Abstract
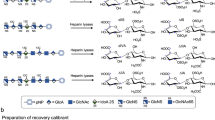
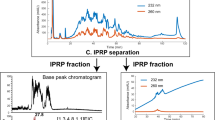
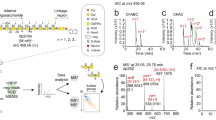
Recently, certain lots of heparin have been associated with an acute, rapid onset of serious side effects indicative of an allergic-type reaction. To identify potential causes for this sudden rise in side effects, we examined lots of heparin that correlated with adverse events using orthogonal high-resolution analytical techniques. Through detailed structural analysis, the contaminant was found to contain a disaccharide repeat unit of glucuronic acid linked β1→3 to a β-N-acetylgalactosamine. The disaccharide unit has an unusual sulfation pattern and is sulfated at the 2-O and 3-O positions of the glucuronic acid as well as at the 4-O and 6-O positions of the galactosamine. Given the nature of this contaminant, traditional screening tests cannot differentiate between affected and unaffected lots. Our analysis suggests effective screening methods that can be used to determine whether or not heparin lots contain the contaminant reported here.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Capila, I. & Linhardt, R.J. Heparin-protein interactions. Angew. Chem. Int. Edn. Engl. 41, 391–412 (2002).
Lepor, N.E. Anticoagulation for acute coronary syndromes: from heparin to direct thrombin inhibitors. Rev. Cardiovasc. Med. 8 (suppl. 3), S9–S17 (2007).
Fischer, K.G. Essentials of anticoagulation in hemodialysis. Hemodial. Int. 11, 178–189 (2007).
Contaminant detected in heparin material of specified origin in the USA and in Germany; serious adverse events reported; recall measures initiated. World Health Organization Alert No. 118 (7 March 2008). <http://www.who.int/medicines/publications/ drugalerts/Alert_118_Heparin.pdf>
Notice of Recall from Rotexmedica to Bfarm (German Regulatory Authorities). Rotexmedica/Bfarm Notice (7 March 2008). <http://www.akdae.de/20/40/Archiv/2008/20080310.pdf>
Acute allergic-type reactions among patients undergoing hemodialysis—multiple states, 2007–2008. Morbid. Mortal. Wkly Rept. 57 (1 February 2008).
2008 Heparin Recall Information. Baxter Investigation Updates (5, 14, 19 March 2008). <http://www.baxter.com/products/ biopharmaceuticals/heparin.html>
Baxter provides update on investigation. Baxter Investigation Update (14 March 2008). <http://www.baxter.com/products/biopharmaceuticals/ downloads/heparin_03-14-08.pdf>
Communication. Information on heparin sodium injection. US Food and Drug Administration. <http://www.fda.gov/cder/drug/infopage/heparin/default.htm>
Holme, K.R. & Perlin, A.S. Nuclear magnetic resonance spectra of heparin in admixture with dermatan sulfate and other glycosaminoglycans. 2-D spectra of the chondroitin sulfates. Carbohydr. Res. 186, 301–312 (1989).
Guerrini, M., Bisio, A. & Torri, G. Combined quantitative 1H and 13C nuclear magnetic resonance spectroscopy for characterization of heparin preparations. Semin. Thromb. Hemost. 27, 473–482 (2001).
Myette, J.R. et al. Molecular cloning of the heparin/heparan sulfate delta 4,5 unsaturated glycuronidase from Flavobacterium heparinum, its recombinant expression in Escherichia coli, and biochemical determination of its unique substrate specificity. Biochemistry 41, 7424–7434 (2002).
Myette, J.R. et al. The heparin/heparan sulfate 2-O-sulfatase from Flavobacterium heparinum. Molecular cloning, recombinant expression, and biochemical characterization. J. Biol. Chem. 278, 12157–12166 (2003).
Kuberan, B. et al. Analysis of heparan sulfate oligosaccharides with ion pair-reverse phase capillary high performance liquid chromatography-microelectrospray ionization time-of-flight mass spectrometry. J. Am. Chem. Soc. 124, 8707–8718 (2002).
Maruyama, T., Toida, T., Imanari, T., Yu, G. & Linhardt, R.J. Conformational changes and anticoagulant activity of chondroitin sulfate following its O-sulfonation. Carbohydr. Res. 306, 35–43 (1998).
Solvolytic desulfation of glycosaminoglycuronan sulfates with dimethyl sulfoxide containing water or methanol. Nagasawa, K., Inoue, Y. & Kamata, T. Carbohydr. Res. 58, 47–55 (1977).
Linhardt, R.J. Analysis of glycosaminoglycans with polysaccharide lyases. in Current Protocols in Molecular Biology (eds. Ausubel, F.A. et al.) 17.13B.1–17B.13.16 (Wiley, New York, 1994).
Kinoshita, A. et al. Novel tetrasaccharides isolated from squid cartilage chondroitin sulfate E contain unusual sulfated disaccharide units GlcA(3-O-sulfate)β1-3GalNAc(6-O-sulfate) or GlcA(3-O-sulfate)β1-3GalNAc. J. Biol. Chem. 272, 19656–19665 (1997).
Hadding, U. et al. Ability of the T cell-replacing polyanion dextran sulfate to trigger the alternate pathway of complement activation. Eur. J. Immunol. 3, 527–529 (1973).
Kang, O.H. et al. Suppressive effect of non-anaphylactogenic anti-IgE antibody on the development of dextran sulfate sodium-induced colitis. Int. J. Mol. Med. 18, 893–899 (2006).
Siebeck, M. et al. Dextran sulfate activates contact system and mediates arterial hypotension via B2 kinin receptors. J. Appl. Physiol. 77, 2675–2680 (1994).
Hojima, Y., Cochrane, C.G., Wiggins, H.C., Austen, K.F. & Steven, R.L. In vitro activation of the contact (Hageman factor) system of plasma by heparin and chondroitin sulfate E. Blood 63, 1453–1459 (1984).
Naggi, A. et al. Sulfamino-galactosaminoglycans, a new class of semi-synthetic polysaccharides. in Biomedical and Biotechnological Advances in Industrial Polysaccharides (eds. Crescenzi, V., Dea, I.C.M. & Paoletti, S.) 101–108 (Gordon & Breach Science, New York, 1989).
Siegmenth, W. & Radi, I. Comparison of glycosaminoglycan polysulfate (Arteparon) and physiological saline solution in arthrosis of the large joints. Results of a multicenter double-blind study. Z. Rheumatol. 42, 223–228 (1983).
Greinacher, A., Michels, I., Schäfer, M., Kiefel, V. & Mueller-Eckhardt, C. Heparin-associated thrombocytopenia in a patient treated with polysulphated chondroitin sulphate: evidence for immunological crossreactivity between heparin and polysulphated glycosaminoglycan. Br. J. Haematol. 81, 252–254 (1992).
Weimann, G. et al. Glucosamine sulfate does not crossreact with the antibodies of patients with heparin-induced thrombocytopenia. Eur. J. Haematol. 66, 195–199 (2001).
Cicero, O., Barbato, G. & Bazzo, R. Sensitivity enhancement of a two-dimensional experiment for the measurement of heteronuclear long-range coupling constants, by a new scheme of coherence selection by gradients. J. Magn. Reson. 148, 209–213 (2001).
Cifonelli, J.C. Reaction of heparitin sulfate with nitrous acid. Carbohydr. Res. 8, 233–242 (1968).
Wishart, D.S. et al. 1H, 13C and 15N chemical shift referencing in biomolecular NMR. J. Biomol. NMR 6, 135–140 (1995).
Acknowledgements
The authors would like to thank Sucharita Roy for work on the chemical synthesis of oversulfated chondroitin sulfate standards, Scott Bailey for assistance in the analysis of selected samples by one- and two-dimensional NMR and Laura Citterio for work in various separation and analytical steps. We also thank James Anderson, Josh Sorafine and Andre Jones for design of composition experiments and HPLC analysis of the heparinase-digested samples. Finally, we thank Ada Ziolkowski for help with manuscript preparation. This work was supported in part by US National Institute of General Medical Sciences grants GM57073 (R.S.) and GM38060 (R.J.L).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
R.L. and R.S. have been directors of Momenta Pharmaceuticals since 2001 and own stock in the company, and both of them have served as scientific advisors to Scientific Protein Labs (SPL) since the end of February 2008. R.J.L. has served as a scientific advisor to Baxter International since mid-February 2008.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 (PDF 1470 kb)
Rights and permissions
About this article
Cite this article
Guerrini, M., Beccati, D., Shriver, Z. et al. Oversulfated chondroitin sulfate is a contaminant in heparin associated with adverse clinical events. Nat Biotechnol 26, 669–675 (2008). https://doi.org/10.1038/nbt1407
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1407
This article is cited by
-
Professor Casu’s contribution to cyclodextrins, the remarkable cage-shaped molecules: a review
Environmental Chemistry Letters (2022)
-
Analytical challenges of glycosaminoglycans at biological interfaces
Analytical and Bioanalytical Chemistry (2022)
-
Silver-assisted gold-catalyzed formal synthesis of the anticoagulant Fondaparinux pentasaccharide
Communications Chemistry (2021)
-
Peroxidase Mimicking Activity of Palladium Nanocluster Altered by Heparin
Catalysis Letters (2021)
-
Bioengineered production of glycosaminoglycans and their analogues
Systems Microbiology and Biomanufacturing (2021)