Abstract
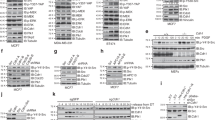
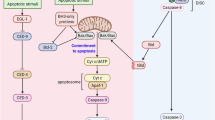
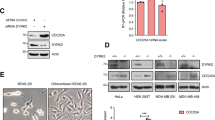
DAP kinase is a pro-apoptotic calcium-regulated serine/threonine kinase, whose expression is frequently lost in human tumours. Here we show that DAP kinase counteracts oncogene-induced transformation by activating a p19ARF/p53-dependent apoptotic checkpoint. Ectopic expression of DAP kinase suppressed oncogenic transformation of primary embryonic fibroblasts by activating p53 in a p19ARF-dependent manner. Consequently, the fibroblasts underwent apoptosis, characterized by caspase activation and DNA fragmentation. In response to c-Myc or E2F-1, the endogenous DAP kinase protein was upregulated. Furthermore, functional or genetic inactivation of the endogenous DAP kinase reduced the extent of induction of p19ARF/p53 and weakened the subsequent apoptotic responses to c-Myc or E2F-1. These results establish a role for DAP kinase in an early apoptotic checkpoint designed to eliminate pre-malignant cells during cancer development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Deiss, L. P. & Kimchi, A. A genetic tool used to identify thioredoxin as a mediator of a growth inhibitory signal. Science 252, 117–120 ( 1991).
Deiss, L. P., Feinstein, E., Berissi, H., Cohen, O. & Kimchi, A. Identification of a novel serine/threonine kinase and a novel 15-kD protein as potential mediators of the gamma interferon-induced cell death. Genes Dev. 9, 15– 30 (1995).
Cohen, O., Feinstein, E. & Kimchi, A. DAP kinase is a Ca2+/calmodulin-dependent, cytoskeletal-associated protein kinase, with cell death-inducing functions that depend on its catalytic activity. EMBO J. 16, 998– 1008 (1997).
Cohen, O. et al. DAP kinase participates in TNF-alpha- and Fas-induced apoptosis and its function requires the death domain. J. Cell Biol. 146, 141–148 (1999).
Inbal, B. et al. DAP kinase links the control of apoptosis to metastasis. Nature 390, 180–184 ( 1997).
Feinstein, E., Wallach, D., Boldin, M., Varfolomeev, E. & Kimchi, A. The death domain: a module shared by proteins with diverse cellular functions. Trends Biochem. Sci. 20, 342–344 ( 1995).
Raveh, T., Berissi, H., Eisenstein, M., Spivak, T. & Kimchi, A. A functional genetic screen identifies regions at the C-terminal tail and death-domain of death-associated protein kinase that are critical for its proapoptotic activity. Proc. Natl Acad. Sci. USA 97, 1572–1577 (2000).
Kissil, J. L. et al. DAP kinase loss of expression in various carcinoma and B-cell lymphoma cell lines: possible implications for role as tumor suppressor gene . Oncogene 15, 403–407 (1997).
Katzenellenbogen, R. A., Baylin, S. B. & Herman, J. G. Hypermethylation of the DAP kinase CpG island is a common alteration in B-cell malignancies. Blood 93, 4347–4353 (1999).
Esteller, M. et al. Detection of aberrant promoter hypermethylation of tumor suppressor genes in serum DNA from non-small cell lung cancer patients. Cancer Res 59, 67–70 ( 1999).
Sanchez-Cespedes, M. et al. Gene promoter hypermethylation in tumors and serum of head and neck cancer patients. Cancer Res. 60, 892–895 (2000).
Manfredi, J. J. & Prives, C. The transforming activity of simian virus 40 large tumor antigen. Biochim. Biophys. Acta 1198, 65–83 ( 1994).
Bryan, T. M. & Reddel, R. R. SV40-induced immortalization of human cells. Crit. Rev. Oncol. 5, 331 –357 (1994).
Nevins, J. R. Cell cycle targets of the DNA tumor viruses. Curr. Opin. Genet. Dev. 4, 130–134 ( 1994).
Pipas, J. M., Peden, K. W. & Nathans, D. Mutational analysis of simian virus 40 T antigen: isolation and characterization of mutants with deletions in the T-antigen gene. Mol. Cell. Biol. 2, 203–213 (1983).
Michalovitz, D., Halevy, O. & Oren, M. Conditional inhibition of transformation and of cell proliferation by a temperature-sensitive mutant of p53. Cell 62, 671–680 (1990).
Shaulian, E., Zauberman, A., Ginsberg, D. & Oren, M. Identification of a minimal transforming domain of p53: negative dominance through abrogation of sequence-specific DNA binding. Mol. Cell. Biol. 12, 5581–5592 ( 1992).
Pietenpol, J. A. et al. Sequence-specific transcriptional activation is essential for growth suppression by p53. Proc. Natl Acad. Sci. USA 91, 1998–2002 (1994).
Crook, T., Marston, N. J., Sara, E. A. & Vousden, K. H. Transcriptional activation by p53 correlates with suppression of growth but not transformation. Cell 79, 817– 827 (1994).
Harvey, D. M. & Levine, A. J. p53 alteration is a common event in the spontaneous immortalization of primary BALB/c murine embryo fibroblasts. Genes Dev. 12B, 2375 –2385 (1991).
Sharpless, N. E. & DePinho, R. A. The INK4A/ARF locus and its two gene products. Curr. Opin. Genet. Dev. 9, 22–30 (1999).
Sherr, C. J. & Weber, J. D. The ARF/p53 pathway . Curr. Opin. Genet. Dev. 10, 94– 99 (2000).
Gottlieb, T. M. & Oren, M. p53 in growth control and neoplasia. Biochim. Biophys. Acta 1287, 77–102 (1996).
Ko, L. J. & Prives, C. p53: puzzle and paradigm . Genes Dev 10, 1054–1072 (1996).
Quelle, D. E., Zindy, F., Ashmun, R. A. & Sherr, C. J. Alternative reading frames of the INK4a tumor suppressor gene encode two unrelated proteins capable of inducing cell cycle arrest. Cell 83, 993–1000 (1995).
Sherr, C. J. Tumor surveillance via the ARF-p53 pathway. Genes Dev. 12, 2984–2991 (1998).
Lowe, S. W. Activation of p53 by oncogenes. Endocr. Relat. Cancer 6, 45–48 (1999).
Pomerantz, J. et al. The Ink4a tumor suppressor gene product, p19ARF, interacts with MDM2 and neutralizes MDM2's inhibition of p53. Cell 92, 713–723 ( 1998).
Zindy, F. et al. Myc signaling via the ARF tumor suppressor regulates p53-dependent apoptosis and immortalization. Genes Dev. 12, 2424–2433 (1998).
de Stanchina, E. et al. E1A signaling to p53 involves the p19(ARF) tumor suppressor . Genes Dev. 12, 2434–2442 (1998).
Bates, S. et al. p14ARF links the tumour suppressors RB and p53. Nature 395, 124–125 ( 1998).
Hermeking, H. & Eick, D. Mediation of c-Myc-induced apoptosis by p53. Science 265, 2091– 2093 (1994).
Schmitt, C. A., McCurrach, M. E., de Stanchina, E., Wallace-Brodeur, R. R. & Lowe, S. W. INK4a/ARF mutations accelerate lymphomagenesis and promote chemoresistance by disabling p53. Genes Dev. 13, 2670– 2677 (1999).
Eischen, C. M., Weber, J. D., Roussel, M. F., Sherr, C. J. & Cleveland, J. L. Disruption of the ARF-Mdm2-p53 tumor suppressor pathway in Myc-induced lymphomagenesis. Genes Dev. 13, 2658–2669 (1999).
Finlay, C. Rat embryo fibroblast complementation assay with ras genes. Methods Enzymol. 255, 389–394 ( 1995).
Jiang, W. & Hunter, T. Analysis of cell-cycle profiles in transfected cells using amembrane-targeted GFP. Biotechniques 24, 352–354 ( 1998).
Pear, W. S., Nolan, G. P., Scott, M. L. & Baltimore, D. Production of high-titer helper-free retroviruses by transient transfection . Proc. Natl Acad. Sci. USA 90, 8392– 8396 (1993).
Takahashi, M. et al. Long-term correction of bilirubin-UDP-glucuronosyltransferase deficiency in Gunn rats by administration of a recombinant adenovirus during the neonatal period. J. Biol. Chem. 271, 26536–26542 (1996).
Acknowledgements
We are grateful to Moshe Oren for helpful discussions and reagents. We thank Charles Sherr for the gift of p19ARF−/− MEFs, Claudio Schneider for kindly providing the anti-p21 antibodies, Tony Hunter for the membrane-targeted GFP plasmid, Doron Ginsberg for the E2F-1 plasmid, and members of the Kimchi lab for critical reading of the manuscript. This work was supported by the Israel Foundation, which is administered by the Israel Academy of Science and Humanities, and by QBI Ltd. M.S.H. is supported by the Forchheimer Foundation. R.A.D is an American Cancer Society Research Professor and is supported by grants from the National Institutes of Health. A.K. is the incumbent of the Helena Rubinstein Chair of Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Raveh, T., Droguett, G., Horwitz, M. et al. DAP kinase activates a p19ARF/p53-mediated apoptotic checkpoint to suppress oncogenic transformation. Nat Cell Biol 3, 1–7 (2001). https://doi.org/10.1038/35050500
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35050500
This article is cited by
-
The role and implication of autophagy in cholangiocarcinoma
Cell Death Discovery (2023)
-
Death-associated protein kinase 1 phosphorylates MDM2 and inhibits its protein stability and function
Archives of Pharmacal Research (2023)
-
MYC: a multipurpose oncogene with prognostic and therapeutic implications in blood malignancies
Journal of Hematology & Oncology (2021)
-
Tumor suppressor death-associated protein kinase 1 inhibits necroptosis by p38 MAPK activation
Cell Death & Disease (2020)
-
DAPK1 loss triggers tumor invasion in colorectal tumor cells
Cell Death & Disease (2019)