling results in nuclear retention of viral ribonucleoprotein complexes (RNPs), impaired function of the nuclear-export protein (NEP/NS2) and concomitant inhibition of virus production. Thus, signalling through the mitogenic cascade seems to be essential for virus production and RNP export from the nucleus during the viral life cycle.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lamb, R. A. & Krug, R. M. in Fields Virology (eds Fields, B. N. et al.) 1353–1395 (Lippincott-Raven, Philadelphia, 1996).
Daum, G., Eisenmann-Tappe, I., Fries, H-W., Troppmair, J. & Rapp, U. R. Trends Biochem. Sci. 19, 474–480 (1994).
Cano, E. & Mahadevan, L. C. Trends Biochem. Sci. 20, 117–122 (1995).
Cohen, P. Trends Cell Biol. 7, 353–361 (1997).
Robinson, M. J. & Cobb, M. H. Curr. Opin. Cell Biol. 9, 180–186 (1997).
Duncia, J. V. et al. Bioorg. Med. Chem. Lett. 8, 2839–2844 (1998).
Favata, M. F. et al. J. Biol. Chem. 273, 18623–18632 (1998).
DeSilva, D. R. et al. J. Immunol. 160, 4175–4181 (1998).
Benn, J., Su, F., Doria, M. & Schneider, R. J. J. Virol. 70, 4978–4985 (1996).
Bruder, J. T. & Kovesdi, I. J. Virol. 71, 398–404 (1997).
Popik, W. & Pitha, P. M. Virology 252, 210–217 (1998).
Rodems, S. M. & Spector, D. H. J. Virol. 72, 9173–9180 (1998).
Scholtissek, C. & Muller, K. Arch. Virol. 100, 27–35 (1988).
Whittaker, G., Bui, M. & Helenius, A. J. Virol. 70, 2743–2756 (1996).
O'Neill, R. E., Talon, J. & Palese, P. EMBO J. 17, 288–296 (1998).
Kistner, O., Müller, K. & Scholtissek, C. J. Gen. Virol. 70, 2421–2431 (1989).
Gregoriades, A., Guzman, G. G. & Paoletti, E. Virus Res. 16, 27–41 (1990).
Richardson, J. C. & Akkina, R. K. Arch. Virol. 116, 69–80 (1991).
Fischer, U., Huber, J., Boelens, W. C., Mattaj, I. W. & Luhrmann, R. Cell 82, 475–483 (1995).
Hope, T. J., Huang, X. J., McDonald, D. & Parslow, T. G. Proc. Natl Acad. Sci. USA 87, 7787–7791 (1990).
Scholtissek, C. & Müller, K. Arch. Virol. 119, 111–118 (1991).
Sebolt-Leopold, J. S. et al. Nature Med. 5, 810–816 (1999).
Ludwig, S. et al. Mol. Cell. Biol. 16, 6687–6697 (1996).
Acknowledgements
We thank P. Palese, R. Rott, C. Scholtissek and P. Staeheli for various influenza virus strains, T. Parslow for the pDM128 plasmid, I. Böhle and H. Häfner for technical assistance, and A. Hoffmeyer, S. Feller, J. Troppmair, G. Neumann, M. Camargo and R. Rott for discussions and critical reading of the manuscipt. This work was supported by grants from the Deutsche Forschungsgemeinschaft and the Fonds der Chemischen Industrie.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Download plugins
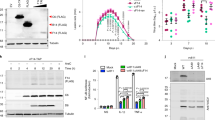
Figure S1. U0126 does not affect vRNA synthesis, transcriptional activity of the viral RDRP or accumulation of viral proteins. RNA isolated from MDCK cells that were infected with WSN-HK or FPV and treated with 50 μM U0126 or DMSO was spotted onto nylon membranes and hybridized with specific probes for nucleoprotein vRNA and mRNA/cRNA, M1 vRNA or GAPDH. The amounts of specific vRNAs synthesized in WSN-HK- or FPV-infected cells in the presence or absence of U0126 were determined in Dot-Blot analyses. b, MDCK cells were incubated with the indicated concentrations of U0126 or DMSO before, during and after infection with transfectant WSN-HK-CAT carrying an extra vRNA-like segment encoding the CAT reporter gene. After 16 h cells were lysed and analysed for CAT activity as an indicator of transcriptional activity of vRDRP. c, MDCK cells were infected with FPV (MOI = 10) in the presence or absence of 50 µM U0126 before, during and after infection. Cell lysates were analysed by western blotting for expression of influenza nucleoprotein, M1 and NS1 at different time points after infection. As a loading control, the same membranes were reblotted with a specific antiserum against ERK2. (PDF 454 kb)
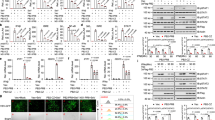
Figure S2. Inhibition of the Raf/MEK/ERK signalling cascade interferes with the nuclear-export activity of NEP/NS2. HeLa cells were transfected with pcDNA3-Rev(1–69)NEP, pDM128 for CAT expression through an RRE-containing mRNA and pCMV-Luc for constitutive expression of firefly luciferase. After 5 h cells were treated with 40, 50 or 60 μM U0126 or the solvent equivalent of 0.6% DMSO. After 25 h cell lysates, normalized for protein content, were assayed for CAT and luciferase activity. CAT activities shown are means from duplicate transfections nor-malized on luciferase values with the solvent control designated as 100%. Data are representative of several independent experiments. b, HeLa cells were transfected as in a together with empty vector or with plasmids expressing dominant negative forms of Raf (Raf(C4B)) or ERK2 (ERK(2B3)) and assayed for reporter-gene activites as in a. CAT activities shown are means of triplicate transfections normalized for luciferase values with the vector control designated as 100%.
Rights and permissions
About this article
Cite this article
Pleschka, S., Wolff, T., Ehrhardt, C. et al. Influenza virus propagation is impaired by inhibition of the Raf/MEK/ERK signalling cascade. Nat Cell Biol 3, 301–305 (2001). https://doi.org/10.1038/35060098
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35060098
This article is cited by
-
Upregulation of galectin-3 in influenza A virus infection promotes viral RNA synthesis through its association with viral PA protein
Journal of Biomedical Science (2023)
-
Carbon dioxide and MAPK signalling: towards therapy for inflammation
Cell Communication and Signaling (2023)
-
ROCK1/MLC2 inhibition induces decay of viral mRNA in BPXV infected cells
Scientific Reports (2022)
-
The MEK1/2-inhibitor ATR-002 efficiently blocks SARS-CoV-2 propagation and alleviates pro-inflammatory cytokine/chemokine responses
Cellular and Molecular Life Sciences (2022)
-
Pleiotropic effects of clopidogrel
Purinergic Signalling (2022)