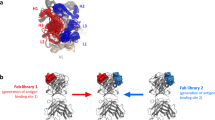
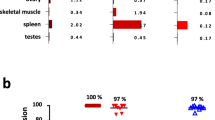
Abstract
For complex diseases in which multiple mediators contribute to overall disease pathogenesis by distinct or redundant mechanisms, simultaneous blockade of multiple targets may yield better therapeutic efficacy than inhibition of a single target. However, developing two separate monoclonal antibodies for clinical use as combination therapy is impractical, owing to regulatory hurdles and cost. Multi-specific, antibody-based molecules have been investigated; however, their therapeutic use has been hampered by poor pharmacokinetics, stability and manufacturing feasibility. Here, we describe a generally applicable model of a dual-specific, tetravalent immunoglobulin G (IgG)-like molecule—termed dual-variable-domain immunoglobulin (DVD-Ig)—that can be engineered from any two monoclonal antibodies while preserving activities of the parental antibodies. This molecule can be efficiently produced from mammalian cells and exhibits good physicochemical and pharmacokinetic properties. Preclinical studies of a DVD-Ig protein in an animal disease model demonstrate its potential for therapeutic application in human diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
van den Berg, W.B., Joosten, L.A., Helsen, M. & van de Loo, F.A. Amelioration of established murine collagen-induced arthritis with anti-IL-1 treatment. Clin. Exp. Immunol. 95, 237–243 (1994).
Maruotti, N., Cantatore, F.P., Crivellato, E., Vacca, A. & Ribatti, D. Angiogenesis in rheumatoid arthritis. Histol. Histopathol. 21, 557–566 (2006).
Uno, T. et al. Eradication of established tumors in mice by a combination antibody-based therapy. Nat. Med. 12, 693–698 (2006).
Strauss, S.J. et al. Multicenter phase II trial of immunotherapy with the humanized anti-CD22 antibody, epratuzumab, in combination with rituximab, in refractory or recurrent non-Hodgkin's lymphoma. J. Clin. Oncol. 24, 3880–3886 (2006).
Holliger, P. & Hudson, P.J. Engineered antibody fragments and the rise of single domains. Nat. Biotechnol. 23, 1126–1136 (2005).
Kriangkum, J., Xu, B., Nagata, L.P., Fulton, R.E. & Suresh, M.R. Bispecific and bifunctional single chain recombinant antibodies. Biomol. Eng. 18, 31–40 (2001).
Marvin, J.S. & Zhu, Z. Recombinant approaches to IgG-like bispecific antibodies. Acta Pharmacol. Sin. 26, 649–658 (2005).
Lacy, S. et al. IL-12/P40 Binding Proteins. Patent Application Number: WO2006US25584A (2007).
Ghayur, T. et al. IL-18 Binding Proteins. United States Patent WO2004US37971A (2005).
Wurm, F.M. Production of recombinant protein therapeutics in cultivated mammalian cells. Nat. Biotechnol. 22, 1393–1398 (2004).
Lattenmayer, C. et al. Protein-free transfection of CHO host cells with an IgG-fusion protein: selection and characterization of stable high producers and comparison to conventionally transfected clones. Biotechnol. Bioeng. 96, 1118–1126 (2007).
Lattenmayer, C. et al. Characterisation of recombinant CHO cell lines by investigation of protein productivities and genetic parameters. J. Biotechnol. 128, 716–725 (2007).
Salfeld, J. et al. Human antibodies that bind human IL-12 and methods for producing. US Patent 6914128 (2005).
Lauw, F.N. et al. Proinflammatory effects of IL-10 during human endotoxemia. J. Immunol. 165, 2783–2789 (2000).
Micallef, M.J. et al. Interferon-gamma-inducing factor enhances T helper 1 cytokine production by stimulated human T cells: synergism with interleukin-12 for interferon-gamma production. Eur. J. Immunol. 26, 1647–1651 (1996).
Yu, J.J., Tripp, C.S. & Russell, J.H. Regulation and phenotype of an innate Th1 cell: role of cytokines and the p38 kinase pathway. J. Immunol. 171, 6112–6118 (2003).
Neumann, D. et al. Injection of IL-12- and IL-18-encoding plasmids ameliorates the autoimmune pathology of MRL/Mp-Tnfrsf6lpr mice: synergistic effect on autoimmune symptoms. Int. Immunol. 18, 1779–1787 (2006).
Tary-Lehmann, M., Saxon, A. & Lehmann, P.V. The human immune system in hu-PBL-SCID mice. Immunol. Today 16, 529–533 (1995).
Joosten, L.A., Helsen, M.M., van de Loo, F.A. & van den Berg, W.B. Anticytokine treatment of established type II collagen-induced arthritis in DBA/1 mice. A comparative study using anti-TNF alpha, anti-IL-1 alpha/beta, and IL-1Ra. Arthritis Rheum. 39, 797–809 (1996).
Dinarello, C.A., Muegge, K. & Durum, S.K. Measurement of soluble and membrane-bound interleukin 1 using a fibroblast bioassay. In Current Protocols in Immunology 6.2.1–6.2.7 (Wiley, Hoboken, New Jersey, USA, 2000).
Sandin, S., Ofverstedt, L.G., Wikstrom, A.C., Wrange, O. & Skoglund, U. Structure and flexibility of individual immunoglobulin G molecules in solution. Structure 12, 409–415 (2004).
Lu, D. et al. A fully human recombinant IgG-like bispecific antibody to both the epidermal growth factor receptor and the insulin-like growth factor receptor for enhanced antitumor activity. J. Biol. Chem. 280, 19665–19672 (2005).
Shen, J. et al. Single variable domain antibody as a versatile building block for the construction of IgG-like bispecific antibodies. J. Immunol. Methods 318, 65–74 (2007).
Marvin, J.S. & Zhu, Z. Bispecific antibodies for dual-modality cancer therapy: killing two signaling cascades with one stone. Curr. Opin. Drug Discov. Devel. 9, 184–193 (2006).
Arend, W.P. Cytokines and cellular interactions in inflammatory synovitis. J. Clin. Invest. 107, 1081–1082 (2001).
Kufer, P., Lutterbuse, R. & Baeuerle, P.A. A revival of bispecific antibodies. Trends Biotechnol. 22, 238–244 (2004).
Clark, E.A., Shu, G. & Ledbetter, J.A. Role of the Bp35 cell surface polypeptide in human B-cell activation. Proc. Natl. Acad. Sci. USA 82, 1766–1770 (1985).
Kung, P., Goldstein, G., Reinherz, E.L. & Schlossman, S.F. Monoclonal antibodies defining distinctive human T cell surface antigens. Science 206, 347–349 (1979).
Arndt, M. & Krauss, J. Bispecific diabodies for cancer therapy. Methods Mol. Biol. 207, 305–321 (2003).
Peipp, M. & Valerius, T. Bispecific antibodies targeting cancer cells. Biochem. Soc. Trans. 30, 507–511 (2002).
Kontermann, R.E. Recombinant bispecific antibodies for cancer therapy. Acta Pharmacol. Sin. 26, 1–9 (2005).
Cheong, H.S., Chang, J.S., Park, J.M. & Byun, S.M. Affinity enhancement of bispecific antibody against two different epitopes in the same antigen. Biochem. Biophys. Res. Commun. 173, 795–800 (1990).
Madrenas, J. et al. Conversion of CTLA-4 from inhibitor to activator of T cells with a bispecific tandem single-chain Fv ligand. J. Immunol. 172, 5948–5956 (2004).
Bonardi, M.A. et al. Delivery of saporin to human B-cell lymphoma using bispecific antibody: targeting via CD22 but not CD19, CD37, or immunoglobulin results in efficient killing. Cancer Res. 53, 3015–3021 (1993).
Vasu, C., Gorla, S.R., Prabhakar, B.S. & Holterman, M.J. Targeted engagement of CTLA-4 prevents autoimmune thyroiditis. Int. Immunol. 15, 641–654 (2003).
Khaw, B.A., Rammohan, R. & Abu-Taha, A. Bispecific enzyme-linked signal-enhanced immunoassay with subattomole sensitivity. Assay Drug Dev. Technol. 3, 319–327 (2005).
Kaufman, R.J. et al. Coamplification and coexpression of human tissue-type plasminogen activator and murine dihydrofolate reductase sequences in Chinese hamster ovary cells. Mol. Cell. Biol. 5, 1750–1759 (1985).
D'Andrea, A. et al. Production of natural killer cell stimulatory factor (interleukin 12) by peripheral blood mononuclear cells. J. Exp. Med. 176, 1387–1398 (1992).
Konishi, K. et al. A simple and sensitive bioassay for the detection of human interleukin-18/interferon-gamma-inducing factor using human myelomonocytic KG-1 cells. J. Immunol. Methods 209, 187–191 (1997).
Joosten, L.A., Helsen, M.M. & van den Berg, W.B. Accelerated onset of collagen-induced arthritis by remote inflammation. Clin. Exp. Immunol. 97, 204–211 (1994).
Acknowledgements
We thank Gerald Carson, Yun Zhang, Liqiang Zhou, Randolph Huelsman, Limary Medina, Michelle Babineau, Shaona Fang, Wendy Gion, Cheryl Thibault, Baofu Ni, Adriana Bajardi-Taccioli, and Elizabeth O'Connor of Abbott Bioresearch Center, for their technical contributions; Jochen Salfeld, Trudi Veldman, Andrew Goodearl, Lisa Olson, Lisa Schopf, Susan Lacy, Robert Hickman, Catherine Tripp, and Peter Isakson of Abbott Bioresearch Center for their support. We also acknowledge, with their permission, Lori Lush, PharmD, of JK Associates, Inc., and Michael Nissen, ELS, of Abbott Laboratories, for their editing and formatting assistance in the development of this manuscript. Type II porcine collagen (lyophilized) was obtained from Marie Griffiths at the University of Utah.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
This study was supported by Abbott Laboratories, Abbott Park, Illinois. The authors are employees of Abbott Laboratories and may own Abbott stock or stock options.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Tables 1–3 (PDF 400 kb)
Rights and permissions
About this article
Cite this article
Wu, C., Ying, H., Grinnell, C. et al. Simultaneous targeting of multiple disease mediators by a dual-variable-domain immunoglobulin. Nat Biotechnol 25, 1290–1297 (2007). https://doi.org/10.1038/nbt1345
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1345
This article is cited by
-
Challenges and strategies for next-generation bispecific antibody-based antitumor therapeutics
Cellular & Molecular Immunology (2020)
-
A novel tetravalent bispecific antibody targeting programmed death 1 and tyrosine-protein kinase Met for treatment of gastric cancer
Investigational New Drugs (2019)
-
Potent and conditional redirected T cell killing of tumor cells using Half DVD-Ig
Protein & Cell (2018)
-
Current progress in innovative engineered antibodies
Protein & Cell (2018)
-
Expanding the Boundaries of Biotherapeutics with Bispecific Antibodies
BioDrugs (2018)