Abstract
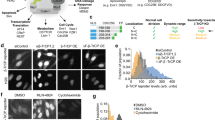
Precise timing of mitosis is essential for high-fidelity cell duplication. However, temporal measurements of the mitotic clock have been challenging. Here we present a fluorescent mitosis biosensor that monitors the time between nuclear envelope breakdown (NEB) and re-formation using parallel total internal reflection fluorescence (TIRF) microscopy. By tracking tens to hundreds of mitotic events per experiment, we found that the mitotic clock of unsynchronized rat basophilic leukemia cells has a marked precision with 80% of cells completing mitosis in 32 ± 6 min. This assay further allowed us to observe delays in mitotic timing at Taxol concentrations 100 times lower than previous minimal effective doses, explaining why Taxol is clinically active at low concentrations. Inactivation of the spindle checkpoint by targeting the regulator Mad2 with RNAi consistently shortened mitosis, providing direct evidence that the internal mitotic timing mechanism is much faster in cells that lack the checkpoint.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ekholm, S.V., Zickert, P., Reed, S.I. & Zetterberg, A. Accumulation of cyclin E is not a prerequisite for passage through the restriction point. Mol. Cell. Biol. 21, 3256–3265 (2001).
Elledge, S.J. Cell cycle checkpoints: preventing an identity crisis. Science 274, 1664–1672 (1996).
Musacchio, A. & Hardwick, K.G. The spindle checkpoint: structural insights into dynamic signalling. Nature Rev. Mol. Cell Biol. 3, 731–741 (2002).
Yu, H. Regulation of APC-Cdc20 by the spindle checkpoint. Curr. Opin. Cell Biol. 14, 706–714 (2002).
Michel, L.S. et al. MAD2 haplo-insufficiency causes premature anaphase and chromosome instability in mammalian cells. Nature 409, 355–359 (2001).
Rieder, C.L. & Khodjakov, A. Mitosis through the microscope: advances in seeing inside live dividing cells. Science 300, 91–96 (2003).
Bajer, A. Cine-Micrographic Studies on mitosis in endosperm. Exp. Cell Res. 14, 245–256 (1958).
Rieder, C.L. Effect of hypothermia (20–25 degrees C) on mitosis in PtK1 cells. Cell Biol. Int. Rep. 5, 563–573 (1981).
Zhang, J., Campbell, R.E., Ting, A.Y. & Tsien, R.Y. Creating new fluorescent probes for cell biology. Nat. Rev. Mol. Cell Biol. 3, 906–918 (2002).
Steyer, J.A. & Almers, W. A real-time view of life within 100 nm of the plasma membrane. Nat. Rev. Mol. Cell Biol. 2, 268–275 (2001).
Tengholm, A., Teruel, M.N. & Meyer, T. Single cell imaging of PI3K activity and glucose transporter insertion into the plasma membrane by dual color evanescent wave microscopy. Science's Stke 2003, PL4 (2003).
Haraguchi, T. et al. Live fluorescence imaging reveals early recruitment of emerin, LBR, RanBP2, and Nup153 to reforming functional nuclear envelopes. J. Cell Sci. 113, 779–794 (2000).
Wang, T.H., Wang, H.S. & Soong, Y.K. Paclitaxel-induced cell death: where the cell cycle and apoptosis come together. Cancer 88, 2619–2628 (2000).
Jordan, M.A., Toso, R.J., Thrower, D. & Wilson, L. Mechanism of mitotic block and inhibition of cell proliferation by taxol at low concentrations. Proc. Nat. Acad. Sci. USA 90, 9552–9556 (1993).
Jordan, M.A. et al. Mitotic block induced in HeLa cells by low concentrations of paclitaxel (Taxol) results in abnormal mitotic exit and apoptotic cell death. Cancer Res. 56, 816–825 (1996).
Sorger, P.K., Dobles, M., Tournebize, R. & Hyman, A.A. Coupling cell division and cell death to microtubule dynamics. Curr. Opin. Cell Biol. 9, 807–814 (1997).
Rieder, C.L., Schultz, A., Cole, R.W. & Sluder, G. Anaphase onset in vertebrate somatic cells is controlled by a checkpoint that monitors sister kinetochore attachment to the spindle. J. Cell Biol. 127, 1301–1310 (1994).
Gorbsky, G.J., Chen, R.H. & Murray, A.W. Microinjection of antibody to Mad2 protein into mammalian cells in mitosis induces premature anaphase. J. Cell Biol. 141, 1193–1205 (1998).
Fang, G., Yu, H. & Kirschner, M.W. The checkpoint protein MAD2 and the mitotic regulator CDC20 form a ternary complex with the anaphase-promoting complex to control anaphase initiation. Genes Dev. 12, 1871–1883 (1998).
Dobles, M., Liberal, V., Scott, M.L., Benezra, R. & Sorger, P.K. Chromosome missegregation and apoptosis in mice lacking the mitotic checkpoint protein Mad2. Cell 101, 635–645 (2000).
Mikhailov, A., Cole, R.W. & Rieder, C.L. DNA damage during mitosis in human cells delays the metaphase/anaphase transition via the spindle-assembly checkpoint. Curr. Biol. 12, 1797–1806 (2002).
Canman, J.C., Salmon, E.D. & Fang, G. Inducing precocious anaphase in cultured mammalian cells. Cell Mot. Cyto. 52, 61–65 (2002).
Myers, J.W., Jones, J.T., Meyer, T. & Ferrell, J.E. Recombinant Dicer efficiently converts large dsRNAs into siRNAs suitable for gene silencing. Nat. Biotechnol. 21, 324–328 (2003).
Teruel, M.N. & Meyer, T. Parallel single-cell monitoring of receptor-triggered membrane translocation of a calcium-sensing protein module. Science 295, 1910–1912 (2002).
Heo, W.D. & Meyer, T. Switch-of-function mutants based on morphology classification of ras superfamily small GTPases. Cell 113, 315–328 (2003).
Campbell, R.E. et al. A monomeric red fluorescent protein. Proc. Nat. Acad. Sci. USA 99, 7877–7882 (2002).
Rekas, A., Alattia, J.R., Nagai, T., Miyawaki, A. & Ikura, M. Crystal structure of venus, a yellow fluorescent protein with improved maturation and reduced environmental sensitivity. J. Biol. Chem. 277, 50573–50578 (2002).
Acknowledgements
We would like to thank Guowei Fang, Thierry Galvez, Marc Fivaz and Angie Hahn for critical reading of the manuscript. We would also like to thank G. Fang for his kind gift of the Mad2 antibody, Tom Wehrman for help with creating the stable line and the Meyer and Ferrell labs for their support. This work was supported by the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Movie 1.
Location of the biosensor during mitosis. Asynchronous RBL-FMB cells were imaged every 15 minutes with a CFP/YFP spinning disc confocal system. 3 cells that enter mitosis are identified by the white arrows. The scale bar in the lower left corner represents 20 µm and the time is in hours:minutes in the lower right corner. (MOV 204 kb)
Supplementary Movie 2.
Localization of the biosensor as monitored by total internal reflection fluorescence (TIRF) microscopy. RBL-FMB cells were imaged with a prism based CFP/YFP TIRF system every two minutes. The rectangular region highlights one cell undergoing mitosis with six cells in total going through mitosis during the course of the time lapse. The scale bar in the lower left corner represents 20 µm and the time is in hours:minutes in the lower right corner. (MOV 1890 kb)
Supplementary Movie 3.
A 10X TIRF view of cycling RBL-FMB cells. RBL-FMB cells were imaged with a prism based CFP/YFP TIRF system. 70 cells undergo mitosis in the field of view during this 5 hour experiment. 75 frames of 149 are represented here. The scale bar in the lower left corner represents 50 µm and the time is represented in hours:minutes in the lower right corner. (MOV 6490 kb)
Supplementary Movie 4.
40X TIRF and transmitted light time lapse of cycling mitotic cells. RBL-FMB cells were imaged with a prism based CFP/YFP TIRF system with near simultaneous transmitted light exposures. The scale bar in the lower left corner represents ten mm and the time is represented in hours:minutes in the lower right corner. (MOV 1325 kb)
Rights and permissions
About this article
Cite this article
Jones, J., Myers, J., Ferrell, J. et al. Probing the precision of the mitotic clock with a live-cell fluorescent biosensor. Nat Biotechnol 22, 306–312 (2004). https://doi.org/10.1038/nbt941
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt941
This article is cited by
-
Delayed APC/C activation extends the first mitosis of mouse embryos
Scientific Reports (2017)
-
Adaptive changes in the kinetochore architecture facilitate proper spindle assembly
Nature Cell Biology (2015)
-
A role for the Rab6A′ GTPase in the inactivation of the Mad2-spindle checkpoint
The EMBO Journal (2006)
-
The pulse of mitosis
Nature Biotechnology (2004)
-
Genome-wide survey of protein kinases required for cell cycle progression
Nature (2004)